- Title
-
EphrinB1/EphB3b Coordinate Bidirectional Epithelial-Mesenchymal Interactions Controlling Liver Morphogenesis and Laterality
- Authors
- Cayuso, J., Dzementsei, A., Fischer, J.C., Karemore, G., Caviglia, S., Bartholdson, J., Wright, G.J., Ober, E.A.
- Source
- Full text @ Dev. Cell
|
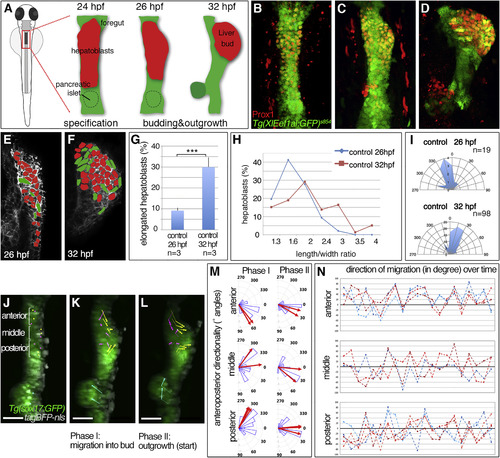
Hepatoblast Polarization Coincides with Liver Budding (A-D) Stages of liver budding: Schematic (A) and confocal projections of corresponding stages with Tg(XlEef1a1:GFP)s854 marking the endoderm and Prox1 hepatoblasts; ventral views (B-D). (E and F) EphrinB1 staining highlights cell shapes at the start of budding (E) and when a bud is apparent (F). Morphometric measurements were performed on serial coronal sections of the bud (E and F); elongated hepatoblasts (L/W ? 2) are shown in green. (G-I) Quantification of hepatoblast shape in control embryos at 26 and 32 hpf: (G) proportion of elongated cells per bud; SEs are shown, (H) L/W distribution for one representative bud; (I) orientation of elongated hepatoblasts with respect to the anteroposterior axis. (J-L) Time lapse of Tg(sox17:GFP)-positive foregut starting around 25 hpf (J) shows distinct hepatoblast movements during liver budding (K) and onset of outgrowth (L); dorsal views. TagBFP-nls (gray) marks nuclei for tracking of liver (yellow), gut (magenta), and pancreas progenitors (cyan). (M and N) Hepatoblasts from different anteroposterior positions migrate with distinct orientation. (M) Rose plots show the distribution of angular displacement with respect to the embryonic midline for 28 min intervals (blue sectors) and the angle of mean displacement per cell for the entire period (red arrow). (N) Line plots representing directionality of displacement over time show individual angular cell displacement for various liver (red hues) and gut progenitors (blue hues). Scale bars represent 40 ?m. ???p < 0.001. See also Figure S1; Movies S1, S2, and S4. |
|
Hepatoblasts Form Filopodia- and Lamellipodia-like Protrusions during Liver Budding (A-B?) Mosaic UAS:lyn-citrine or ubi:lyn-tdTomato expression shows EphrinB1+ hepatoblasts form lamellipodia (arrows in A?) and filopodia-like extensions (B and B?). Extensions (white arrowheads) connect the LPM (yellow arrowhead) and hepatoblasts (red arrowhead) over several cell diameters (B and B?). Dashed lines outline the LPM (white) and endoderm (green). (C and C?) Utrophin-GFP highlights actin in the cortical network and protrusions of hepatoblasts (arrowheads). Dashed lines delineate endoderm (green) and LPM (white). (D) Quantification of hepatoblast protrusions shows an increase of lamellipodia and decrease of filopodia-like extensions during budding. (E) Time lapse of migrating hepatoblasts during liver budding and early outgrowth; dorsal views. Membrane labeling with UAS:lyn-Citrine shows filopodia in the direction of outgrowth (white arrows) and toward the LPM (yellow arrows) and lamellipodia (red arrowheads); stills of Movie S3. Scale bars represent 30 ?m. ??p < 0.01, ???p < 0.001). See also Movie S3. |
|
Compromised Protrusion Formation Correlates with Liver Budding Defects (A-F) Latrunculin B (0.1 ?g/ml) treatment during liver budding (26-32 hpf) leads to significantly less hepatoblast protrusions (A-C; DMSO clones, n = 7; Lat B clones, n = 9, N = 1), and a significantly longer Prox1 domain (bracket) (D?F; DMSO, n = 23; Lat B, n = 13, N = 2). (G-L) Hepatoblasts expressing Cdc42T17N-GFP during liver budding (26?32 hpf) form significantly less protrusions compared to controls and Cdc42-GFP (G-I; control clones, n = 10; Cdc42-GFP clones, n = 16; Cdc42T17N-GFP clones, n = 18, N = 1) and a significantly longer EphrinB1 domain (bracket) (J?L; control, n = 7; Cdc42-GFP, n = 18; Cdc42T17N-GFP, n = 16, N = 2). N indicates the number of experiments. ?p < 0.05, ??p < 0.01, ???p < 0.001. |
|
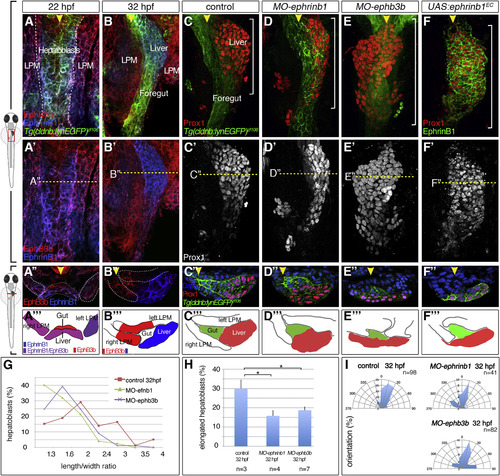
Complementary EphrinB1 and EphB3b Expression Controls Hepatoblast Positioning and Cell-Shape Changes in Liver Bud Formation (A-F?) At 22 hpf, EphrinB1 and EphB3b expression largely overlaps in future hepatoblasts and the LPM (A-A?). Complementary expression of both factors coincides with the start of liver budding: EphrinB1 in hepatoblasts and EphB3b in the gut and LPM (B?B?). At 32 hpf, hepatoblasts are located more posteriorly and medially compared with controls in MO-ephrinb1 (C-D?), in MO-ephb3b (E-E?), and upon conditional UAS:ephrinb1EC expression (F-F?). (A-F?) ventral views of confocal projections, anterior to the top; (A??F?) transverse sections of the foregut, as indicated by the dashed line in (A?-F?), and matching schematics (A?-F?); yellow arrowheads specify the midline and white brackets the length of the Prox1 domain. (G?I) Cell shapes were determined with EphrinB1-staining at 32 hpf (see Figure 1F). Quantification of hepatoblast shape in control, MO-ephrinb1, and MO-ephb3b embryos: (G) L/W distribution for one representative bud; (H) proportion of elongated cells per bud; SEs are shown; and (I) orientation of elongated hepatoblasts with respect to the anteroposterior axis. ?p < 0.05. See also Figure S2. EXPRESSION / LABELING:
PHENOTYPE:
|
|
EphB3b Controls LPM Polarity and Asymmetric Movement (A-E?) EphrinB1 and EphB3b regulate LPM polarity in the foregut region. ?-ZO-1 staining in MO-ephb3b embryos reveals that junctions in the LPM form similar to controls at 22 hpf (A-B?); while at 32 hpf, ZO-1 is mislocalized (arrowheads) in MO-ephrinb1 and MO-ephb3b embryos (C-E?). Yellow arrowheads specify the midline and reveal impaired gut looping in MO-ephrinb1 and MO-ephb3b (C-E). (F-G?) casanova mutants exhibit ZO-1 localization defects in the LPM (arrowheads) at 32 hpf. (A-G?) Transverse sections at liver level, left side to the right; dashed lines delineate the endoderm (green) and LPM (yellow); (A?-G?) are magnifications of the areas indicated by a box in (A-G). EXPRESSION / LABELING:
PHENOTYPE:
|
|
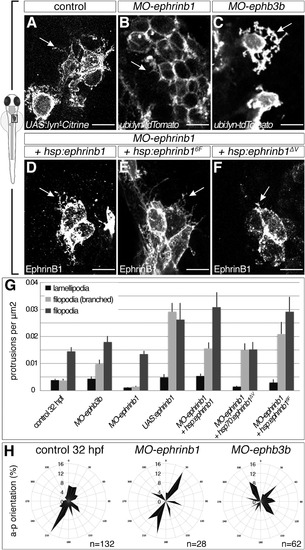
Opposing EphrinB1 and EphB3b Functions Control Formation of Hepatoblast Protrusions (A-G) EphrinB1 and EphB3b regulate hepatoblast protrusion number and morphology. Sparse labeling reveals fewer and shorter filopodia in MO-ephrinb1 hepatoblasts than in controls (A and B). Conversely, MO-ephb3b hepatoblasts show more complex, branched extensions (C). In contrast to EphrinB1 and EphrinB16F (D and E), EphrinB1?V fails to rescue extension formation in MO-ephrinb1 (F). Arrows indicate representative protrusions (A-F). Quantification of hepatoblast protrusion types in various conditions; comparative p values are shown in Table S1 (G). (H) Hepatoblast lamellipodia orientation is randomized in MO-ephb3b. (A-C) lyn-Citrine and lyn-Tomato outline hepatoblasts, (D-F) EphrinB1 staining highlights overexpression of different forms of EphrinB1, (A-F) ventral projections, anterior to the top. SEs are shown. Scale bars represent 10 ?m. See also Figure S3;Table S1. PHENOTYPE:
|
|
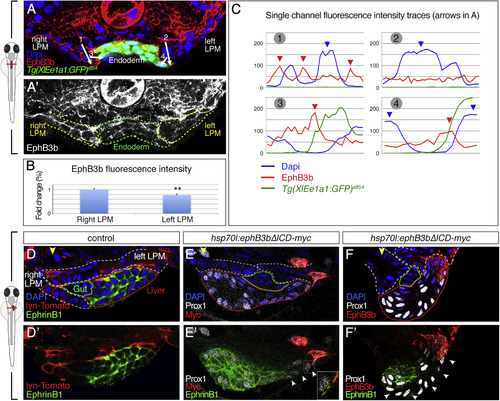
Asymmetric EphB3b Can Exert Repulsive Activity during Liver Budding (A-C) EphB3b expression is higher on the right than the left LPM at 24 hpf (A and A?). Quantification of EphB3b expression measuring overall fluorescent intensity (B) and intensity profile (C). Fluorescent intensity profiles show high EphB3b at cell membranes (red arrowheads) of the right but not left LPM; numbered arrows in (A) indicate the position of profiles. The right LPM-hepatoblast interface (profile 3) shows highest EphB3b expression. High DAPI levels indicate nuclei position (blue arrowheads). (D-F) Ectopic EphB3b?ICD expression alters hepatoblast position. Compared with mosaic lyn-Tomato (D), mosaic EphB3b?ICD expression on the left LPM (E) or left LPM and hepatoblasts (F) at 26 hpf causes positioning of Prox1+ hepatoblasts away from the clone at 32 hpf. EphrinB1 is absent from membranes in 3-7 hepatoblasts next to EphB3b?ICD clones (white arrowheads), without altering Prox1 expression (E? and F?). (E?) Inset shows an EphrinB1 signal in EphB3b?ICD protrusion, indicating direct cell interaction. (A, A? and D?F?) Transverse sections at liver level, left side to the right; yellow arrowhead specifies the midline; lines delineate the left and right LPM (white), gut (green), hepatoblasts (red). ??p < 0.01. See also Figures S4 and S5. |
|
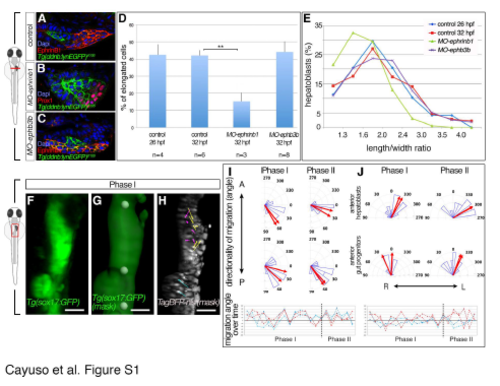
Hepatoblasts undergo dynamic cell shape changes during liver budding. Related to Figure 1. (A-C) Cell morphology was determined with EphrinB1- or Tg(-0.8cldnb:lynEGFP)zf106-expression outlining the hepatoblasts in control (A), MO-ephrinB1 (B) and MO-ephB3b embryos (C); transverse sections with liver oriented to the right. Morphometric measurements were carried out on serial sections through the bud. (D,E) Quantification of hepatoblast characteristics during liver budding in control, MO-ephrinB1 and MO-ephB3b embryos: (D) population of elongated cells (length/width>2) per bud and (E) hepatoblast distribution according to length/width ratio; only MO-ephrinB1M exhibit a significantly decreased length/width ratio). Error bars represent standard error of the mean; ** = p<0.01. (F-H) Processing steps of 4D dataset of a Tg(sox17:GFP)z99 embryo injected with tagBFP-nls mRNA capturing liver budding (see Figure 1J-N, movie S1). sox17:GFP expression (F) marks the endoderm and was used to generate a foregut mask (G) which was subsequently applied to the TagBFP-nls channel (H). Three manually selected anatomical landmarks in 3D were used for drift correction (G, round spheres). Scale bar - 40?m. (I,J) Rose plots represent distribution of angular displacement (blue sectors) for hepatoblasts and gut progenitors from the same anterior position and angle of mean displacement per cell for the entire period (red arrow). Displacements were recorded every 28 min with respect to the embryonic midline and left-right distribution. Line plots representing directionality of displacement over time reveal differences in individual angular cell displacement for various hepatoblasts (red hues) and gut progenitors (blue hues). Data are derived from movie S1 (see also Figure 1 J-N). |
|
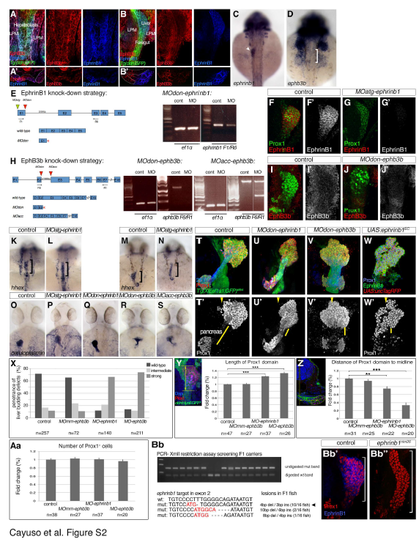
EphrinB1 and EphB3b expression and validation of knock down and knock out strategies. Related to Figure 4. (A-B') Single channel views of immunostainings for EphrinB1 and EphB3b at 22 and 32 hpf shown in Figure 4A-B'''. (C,D) ephrinb1and ephb3b are expressed in the foregut area. At 30 hpf, ephrinb1 is expressed in the liver (arrowhead) and ephb3b in hepatic area (bracket). (E) Schematic representation of ephrinB1 knock-down strategy, with relevant MOs indicated. RT-PCR analysis of MOdon-ephrinB1 embryos at 28 hpf shows a strong knock-down. In addition, a stop codon in the intronic sequence terminates the resulting protein 3 bps after the exon1-intron boundary. Both MO-ephrinB1 produce similar phenotypes (see O-Q), MOatg-ephrinB1 were used for all experiments. (F-G') EphrinB1 expression is abolished or barely detectable in MOatg-ephrinB1 embryos at 32 hpf. (H) Schematic representation of ephB3b knock-down strategy, with relevant MOs indicated. ephB3b maps to linkage group 22 and is partly encoded by EST CT 623350. By 5'RACE, we identified a 2970 bp coding transcript encoded by 16 exons containing all motifs conserved between EphB-receptors, including a N-terminal signal peptide and Ephrin-binding domain. In addition, we found an alternative transcript missing the first two exons, replaced by a N-terminal extension of exon 3 and lacking a signal peptide and a clear transcriptional start site. Its functional significance is unclear and it was not targeted by our knock-down strategy. RT-PCR analysis of MOdon-ephB3b embryos at 28 hpf shows a complete knock-down, indicated by a complete loss of the Ephrin-binding domain-containing exon3. In addition, a stop codon in the intronic sequence would terminate the resulting protein 108 bps after the exon 2- intron boundary. RT-PCR analysis of MOacc-ephB3b embryos at 28 hpf reveals a partial knock-down. This is consistent with the loss of the Ephrin-binding domain-containing exon 3 in a fraction of the transcripts, as indicated by the appearance of a 500bp smaller ectopic band in the MOacc-ephB3b lane compared to controls; control and representative MOacc-ephB3b sample are from different parts of the same gel, as indicated by the separating line. Although, both MO-ephB3b produce similar phenotypes (see O,R,S), the MOdon-ephB3b knock-down is consistently more complete and was used for all experiments. (I-J') EphB3b expression is abolished or barely detectable in MOdon-ephB3b embryos at 32 hpf. (K-N) hhex mRNA expression at 25 hpf reveals that the liver anlage (bracket) forms in MOephrinB1 embryos (L) similar to controls (G), while impaired hepatoblast (bracket) movement into the liver bud in MO-ephrinB1 embryos is apparent at 28 hpf (M,N). (O-S) Two independent MOs targeting ephrinB1 or ephB3b produce equivalent phenotypes, with hepatoblasts positioned more posterior and medial (P,Q and R,S, respectively) compared to controls at 48 hpf (O). Asymmetric hepatoblast positioning is generally more severely disrupted in MO-ephB3b than MO-ephrinB1 embryos. (T-W') Confocal projections reveal liver morphogenesis defects in MO-ephrinB1 (U,U'), MO-ephB3b (V,V'), as well as embryos expressing EphrinB1EC in the context of the entire foregut domain (W,W'). Tg(XlEef1a1:GFP)s854 marks the foregut endoderm, 2F11 the hepatopancreatic ducts and Prox1 the developing liver and pancreas at 54 hpf. Upon loss of EphrinB1 or EphB3b functions hepatoblasts are ectopically positioned in the hepatopancreatic duct domain compared to controls, as indicated by shorter yellow bars, and across the midline (arrowheads; T'-V'). Similar defects are observed upon conditional expression of EphrinB1EC (W,W'). (C,D, K-S) dorsal views, (A-B, F-G', I-J',T-W') confocal projections of ventral views; all anterior to the top. (A?,B?) confocal images of transverse sections, left is oriented to the right. (X-Aa) Quantification and morphometric analyses of liver bud development in MO-ephrinB1 and MOephB3b embryos at 32 hpf. (X) Graph showing the proportion of embryos exhibiting hepatoblast positioning defects in un-injected and MOmm-ephB3b controls, and following EphrinB1- or EphB3b knockdown. In whole-mount preparations the length (Y) and width of the Prox1-domain and its proximity to the embryonic midline (Z) were used to classify the liver budding defects into three categories. (Y) Morphometric measurements showed a significant increase in Prox1-domain length (yellow double arrow) in MO-ephrinB1 and MO-ephB3b embryos compared to controls. (Z) Morphometric measurements revealed a significant reduction in asymmetric displacement of the Prox1-domain from the midline in MO-ephrinB1 and MO-ephB3b embryos compared to controls. Specifically, the distance of the centre of the Prox1-domain to the embryonic midline was determined in transverse sections (yellow double arrow). (Aa) EphrinB1 or EphB3b knockdown does not significantly change the number of Prox1-expressing hepatoblasts; p-values, for all conditions >0.57. (c,e,f) Standard errors are shown; asterisks indicate the p-value for each condition (**, p<0.01; ***, p<0.001). (Bb-Bb'') Stable genetic ephrinb1 mutant was generated using Crispr/Cas9 genome editing. F1 fish were screened for germline transmission of lesions by XmlI digest of amplified target DNA; undigested PCR bands indicate transmission of genetic lesion. Sequencing revealed that F1 fish carry different indels. Intercrosses of carriers with a 4bp deletion and 3bp insertion show a complete loss of ?-EphrinB1 staining in homozygous carriers (Bb'') compared to sibling controls (Bb'). A stable line, ephrinb1nim26, was generated from these carriers. |
|
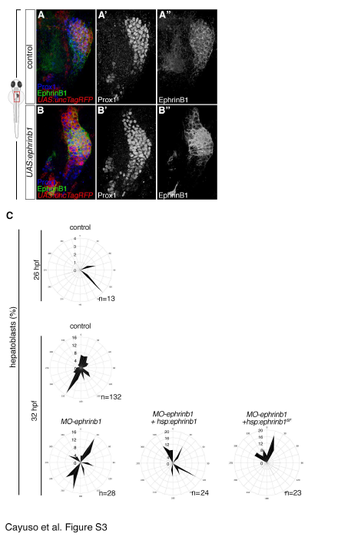
Altering EphrinB1 and EphB3b levels leads to defects in lamellipodia orientation and hepatoblast positioning. Related to Figure 6. (A-B?) Conditional expression of UAS:ephrinB1 causes a posterior accumulation of hepatoblasts and morphological defects of the liver domain (B-B''). (C) Hepatoblast lamellipodia are oriented with respect to the anteroposterior axis. Expression of EphrinB1 and EphrinB16F randomises lamellipodia orientation in comparison to control and MO-ephrinB1 embryos, and similar to compromised EphB3b function (see Figure 6H). |
|
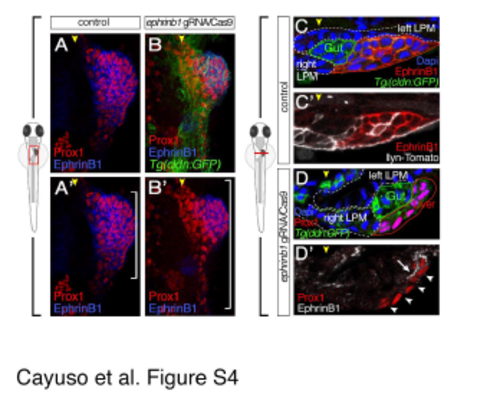
Mosaic inactivation of EphrinB1 by Crispr/Cas9 editing, leads to defects in hepatoblast positioning and indicate long-distance contacts. Related to Figure 7. (A-B') at 32 hpf, EphrinB1+-hepatoblasts transiently expressing lyn-Tomato distribute randomly throughout the bud (A,A?), while EphrinB1+ hepatoblasts (blue) aggregate mostly anterior and away from the right LPM and hepatoblasts lacking EphrinB1 form an elongated Prox1 domain (B,B?) closer to the midline (yellow arrowhead). White brackets indicate the length of the Prox1-domain. Confocal projections of ventral views; anterior to the top. (C-D') EphrinB1+ hepatoblasts (white) aggregate mostly away from the right LPM, often with a gap of 1-6 hepatoblasts lacking EphrinB1 (white arrowheads) indicating long-distance cell-cell contacts sensing repulsive EphB3b from the LPM. Consistently, hepatoblast protrusions (arrow) are also EphrinB1+ (D'). Transverse sections with dorsal to the top, and left to the right. |
Reprinted from Developmental Cell, 39, Cayuso, J., Dzementsei, A., Fischer, J.C., Karemore, G., Caviglia, S., Bartholdson, J., Wright, G.J., Ober, E.A., EphrinB1/EphB3b Coordinate Bidirectional Epithelial-Mesenchymal Interactions Controlling Liver Morphogenesis and Laterality, 316-328, Copyright (2016) with permission from Elsevier. Full text @ Dev. Cell