- Title
-
Small heat shock proteins are necessary for heart migration and laterality determination in zebrafish
- Authors
- Lahvic, J.L., Ji, Y., Marin, P., Zuflacht, J.P., Springel, M.W., Wosen, J.E., Davis, L., Hutson, L.D., Amack, J.D., and Marvin, M.J.
- Source
- Full text @ Dev. Biol.
|
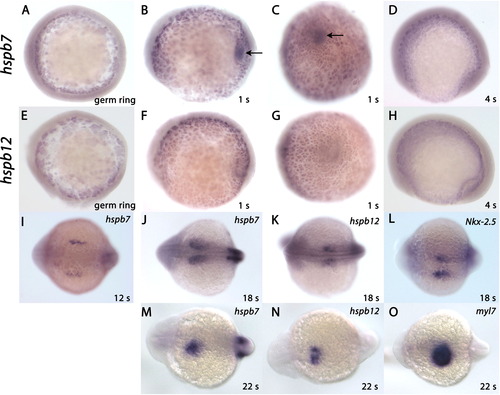
Expression patterns of hspb7 and hspb12 during the first 22 h of development. (A) hspb7 was first detected by in situ hybridization at 5.7 hpf in the yolk syncitial layer (YSL) (B) hspb7 expression continued in the YSL and was transiently expressed in the forming Kupffer′s vesicle (KV) at the tailbud-1 somite stage. (C) Posterior view of hspb7 expression in KV (arrow) at tailbud stage (dorsal to the left). (D) hspb7 disappeared from KV (arrow) by the four somite stage but remained in the YSL. (E) hspb12 was similarly expressed in the YSL at 5.7 hpf. (F) hspb12 was expressed in the YSL at 1-somite stage, but was not detected within KV. (G) Posterior view showing that hspb12 hybridization was not visible in KV. The surface of the YSL is depressed beneath the tailbud. This causes an edge-on view of the YSL surface, but the apparent staining was not visible at other angles. (H) hspb12 was absent from KV in four somite stage embryos, but YSL expression continued until approximately the 10 somite stage. (I) hspb7 was expressed in the bilateral heart fields beginning at the 12 somite stage. (J) hspb7 in the heart field and somites at the 18 somite stage. (K) hspb12 expression began in the bilateral cardiac precursors at the 18 somite stage. (L) Nkx-2.5 is expressed in ventricular precursors and corresponds to the expression sites of hspb7 and hspb12 at 18 somites. (M) hspb7 and (N) hspb12 were expressed in the fused heart fields as the leftward jog of the primitive heart tube begins at 21 somites. (O) Expression of myl7 (cmlc2) at 21-22 somites overlapped hspb7 and hspb12 hybridization. EXPRESSION / LABELING:
|
|
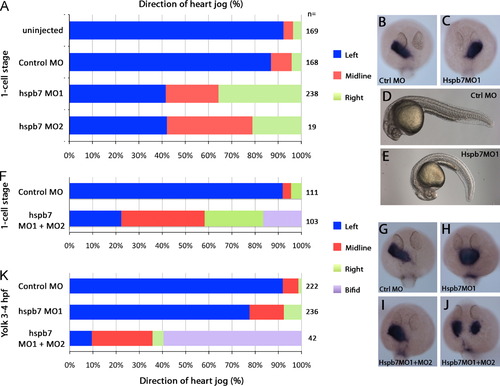
Heart jog direction was randomized by hspb7 knockdown. (A) Two splice-blocking morpholinos that target hspb7 both significantly increased right and midline heart jogs at the expense of leftward heart jogs (Χ2 test, p<0.001). (B, C) In situ hybridization of myl7 (cmlc2) in embryos injected with hspb7MO1. (B) Left jog and (C) right jog. (D) Live embryo at 26 hpf, injected with control MO. (E) Live embryo at 26 hpf, injected with hspb7MO1, showing characteristic downward curl of the body. (F) The combination of hspb7MO1 (0.25 pmole) and hspb7MO2 (0.17 pmole) prevented heart field fusion when targeted to the whole embryo at 1-4 cell stages. (G-I) In situ hybridization for myl7 (cmlc2). (G) Embryo injected with control MO at 1 cell stage had normal leftward heart jogs; the anterior boundary of the heart is midway across the eye at 26 hpf. (H) Midline heart in embryo injected with hspb7MO1+MO2 at 1 cell stage, showing slowed anterior migration, with the anterior boundary of the heart at the posterior end of the eye. (I) hspb7MO1+MO2 injected embryo, with normal leftward jog but delayed anterior migration and wide heart. (J) hspb7MO1+MO2 injected embryo, with cardia bifida. (K) Yolk injection between 3 and 4 hpf of control MO produced a small number of midline hearts; hspb7MO1 (0.5 pmole) induced midline hearts; hspb7MO1 (0.25 pmole) and hspb7MO2 (0.25 pmole) induced cardia bifida and midline hearts. |
|
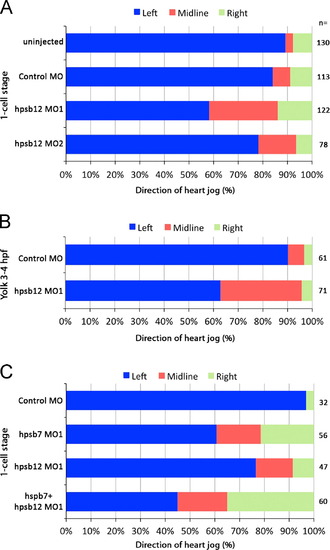
Heart jog direction was randomized by hspb12 knockdown (A) Two morpholinos that target hspb12 significantly increased right and midline heart jogs at the expense of leftward jogs compared to control MO (Χ2 test; p<0.001 for hspb12MO1 and p<0.05 for hspb12MO2). (B) Yolk injections of hspb12MO1 increased midline hearts but not rightward jogs. Combinations of both hspb12 MOs were lethal. (C) Combined hspb7MO1 and hspb12MO1 (0.25 pmol individually or in combination) injected at the 1-cell stage increased midline hearts and rightward jogs significantly compared to control (Χ2 test; all p<0.001). Combined hspb7MO1+hspb12MO1 (0.25 pmol each) gave significantly more abnormal jogs than 0.25 pmol hspb7MO1 alone (Χ2 test; p<0.05). PHENOTYPE:
|
|
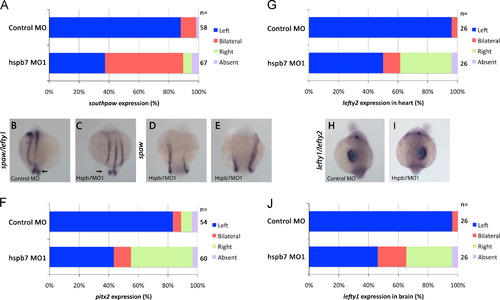
hspb7 was necessary for the expression of laterality genes in the left lateral plate mesoderm (LPM). (A) Embryos injected with 0.5 pmol control MO had primarily left-sided expression of spaw in the lateral plate mesoderm. In contrast, hspb7MO1 induced bilateral spaw expression (Χ2 test; p<0.001). (B-E) 14-18 somite stage embryos. (B) Embryos injected with control MO had normal spaw and lefty1 expression in left LPM and notochord, respectively. In the tail, bilateral peri-KV spaw domains were present (arrow). (C) hspb7MO1-injected embryos expressed spaw bilaterally. lefty1 in the notochord and peri-KV expression of spaw were not disturbed. (D, E) hspb7MO1-injected embryo with bilateral spaw with (D) left side leading leading and (E) right side leading. (F) hspb7MO1 randomized pitx2 expression (Χ2 test; p<0.001), with largely left or right sided expression and a small fraction of bilateral expression. (G) lefty2 expression in the left heart field was randomized or made bilateral by hspb7MO1 injection (Χ2 test; p<0.001). (H) embryos injected with control MO express lefty1 and lefty2 on the left side of the heart and brain at 22?24 somite stages. (I) hspb7MO1 injected embryos expressing lefty1 and lefty2 on the right. (J) lefty1 expression in brain was randomized by hspb7MO1 injection (Χ2 test; p<0.001). EXPRESSION / LABELING:
PHENOTYPE:
|
|
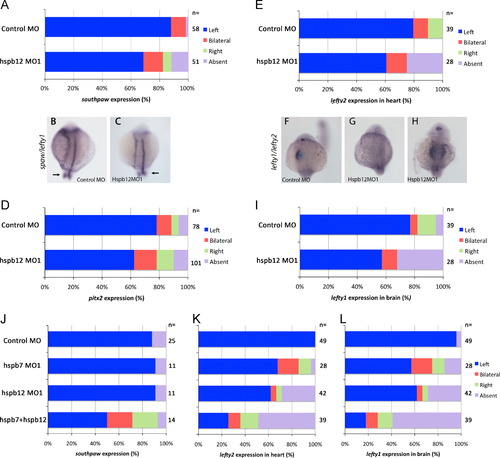
hspb12 knockdown randomized spaw, pitx2, lefty2 and lefty1. (A) 0.5 pmol hspb12 MO1 increased bilateral spaw expression compared to control MO (Χ2 test; p<0.001). (B, C) 14-18 somite embryos. (B) Control MO injected embryo with left sided spaw and midline lefty1 expression. (C) hspb12MO1-injected embryo with normal spaw and lefty1 expression. (D) Hspb12MO1 randomized pitx2 expression (Χ2 test; p<0.001). (E) hspb12 MO1 injection did not induce ectopic lefty2, but increased the number of embryos lacking lefty2 expression (Χ2 test; p<0.001). (F-H) lefty1 and lefty2 in situ hybridization at 22-24 somites. (F) Control MO with normal lefty1 and lefty2 in left brain and heart field respectively. (G) In this embryo, Hspb12MO1 suppressed normal lefty1 and lefty2. (H) In another embryo, Hspb12MO1 induced bilateral lefty1 and lefty2. lefty1 and lefty2 were never observed exclusively on the right. (I) Hspb12MO1 injection caused bilateral or absent lefty1 expression in the brain (Χ2 test; p<0.001). (J) Injection of 0.25 pmole of hspb7MO1 or hspb12MO1 individually did not affect left-sided spaw expression in the LPM, but injection of both MO together induced bilateral and right-sided spaw. (K) A dose of 0.25 pmole of hspb12MO1 or hspb7MO1 individually perturbed expression of lefty2 in heart, while the combination of 0.25 pmole each of hspb7MO1 and hspb12MO1 induced an even greater degree of abnormal expression. (L) Similarly, 0.25 pmole of hspb7MO1 or hspb12 MO1 induce a similar alteration in the expression of lefty1 in the brain, while the combination induces further abnormal expression. EXPRESSION / LABELING:
PHENOTYPE:
|
|
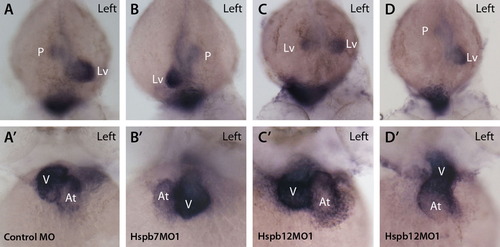
Concordance of asymmetry between heart and gut in hspb7 and hspb12 knockdown embryos. (A-D) Dorsal view, rostral toward bottom, left side as indicated in (A). Expression of foxa3 is strong in liver (Lv) and weaker in gut and pancreatic bud (P) at 48 hpf. (A′-D′) myl7 (cmlc2) is strongly expressed in ventricle (V) and more weakly in atrium (At) at 48 hpf. Anterior view, left side as indicated in (A). (A, A′) Control MO-injected embryo demonstrated the normal position of atrium to the left of the embryo′s midline and of the ventricle on the right (dextral loop). foxa3 is concentrated in the normal, left-sided liver and weaker in the pancreatic bud in control MO-injected embryo. (B, B′) hspb7 morphant with right-sided liver and ventricle. (C, C′) hspb12MO1-injected embryo with bilateral liver but normal position of the atrium and ventricle. (D, D′) hspb12MO1-injected embryo with left-sided liver and a heart that failed to jog or loop (midline heart). |
|
hspb7 knockdown disrupts cilia motility and asymmetric flow in Kupffer′s vesicle. (A) Analysis of cilia motility revealed that many cilia in hspb7 MO embryos were immotile or beat abnormally relative to controls. (B-C) Maximum projections of fluorescent bead movements superimposed on images of KV. Beads moved in a counterclockwise direction in control MO embryos (n=7/7 embryos; B), whereas this directional flow was absent in most hspb7 MO embryos (n=6/9 embryos; C). (D-F) Transmission electron micrographs showing: (D) transverse section of cilium from embryo injected with control MO showing 9+2 configuration of microtubules, (E) transverse section showing 9+0 configuration of microtubules in a cilium from embryo injected with hspb7 MO1+MO2, and (F) transverse section shows the abnormal central microtubule configuration in a cilium from embryo injected with hspb7 MO1. Arrows: outer dynein arms. PHENOTYPE:
|
Reprinted from Developmental Biology, 384(2), Lahvic, J.L., Ji, Y., Marin, P., Zuflacht, J.P., Springel, M.W., Wosen, J.E., Davis, L., Hutson, L.D., Amack, J.D., and Marvin, M.J., Small heat shock proteins are necessary for heart migration and laterality determination in zebrafish, 166-180, Copyright (2013) with permission from Elsevier. Full text @ Dev. Biol.