- Title
-
The Integrator subunits function in hematopoiesis by modulating Smad/BMP signaling
- Authors
- Tao, S., Cai, Y., and Sampath, K.
- Source
- Full text @ Development
|
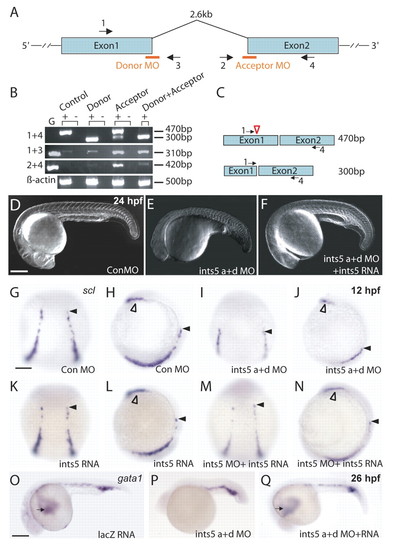
Ints5 functions during hematopoiesis. (A) Schematic representation of the ints5 genomic locus; orange bars indicate target sites of the donor and acceptor morpholinos (MOs); numbered black arrows show the position of the primers used in RT-PCRs to examine splicing of ints5 RNA. (B) RT-PCR to detect splicing of ints5 and control β-actin (encoded by bactin1). RNA from sphere stage (4 hours post fertilization, hpf) embryos injected with control or ints5 donor and acceptor morpholinos at the 1-cell stage. Primers used (left) and transcript sizes (right) are indicated. G, genomic DNA. (C) Schematic representation of correctly spliced (upper) and aberrantly spliced (lower) ints5 transcripts. The size of bands amplified by primer pair 1 and 4 is indicated on the right. Red open arrowhead indicates the position of a cryptic splice site in exon 1. (D-F) DIC images of live embryos at 24 hpf injected with control morpholinos (D), ints5 acceptor and donor (a+d) morpholinos (E) or co-injected with ints5 morpholinos and ints5 RNA (F). (G-Q) Whole-mount in situ hybridization to detect expression of scl (G-N) at 12 hpf and gata1 (O-Q) at 26 hpf shows reduced expression of both genes in ints5 morphants (I,J,P), in comparison to control morpholino- or lacZ RNA-injected embryos (G,H,O). Black arrowheads indicate the anterior limit of scl expression in the intermediate cell mass (ICM); open arrowheads indicate scl expression in the rostral blood island (RBI); black arrows indicate anterior gata1 expression, which represents the circulating blood cells. Co-injection of ints5 RNA can restore scl and gata1 expression in ints5 morphants (M,N,Q), whereas embryos injected with ints5 RNA alone show normal scl expression at 12 hpf (K,L). D-F,O-Q show lateral views of embryos with anterior to the left; G,I,K,M show dorsal views of embryos with anterior at the top; H,J,L,N show lateral views of embryos with dorsal to the right. Scale bars: 250 μm in D,O; 50 μm in G. EXPRESSION / LABELING:
PHENOTYPE:
|
|
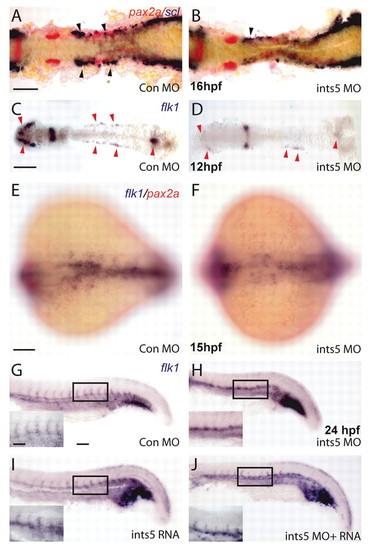
Ints5 knockdown affects hematopoietic progenitors but not pronephric cells. (A-F) In situ hybridization to detect expression of scl (purple) in hematopoietic cells, pax2a (red) in the pronephric cells and flk1 (purple) in endothelial cells. (A,B) scl and pax2a expression in control (A) and ints5 morphants (B) at 16 hpf. Black arrowheads indicate the anterior scl expression in the ICM, and its reduction in ints5 morphants; pax2a expression is unaffected. (C-F) Expression of flk1 in control (C,E) and ints5 morphants (D,F) at 12 hpf (C,D) and 15 hpf (E,F). Red arrowheads indicate flk1 expression, which is much reduced in ints5 morphants at an early stage (C,D) but recovers later in development (E,F). (G-J) Expression of flk1 in injected embryos at 24 hpf. Insets show enlarged views of the boxed areas. Inter-segmental flk1 expression is not detected in ints5 morphants (H), in comparison to control embryos (G) and ints5 RNA-injected embryos (I). Expression of flk1 in inter-segmental vessels is rescued by co-injection of ints5 RNA (J). A-D show dorsal views of flat-mounted embryos; E,F show dorsal views with anterior to the left; G-J show lateral views of the trunk with anterior to the left. Scale bars: 100 μm in A,C,E,G; 50 μm in inset in G. EXPRESSION / LABELING:
PHENOTYPE:
|
|
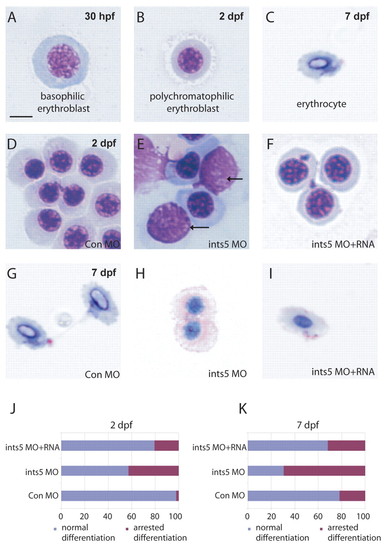
Ints5 is required for erythrocyte differentiation. (A-C) May-Grunwald Giemsa staining shows normal red blood cells (RBCs) at various stages of differentiation at the indicated embryonic stages. (D-F) RBCs in embryos injected with control morpholinos (D), ints5 morpholinos (E), or co-injected with ints5 RNA and morpholinos (F) at 2 dpf. Arrows indicate the RBCs arrested at the basophilic erythroblast stage (E). (G-I) RBCs in embryos injected with control morpholinos (G), ints5 morpholinos (H), or co-injected with ints5 RNA and morpholinos (I) at 7 dpf. (J,K) Histograms showing the percentage of RBCs with normal (blue) or arrested (magenta) differentiation in injected embryos at 2 dpf (J) and 7 dpf (K). Scale bar: 10 μm. PHENOTYPE:
|
|
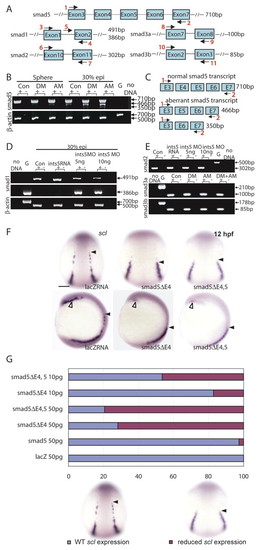
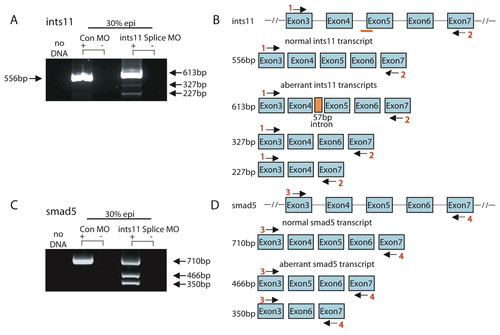
Knockdown of Ints5 perturbs splicing of smad1 and smad5 RNA. (A) Schematic representation of the smad5, smad1, smad2, smad3a and smad3b genomic loci. Numbered black arrows indicate the position of primers used in RT-PCRs to detect splicing. The sizes of the predicted products using various primer pairs are indicated on the right. (B) At both sphere and 30% epiboly stages, aberrantly spliced smad5 transcripts accumulate in ints5 donor morpholino (DM)- and acceptor mopholino (AM)-injected embryos. β-actin control transcripts are correctly spliced to yield a 500 bp product from cDNA in comparison to a 700 bp genomic DNA product, G. (C) Schematic representation to show the wild-type 710 bp smad5 transcript, and aberrant smad5 transcripts that lack exon 4 (466 bp product), or lack both exon 4 and exon 5 (350 bp product). (D) Unspliced smad1 (386 bp product with primer pair 4 and 5) and correctly spliced smad1 (491 bp with primer pair 3 and 4) transcripts in 30% epiboly ints5 morpholino-injected embryos. (E) Amplification of smad2, smad3a and smad3b with the primer pairs indicated in A shows correctly spliced products in ints5 morphants. Amplification with primers spanning other regions of these genes also did not show aberrant splicing (data not shown). (F,G) Overexpression of truncated smad5 transcripts causes hematopoiesis defects, similar to ints5 morphants. (F) Whole-mount in situ hybridization to detect scl in control embryos (left), and in embryos injected with smad5ΔE4 RNA (middle) or smad5ΔE4,5 RNA (right) at 12 hpf. Black arrowheads indicate the anterior limit of scl expression in the ICM; open arrowheads mark scl expression in the RBI. Upper panel shows dorsal views with anterior to the top; lower panel shows lateral views with dorsal to the right. (G) Histogram showing the percentage of injected embryos with wild-type (blue) or reduced (magenta) scl expression levels. Scale bar: 50 μm. PHENOTYPE:
|
|
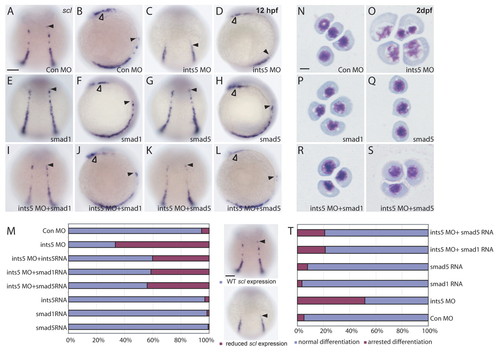
The hematopoiesis defects induced by Ints5 knockdown are rescued by smad1 and smad5 RNA. (A-L) Whole-mount in situ hybridization to detect scl expression in 12 hpf embryos injected with control morpholinos (A,B), ints5 morpholinos (C,D), smad1 RNA (E,F), smad5 RNA (G,H), ints5 morpholinos with smad1 RNA (I,J), or ints5 morpholinos with smad5 RNA (K,L). Black arrowheads indicate the anterior limit of scl expression in the ICM; open arrowheads indicate scl expression in the RBI. A,C,E,G,I,K show dorsal views of embryos with anterior to the top; B,D,F,H,J,L show lateral views of embryos with dorsal to the right. (M) Histogram showing the percentage of injected embryos with wild-type-like (blue) or reduced (magenta) scl expression. The total number of embryos for each staining is >180, and all experiments were carried out three independent times (see Table S1 in the supplementary material). (N-S) May-Grunwald Giemsa staining to show RBC differentiation in embryos injected with control morpholinos (N), ints5 morpholinos (O), smad1 RNA (P), smad5 RNA (Q), ints5 morpholinos with smad1 RNA (R), or ints5 morpholinos with smad5 RNA (S) at 2 dpf. (T) Histogram showing the percentage of RBCs with normal (blue) or arrested (magenta) differentiation in injected embryos at 2 dpf. The total number of red blood cells counted for each sample is >200, and all experiments were carried out three independent times. Scale bars: 50 μm in A,M; 10 μm in N. EXPRESSION / LABELING:
PHENOTYPE:
|
|
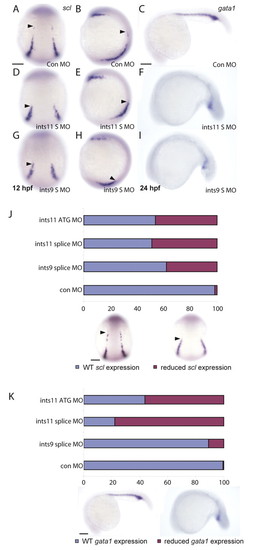
Multiple subunits of the Integrator complex regulate primitive hematopoiesis. (A-I) Whole-mount in situ hybridization to detect scl (A,B,D,E,G,H) and gata1 (C,F,I) expression in embryo injected with control morpholinos (A-C), ints11 splice-junction morpholinos (S MO, D-F) and ints9 splice-junction morpholinos (G-I). Black arrowhead indicates the anterior limit of scl expression in the ICM. The ints9 and ints11 morphants show substantially reduced scl and gata1 expression in comparison to control morphants. (J,K) Histograms showing the percentage of injected embryos with wild-type (blue) or reduced (magenta) scl (J) and gata1 (K) expression levels. Scale bars: 50 μm in A,J; 250 μm in C,K. EXPRESSION / LABELING:
PHENOTYPE:
|
|
Knockdown of ints11 perturbs smad5 splicing. (A)RT-PCR analysis shows that the ints11 splice-junction morpholino efficiently blocks the correct splicing of ints11 RNA, and several aberrant transcripts are detected. Transcripts are indicated by black arrows, with sizes indicated. (B) Schematic representation of the ints11 genomic locus (upper panel); schematic representation of normal or aberrant ints11 transcripts is shown in the lower panel. Numbered black arrows indicate the position of primers used in RT-PCRs to detect splicing. Orange bar shows the target site of the ints11 splice-site morpholino. (C)RT-PCR analysis shows that splicing of smad5 is impaired in ints11 morphants. (D) Schematic representation of the smad5 genomic locus (upper panel); the lower panel shows schematic representations of normal and aberrantly spliced smad5 transcripts. EXPRESSION / LABELING:
PHENOTYPE:
|
|
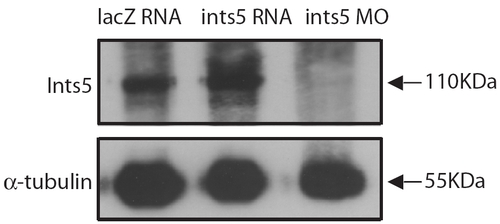
ints5 morpholinos abolish Ints5 protein in embryos. Western blots on extracts of embryos injected with lacZ RNA, ints5 RNA or ints5 morpholinos. Proteins of each sample were harvested from 20 injected embryos at 8 hpf. Ints5 proteins were detected by using rabbit anti-Ints5 antibody (upper panel); lower panel shows expression of α-Tubulin control detected using anti-α-Tubulin antibodies. |
|
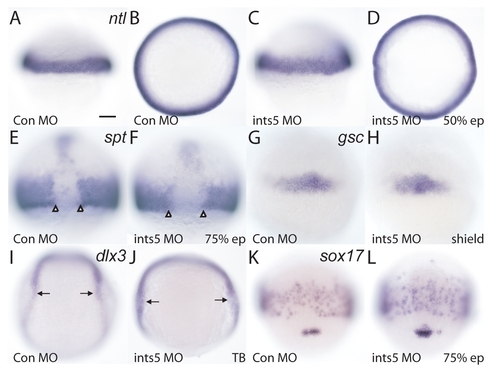
Knockdown of ints5 does not affect germ-layer specification. (A-H) Whole-mount in situ hybridization to detect expression of notail (ntl) (A-D) and spadetail (spt) (E,F) at 50% epiboly (5.3 hpf) and 75% epiboly (8 hpf), respectively, and goosecoid (gsc) (G,H) in shield-stage embryos (6 hpf), injected with ints5 morpholinos (C,D,F,H), or control morpholinos (A,B,E,G). Open arrowheads in E and F indicate the distance between spt-positive cells in the midline. (I-L) Expression of dlx3 (I,J) at the neural plate boundary in tailbud stage (TB, 10 hpf) embryos, and sox17 (K,L) in the endoderm of 75% epiboly embryos injected with ints5 morpholinos (J,L) or control morpholinos (I,K). Black arrows in I and J mark the distance between the boundary of dlx3 expression. A and C show lateral views of embryos with animal pole to the top; B and D show animal pole views; E-L show dorsal views of embryos with animal pole to the top. The total number of embryos for each marker was >50, from three independent injection experiments. Scale bar: 50 μm. |
|
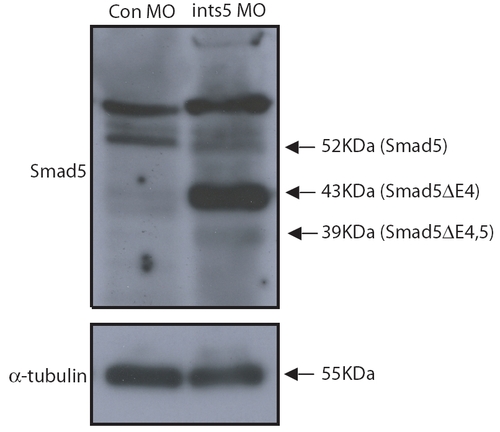
ints5 morpholinos lead to truncated Smad5 protein in embryos. Western blots on extracts of embryos injected with control or ints5 morpholinos. Proteins were harvested from 20 injected embryos at 8 hpf. Smad5 proteins were detected using rabbit anti-Smad5 polyclonal antibody (upper panel); the lower panel shows expression of α-Tubulin control detected with mouse monoclonal anti-α-Tubulin antibodies. The black arrows indicate the size of corresponding peptides. |
|
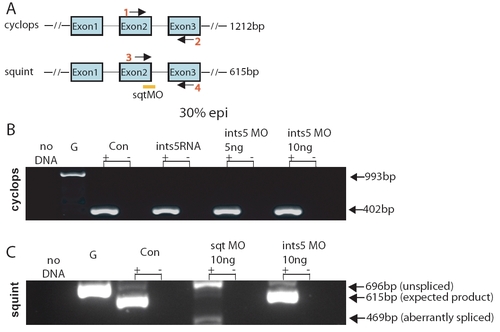
Knockdown of Ints5 does not affect splicing of cyclops and squint RNA. (A) Schematic representation of cyclops and squint genomic loci. Arrows indicate the position of primer pairs used in RT-PCRs to detect splicing. Orange bar indicates the position of the squint splice junction morpholino at the exon2-intron2 boundary (Gore et al., 2005). Expected sizes of the amplified products are shown on the right. At 30% epiboly, a 402 bp cyc product is detected from all cDNA templates (B), whereas a 993 bp band is amplified from genomic DNA. (C) Embryos injected with ints5 morpholinos or control morpholinos show the expected 615 bp squint product. By contrast, embryos injected with squint splice-junction morpholinos show either unspliced (696 bp) or aberrantly spliced (469 bp) squint transcripts. |
|
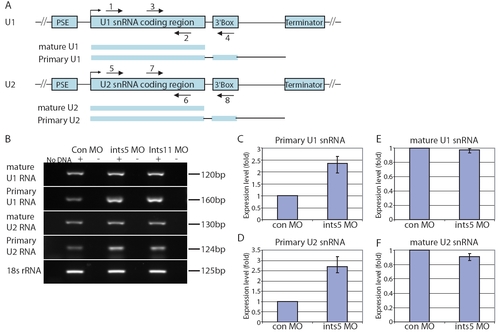
Knockdown of ints5 leads to the accumulation of unprocessed primary U1/U2 snRNAs. (A) Diagrams of U1 and U2 snRNA genes. Location of the proximal promoter elements (PSE), snRNA coding region, the 3′ end formation signals (3′ Box) and terminators are indicated in blue boxes. Schematic representation below shows mature and primary immature forms of U snRNA transcripts. Numbered arrows indicate the position of primers used to detect expression of the various U snRNAs. (B) Representative RT-PCRs to detect mature and primary U1 and U2 snRNAs at 8 hpf with the following primer pairs: mature U1, 1+2; primary U1, 3+4; mature U2, 5+6; primary U2, 7+8. Expression of 18s rRNA is shown as a control. (C-F) Histograms to show expression level of primary U1 (C), primary U2 (D), mature U1 (E) and mature U2 (F) transcripts in injected embryos. Expression levels were measured at 8 hpf by semi-quantitative RT-PCR, normalized with respect to the level of the 18s rRNA internal control, and shown as a fold change relative to control embryos. Histograms show data obtained from three independent injection experiments. |
|
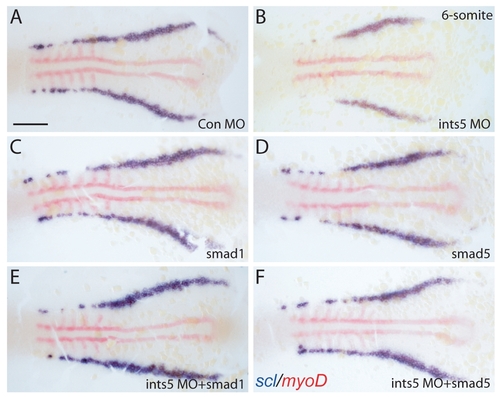
The hematopoiesis defects induced by ints5 knockdown are rescued by smad1 and smad5 RNA. (A-F) Whole-mount in situ hybridization to detect scl (purple) and myoD (red) expression in 6-somite stage embryos injected with control morpholinos (A), ints5 morpholinos (B), smad1 RNA (C), smad5 RNA (D), ints5 morpholinos with smad1 RNA (E), or ints5 morpholinos with smad5 RNA (F). Images show flat-mounted embryos with anterior to the left. For each marker, n>50 embryos, from three independent injection experiments. Scale bar: 100 μm. |
|
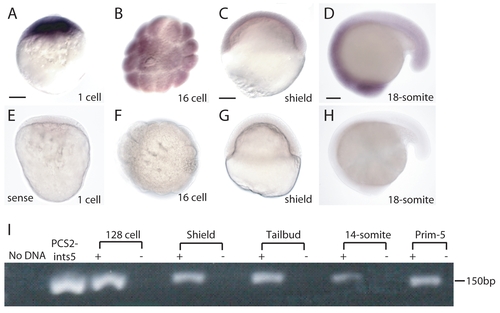
ints5 is expressed both maternally and zygotically in zebrafish embryos. Whole-mount in situ hybridization to detect ints5 expression in wild-type embryos at the indicated stages (A-D). (E-H) Embryos hybridized with control ints5 sense strand probe. A,C-E,G,H show lateral views of embryos; B,F show animal pole views. (I) RT-PCR to show ints5 RNA expression at the indicated embryonic stages. Scale bars: 50 μm in A,C; 100 μm in D. |
|
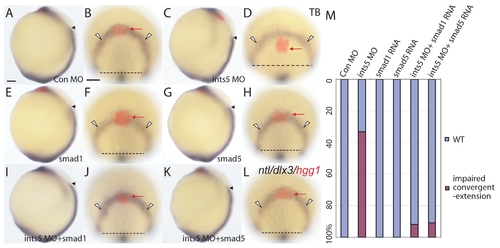
Convergent-extension defects in ints5 morphants are rescued by smad1 and smad5 RNA. (A-L) Whole-mount in situ hybridization to detect ntl, dlx3 and hgg1 (red) expression in tailbud (TB, 10 hpf) embryos injected with control morpholinos (A,B), ints5 morpholinos (C,D), smad1 RNA (E,F), smad5 RNA (G,H), ints5 morpholinos with smad1 RNA (I,J), or ints5 morpholinos with smad5 RNA (K,L). Black arrowheads indicate the anterior limit of ntl expression in the midline, and red arrows indicate hgg1 staining in the anterior prechordal plate mesendoderm cells. White arrowheads and dashed lines indicate the distance between the lateral limits of dlx3 expression in the neuroectoderm. A,C,E,G,I,K show lateral views of embryos with dorsal to the right; B,D,F,H,J,L show animal pole views of embryos. (M) Histogram showing percentage of injected embryos with wild-type (WT, blue) or impaired (magenta) cell movement marked by ntl/dlx3/hgg1 expression. n>50 embryos, from three independent injection experiments. Scale bars: 50 μm. |