- Title
-
Interaction of Wnt and caudal-related genes in zebrafish posterior body formation
- Authors
- Shimizu, T., Bae, Y.K., Muraoka, O., and Hibi, M.
- Source
- Full text @ Dev. Biol.
|
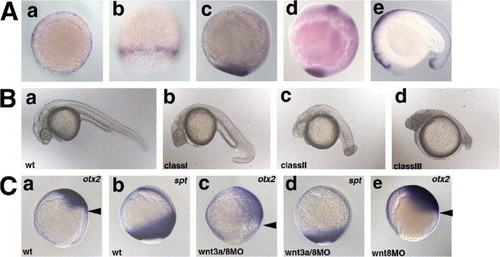
Redundant role of Wnt3a and Wnt8 in early embryogenesis. (A) Expression of wnt3a. a, shield stage; b, 80% epiboly stage; c, bud stage; d, 8-somite stage (13 hpf); e, 17-somite stage (17 hpf). a, animal pole view with dorsal to the right; b, dorsal view; c, d, lateral view with dorsal to the right; e, lateral view with anterior to the left. (B) Phenotype of the wnt3aMO- and/or wnt8MO-injected pharyngula-stage (24 hpf) embryos. The phenotypes of the injected embryos were classified into three categories. Class I embryos had a ventrally bent tail structure and a normal number of somites (30?34). Class II embryos had an enlarged head, and a short and twisted tail (number of the somites 20?30; average 27.4 (n = 13)). Class III embryos had an enlarged head and a truncated tail structure (less than twenty somites; average 13.7 (n = 8)). The percentage of embryos in each class is shown in Table 1. (C) Expression of the anterior neuroectoderm marker otx2 (a, c, e) and the paraxial mesodermal marker spadetail (spt) (b, d) at the 90% epiboly stage in wild-type embryos (wt, a, b), and embryos injected with both wnt3aMO and wnt8MOs (wnt8-1MO and wnt8-2MO, c, d) or wnt8MOs alone (e). Lateral views with dorsal to the right. Arrowheads (a, c, e) indicate the posterior limits of the otx2 expression. EXPRESSION / LABELING:
|
|
Interaction between the Wnt3a/8 signaling and the ventrally expressed homeobox genes vox, vent, and ved for restriction of the dorsal organizer domain. (A) Expression of organizer genes in the wnt3a/wnt8 morphant embryos. The embryos received 4.2 ng each of wnt3aMO, wnt8-1MO, and wnt8-2MO (wnt3a/8MO, wnt3a/wnt8 morphant embryos) and were fixed at the shield stage. The expression of goosecoid (gsc), floating head (flh), and chordino (din) was expanded ventrally in the wnt3a/wnt8 morphant embryos compared with wild-type embryos (wt) or embryos receiving an injection of wnt3aMO (4.2 ng) alone (data not shown). In contrast, the expression of dickkopf1 (dkk1) was strongly reduced in the wnt3a/wnt8 morphant embryos. a?c, e?f animal pole views with dorsal to the right. d,h, Dorsal views. (B) Expression of vox and ved in the wnt3a/wnt8 morphant embryos, and expression of wnt3a and wnt8 in the vox/vent/ved morphant embryos. The vox/vent/ved triple morphant embryos were prepared by injecting 0.3 ng each of voxMO, ventMO, and vedMO, as published previously (Shimizu et al., 2002). a, b, e, f, shield stage, animal pole views with dorsal to the right. c, d, g, h, 80% epiboly stage, lateral views with dorsal to the right. Arrowheads indicate the dorsal limit of the vox and ved expression. The expression of vox and ved was reduced on the dorsal side in the wnt3a/wnt8 morphant embryos. The expression of wnt3a and wnt8 in the blastoderm margin was reduced in the vox/vent/ved morphant embryos. (C) Ved is epistatic to the Wnt3a/Wnt8 signaling. ved RNA (100 pg) was injected alone (b) or with wnt3aMO and wnt8MOs (c). The co-injection of ved RNA suppressed the head enlargement but did not suppress the reduction in posterior body observed in the wnt3a/wnt8 morphant embryos. EXPRESSION / LABELING:
|
|
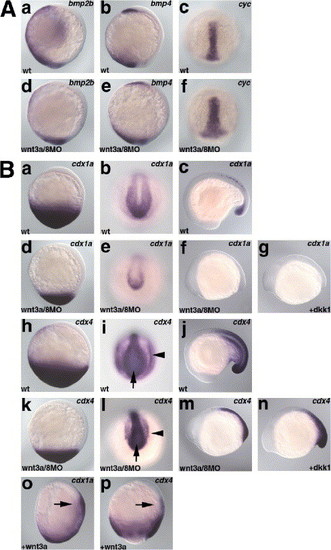
Tailbud expression of caudal-related genes cdx1a and cdx4, but not bmps or cyclops is dependent on the Wnt3a/Wnt8 signaling. (A) Expression of bmp2 and bmp4 at the bud stage, and expression of cyclops (cyc) at the 80% epiboly stage in the wild-type (wt, a, b, c) and wnt3a/wnt8 morphant (wnt3a/8MO, d, e, f) embryos. Expression of bmp2b and bmp4 in the ventro-lateral domains but not in the tailbud was reduced in the wnt3a/wnt8 morphant embryos. Expression of cyc in the axial mesoderm was slightly expanded in the wnt3a/wnt8 morphant embryos. a, b, d, e, lateral views with dorsal to the right. c, f, dorsal views. (B) Expression of cdx1a and cdx4 in the wnt3a/wnt8 morphant embryos and dkk1 RNA (25 pg)-injected embryos, and in embryos misexpressing wnt3a. Expression of cdx1a and cdx4 was reduced in the wnt3a/wnt8 morphant embryos, compared with wild-type embryos, at the bud (a, d, h, k), 8-somite (13 hpf, b, e, i, l), and 17-somite (17 hpf, c, f, j, m) stages. Expression of cdx1a and cdx4 was also reduced in the dkk1 RNA-injected embryos at 17 hpf (g, n). Note that the expression of cdx4 in the posterior neuroectoderm (arrows) was less reduced than in the paraxial mesoderm (arrowheads) in the wnt3a/wnt8 morphant embryos. Ectopic expression of cdx1a and cdx4 was detected in the embryos injected with 6 pg of pCS2 + wnt3a plasmid DNA at the bud stage (o, p, arrows). a, d, h, k, o, p, lateral views with dorsal to the right. b, e, i, l, tailbud views. c, f, g, l, m, n, lateral views with anterior to the left. EXPRESSION / LABELING:
|
|
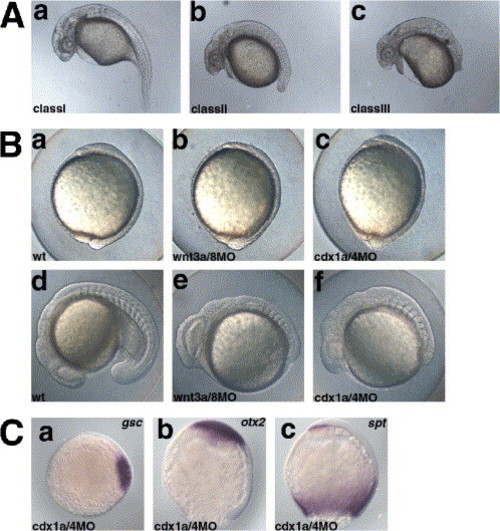
Cooperation of Cdx1a and Cdx4 in formation of the posterior body. (A) Phenotypes of cdx1aMO and/or cdx4MO-injected pharyngula-stage (24 hpf) embryos. The phenotypes were classified into three categories. In Class I embryos, the tail was extended but its size was reduced, and the number of somites was not changed (30?34 somites). Class II embryos had a truncated tail structure (number of the somites 20?30, average 25.2 (n = 10)). Class III embryos had no evident structure protruding from the yolk (no tail structure; less than twenty somites, average 10.3 (n = 12)). The percentage of embryos in each class is shown in Table 2. (B) Comparison between the wnt3a/wnt8 and cdx1a/cdx4 morphant embryos. The phenotypes of wild-type (non-injected) control embryos (wt, a, d), embryos receiving an injection of 4.2 ng of wnt3aMO and wnt8MOs (wnt3a/8MO, b, e), and embryos receiving an injection of 2.1 ng of cdx1aMO and cdx4MO (cdx1a/4MO, cdx1a/cdx4 morphant embryos, c, f), at the bud stage (a?c, lateral views with dorsal to the right) and 17 hpf (d?f, lateral views, with anterior to the left). Neither the wnt3a/wnt8 nor the cdx1a/cdx4 morphant embryos showed strong morphological abnormalities at the bud stage. At 17 hpf (17-somite stage for the wild-type), both the wnt3a/wnt8 and cdx1a/cdx4 morphant embryos displayed a truncated tail, whereas only the wnt3a/wnt8 morphant embryos had an enlarged head and eyes. (C) Expression of goosecoid (gsc) (a) at the shield stage, and otx2 (b) and spadetail (spt) (c) at the 90% epiboly stages in the cdx1a/cdx4 morphant embryos. Expression of these genes was not significantly affected in the cdx1a/cdx4 morphant embryos, compared with the wild-type embryos (Fig. 1). a, animal pole view with dorsal to the right. b, c lateral views with dorsal to the right. EXPRESSION / LABELING:
|
|
Cooperation of FGF and Wnt signaling for the cdx1a and cdx4 expression. Expression of cdx1a and cdx4 at the 3-somite stage in wild-type embryos (wt, A, B), SU5402 (an inhibitor of FGF receptors)-treated embryos (C, D), a dominant negative Xenopus FGF receptor 1 (XFD) RNA (500 pg)-injected embryos (E, F), SU5402-treated wnt3a/wnt8 morphant embryos (G, H), and embryos receiving an injection of 6 pg of pCS2 + fgf8 (I, J). The cdx1a and cdx4 expression was slightly reduced in the SU5402-treated embryos, and more strongly reduced in the embryos defective in both Wnt3a/Wnt8 and FGF signaling. In contrast, expression of cdx1a and cdx4 was expanded anteriorly in the fgf8-misexpressing embryos. Lateral views with dorsal to the right. EXPRESSION / LABELING:
|
|
Regulation of fgf3 and fgf8 expression by Wnt3a/Wnt8 and Cdx1a/Cdx4. (A?F) Expression of fgf3 and fgf8 in the tailbud (bud stage) was reduced in the wnt3a/wnt8 (B, E) and cdx1a/cdx4 (C, F) morphant embryos (A, D, wild-type control). Note that in the wnt3a/wnt8 morphant embryos, fgf3 expression in the forebrain was expanded posteriorly, and in the hindbrain it was shifted posteriorly. Similarly, fgf8 expression in the midbrain?hindbrain boundary (MHB) was shifted posteriorly in the wnt3a/wnt8 morphant embryos. These changes in the fgf3 and fgf8 expression were probably due to anteriorization of the neuroectoderm in the wnt3a/wnt8 morphant embryos. Lateral views with dorsal to the right. (G?L) Reduction of fgf3 and fgf8 expression in the tail region of the wnt3a/wnt8 (H, K) and cdx1a/cdx4 (I, L) morphant embryos (G, J, control). 13 hpf (8-somite stage for wild-type embryos). Posterior (tail) views with dorsal to the top. (M?R) Reduction or loss of fgf3 and fgf8 expression in the tail of wnt3a/wnt8 (N, Q) and cdx1a/cdx4 (O, R) morphant embryos (M, P, control). 17 hpf (17-somite stage for wild-type embryos). Lateral views with anterior to the left. EXPRESSION / LABELING:
|
|
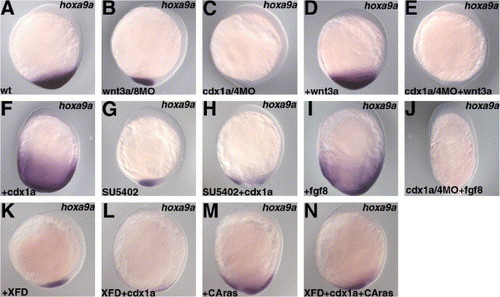
Involvement of Wnt, Cdx1a/4, and FGF in hoxa9a expression. Expression of hoxa9a at the 3-somite stage (A for the wild-type embryos). Expression of hoxa9a was reduced in the wnt3a/wnt8 morphant embryos (B) and it was not detected in the cdx1a/cdx4 morphant embryos (C). Although 6 pg of pCS2 + wnt3a plasmid injection led to increased hoxa9a expression (D), no hoxa9a expression was detected in the wnt3a-injected cdx1a/cdx4 morphant embryos (E). The hoxa9a expression was expanded in the cdx1a RNA (25 pg)-injected embryos (F), but was reduced in the SU5402-treated cdx1a RNA-injected embryos (H), as in the SU5402-treated non-injected embryos (G). The hoxa9a expression was also expanded in the 6 pg of pCS2 + fgf8-injected embryos (I), but was not detected in the embryos receiving injections of both pCS2 + fgf8- and cdx1a/4MO (J). The hoxa9a expression was reduced in the XFD RNA (500 pg)-injected (K), and XFD (500 pg) and cdx1a RNA (25 pg)-injected embryos (L). Co-injection of RNA for a constitutively active Ras (RasV12, CAras, 250 pg) with XFD (500 pg) and cdx1a RNA (25 pg) restored the expression of hoxa9a (N). (M) Expression of hoxa9a in the CAras RNA-injected embryos. Lateral views with dorsal to the right. EXPRESSION / LABELING:
|
|
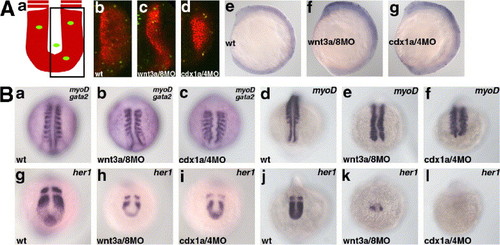
The Wnt-Cdx cascade is involved in morphogenesis of the posterior body. (A) Cell proliferation and cell death was not strongly impaired in the in the wnt3a/wnt8 (c, f) and cdx1a/cdx4 morphant embryos (d, g) (b, e, control embryos). The morphant embryos displayed the Class III phenotype. The embryos were fixed at 17 hpf and stained with the paraxial mesoderm marker papc probe (red) and with anti-phosphohistone H3 antibodies (green). (Aa) Schematic representation of the stained embryos and (Ab?d) confocal images of representative embryos. Note that the number of phosphoH3-positive cells was not significantly different among the wild-type, wnt3a/wnt8-morphant, and cdx1a/cdx4 morphant embryos. (Ae?g) The embryos were fixed at 13 hpf and subjected to Tunel staining. (B) Impairment of the posterior somite formation. Expression of myoD at 13 hpf (a?c, gata2 was also stained) and 17 hpf (d?f). Expression of her1 at 13 hpf (g?i) and 17 hpf (j?l). (a?f) dorsal views and (g?l) posterior views. her1 expression was reduced but displayed normal oscillation in the wnt3a/wnt8 and cdx1a/cdx4 morphant embryos at the early segmentation stage, which had normal anterior somites. At the mid-to-late segmentation periods, her1 expression was strongly reduced or abrogated in the wnt3a/wnt8 and cdx1a/cdx4 morphant embryos, and segmentation stopped for the posterior somites. EXPRESSION / LABELING:
|
|
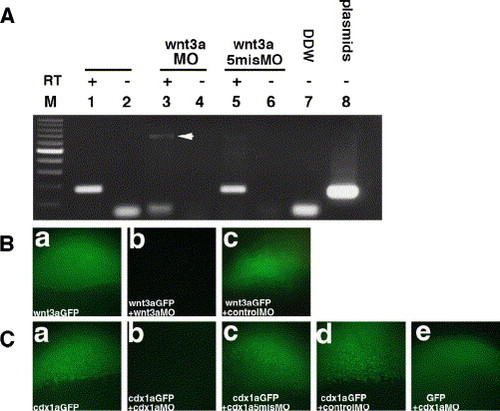
MO-mediated loss of function of Wnt3a and Cdx1a. (A) wnt3aMO inhibits the splicing of the first intron. Embryos injected with wnt3aMO (lanes 3 and 4), wnt3amisMO (lanes 5 and 6), or pCS2+wnt3a plasmid (lane 8) were harvested (lanes 1 and 2, non-injected control embryos) at dome stage, and RNA were isolated from these embryos. cDNAs were synthesized (lanes 1, 3, 5, 7, and 8; lanes 2, 4, and 6 are control without reverse transcriptase) and subjected to PCR using the primers 5′-TGTGGAGTTCGGCAGCATGGT-3′ and 5′-ACCAGCAGGTTTTCACCTCGC-3′ surrounding the first intron. Lane 7 control without template. The correctly spliced wnt3a RNA generates 182 bp PCR products, whereas the RNA deficient in the splicing should produce 814 bp products. Injection of wnt3aMO but not wnt3amisMO inhibited the splicing and generates 814 bp PCR products. (B) wnt3aatgMO inhibits translation of GFP-tagged Wnt3a, in which GFP is tagged at the carboxy terminal of Wnt3a. Expression of Wnt3a-GPF in the embryos that received injection of 50 pg of wnt3a-gfp RNA (a), wnt3a-gfp RNA and wnt3aatgMO (2.1 ng) (b), or wnt3a-gfp RNA and the control MO (c), at 4.5 hpf. wnt3aatgMO but not the control MO inhibited generation (translation) of Wnt3a-GFP. (C) cdx1aMO inhibits translation of GFP-tagged Cdx1a, in which GFP is tagged at the carboxy terminal of Cdx1a. Expression of Cdx1a-GFP in the embryos that received the injection of 100 pg of cdx1a-gfp RNA (a), cdx1a-gfp RNA and cdx1aMO (b), cdx1a-gfp RNA andcdx1amisMO, cdx1a-gfp RNA and the control MO, and expression of GFP in the embryos injected with gfp RNA and cdx1aMO. cdx1aMO inhibited the generation of Cdx1a-GFP protein but not GFP. Neither cdx1amisMO nor the control MO inhibited the translation of Cdx1a-GFP. |

ZFIN is incorporating published figure images and captions as part of an ongoing project. Figures from some publications have not yet been curated, or are not available for display because of copyright restrictions. PHENOTYPE:
|

ZFIN is incorporating published figure images and captions as part of an ongoing project. Figures from some publications have not yet been curated, or are not available for display because of copyright restrictions. PHENOTYPE:
|

Unillustrated author statements EXPRESSION / LABELING:
|
Reprinted from Developmental Biology, 279(1), Shimizu, T., Bae, Y.K., Muraoka, O., and Hibi, M., Interaction of Wnt and caudal-related genes in zebrafish posterior body formation, 125-141, Copyright (2005) with permission from Elsevier. Full text @ Dev. Biol.