- Title
-
Pharyngeal morphogenesis requires fras1-itga8- dependent epithelial-mesenchymal interaction
- Authors
- Talbot, J.C., Nichols, J.T., Yan, Y.L., Leonard, I.F., BreMiller, R.A., Amacher, S.L., Postlethwait, J.H., Kimmel, C.B.
- Source
- Full text @ Dev. Biol.
|
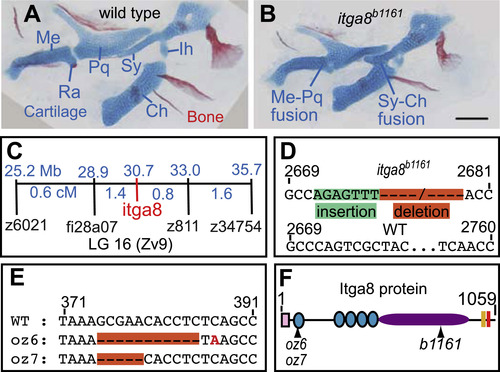
Skeletal defects in b1161 mutants are caused by lesions in itga8. (A, B) Alcian and alizarin staining of (A) wild type and (B) itga8b1161 homozygotes shows skeletal defects caused by itga8 mutations. Homozygous itga8 mutants often show cartilage fusions in the first two pharyngeal arches and defects in symplectic length. (C) Linkage analysis reveals no recombinants (0/696 individuals) between b1161 and the itga8b1161, placing them at the same map position. Map distances (blue) of additional markers from b1161 are shown above in Mb and below in cM. (D) Sequence of the itga8b1161 lesion reveals a 7 bp insertion in exon 25 (green), followed by a 79 bp deletion (orange) (wild type: GenBank JN399198, b1161: GenBank JN399198). This lesion results in protein truncation after amino acid 845 of 1059 with the predicted addition of 21 aberrant amino acids (EFTHWSWRPRLFRTLQSYWAS). (E) Sequence of two CRISPR-induced itga8 lesions, itga8oz6 (11 bp deletion) and itga8oz7 (5 bp deletion), reveal that both introduce frameshifts after amino acid 79 of 1059; itga8oz6 causes an immediate stop codon after the frameshift, while itga8oz7 introduces 30 aberrant amino acids (HLSAGDCGGRSGVLLPLAGIRPRLLPPDPL) before terminating. (F) Itga8 protein diagram with locations of oz6, oz7, and b1161 mutations along with predicted protein motifs. Protein motifs are designated as follows: signal sequence (pink box), integrin beta domains (teal circles); integrin alpha domain (purple oval), transmembrane domain (peach box), and an intracellular integrin domain (red box). Cartilage abbreviations: Meckel’s (Me), Retroarticular process (Ra), Palatoquadrate (Pq), Symplectic (Sy), Ceratohyal (Ch), Interhyal (Ih). Scale bar (100 µm) in B also applies to A. PHENOTYPE:
|
|
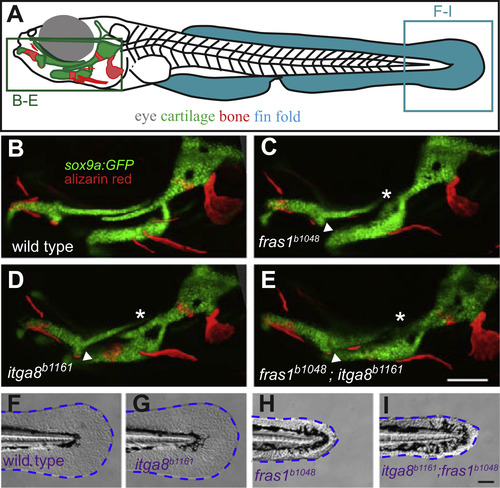
Comparison of skeletal, endodermal, and tail ectoderm phenotypes between wild type, itga8b1161 mutants, fras1b1048 mutants, and itga8b1161;fras1b1048 double mutants. (A) Illustration of a zebrafish larva, indicating regions shown in subsequent (B-I) panels. (B-E) Skeletal morphology is revealed using sox9a:EGFP expression (cartilage) and alizarin red staining (bone) at 7 dpf; itga8 and fras1 single and double mutants display similar cartilage defects, in particular, Meckel’s-palatoquadrate joint fusion (arrowhead) and symplectic-ceratohyal fusions (asterisk). (F-I) Bright field images showing normal fin fold morphology (outlined blue) in (F) wild type and (G) itga8b1161 individuals versus the “blister phenotypes” in (H) fras1b1048 and (I) itga8b1161;fras1b1048 mutants. Scale bars (E and I) are 100 µm. Scale bar in E applies to B-E; Scale bar in I applies to F-I. |
|
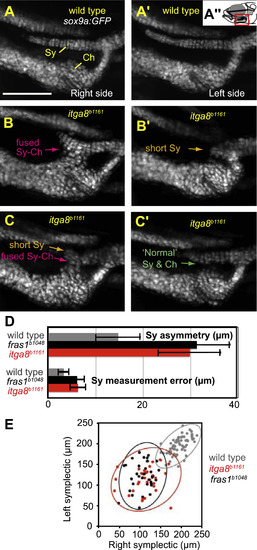
Phenotypic variation in fras1 and itga8 mutant phenotypes shows fluctuating asymmetry. (A-C′) Facial cartilage skeleton marked by sox9a:GFP expression at 7.5 dpf, region shown is boxed in red in A”. Compared to wild type (A), the itga8b1161 mutant skeleton is often asymmetric (B and C). For instance, the fish in (B) shows an extended symplectic cartilage fused to the ceratohyal cartilage on the right side and an unfused, severely shortened, symplectic phenotype on its left side (B′). In another example, both “Short Sy” and “Fused Sy-Ch” symplectic phenotypes (C) are found in a fish presenting only subtle defects on the opposite side (C′). (D and E) At 7.5 dpf, average symplectic length in itga8 mutants is shorter than wild type, but comparable to fras1 mutants. Symplectic cartilages in itga8 mutants show asymmetry similar to fras1 mutants, which is twice as high as wild-type asymmetry. (E) Plot of symplectic lengths measured on left and right sides, with grouped 95% density ellipses. Symplectic lengths for wild types were along the diagonal as expected for a high degree of left/right correlation, but for itga8b1161 and fras1b1048 mutants, symplectics were much shorter and do not correlate well between sides. Scale bar (A) is 100 µm, applicable to A-C′. Error bars (D) show 95% confidence intervals: 1.95 times standard error. PHENOTYPE:
|
|
fras1 and itga8 are expressed in adjacent facial tissues. (A–D) Colorimetric in situ hybridization, developed using NBT/BCIP on wild-type sections, showing fras1 expression in pharyngeal and ectodermal epithelia (A, B) and itga8 expression in mesenchyme (C and D) at 36 hpf (A and C) and 72 hpf (B andD). (E) Color-coded diagram of lateral and transverse sections from a wild-type 60 hpf embryo, showing the first two pharyngeal arches (green), ectoderm (orange), cartilage (blue), and endoderm (red). (F and G) Fluorescent RNA in situ for fras1, itga8, and col2a1 expression in a 60 hpf transverse section of wild-type embryos at (F) low and (G) higher magnification. All scale bars: 50 µm. EXPRESSION / LABELING:
|
|
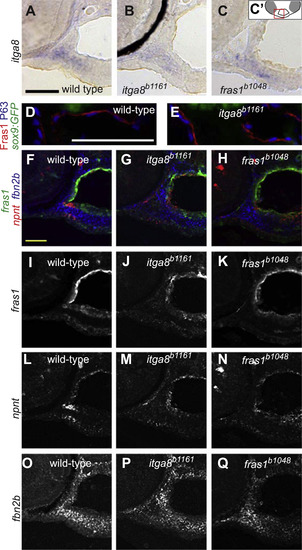
fras1 and itga8 do not regulate one another, nor do they regulate candidate targets npnt or fbn2b. (A-C) RNA in situ hybridization for itga8 transcripts on transverse sections in 60 hpf wild-type and mutant embryos, developed colorimetrically, oriented as shown in C’. These transcripts appear similar in (A) wild type, (B) itga8 mutants, and (C) fras1 mutants. The black crescent in (B) is eye pigment. (D and E) 72 hpf tissue sections, labeled for Fras1 protein, epithelial nuclei (anti-P63) and cartilage (sox9a:GFP). Fras1 protein levels and localization appears similar in wild type (D) and itga8b1161 mutants (E). (F-Q) Triple in situ hybridization for fras1, npnt, and fbn2b transcripts on 60 hpf tissue sections, oriented as in Fig. 4 (E). (F-H) Merged overlays of all three probes, which are also shown separately for fras1 (I-K), npnt (L-N), and fbn2b (O-Q). For all three genes, expression is similar between wild type, fras1 mutants, and itga8 mutants. All scale bars: 50 µm. Scale bar in A applies to A-C. Scale bar, in D applies D, E. Scale bar in F applies to F-Q. EXPRESSION / LABELING:
|
|
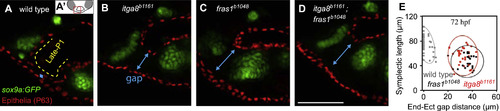
Late-p1 is severely affected in itga8 and fras1 single and double mutants. (A-D) Tissue sections labeled with anti-P63 (epithelial nuclei) and sox9a:EGFP (cartilage) at 72 hpf, and oriented as shown in A′. The late-forming outpocketing of pouch-1 (late-p1) is indicated with a yellow dotted line in wild type. However, late-p1 is absent in (B) itga8b1161, (C) fras1b1048, and (D) fras1b1048;itga8b1161 double mutants, leaving a large gap (blue line) between endoderm and ectoderm. (E) Symplectic cartilage length and ectoderm-endoderm gap distance were measured on confocal stacks of whole-mounted embryos expressing sox9a:GFP and labeled with anti-P63. Each measurement pair is plotted as a dot, with 95% density ellipses shown for each genotype. EXPRESSION / LABELING:
PHENOTYPE:
|
|
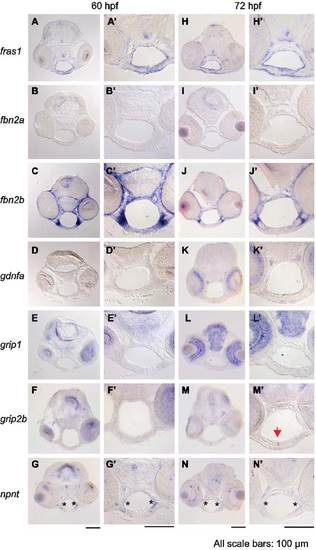
Expression of fras1-related and itga8-related genes in facial tissues. (A-N′) RNA in situ hybridization on wild type tissue sections at (A-G′) 60 hpf or (H-N′) 72 hpf. Each image in A-N shows a lower magnification image of individual sections and A′-N′ shows higher magnification images of these sections, focusing on the pharyngeal region. (A and H) fras1 expression in facial endoderm and ectoderm is shown as a positive control. (B and I) fbn2a shows no detectable facial expression. (C and J) fbn2b is strongly expressed in arch mesenchyme. (D and K) The fras1 processing gene grip1 is expressed in brain and eyes, but not in the pharyngeal regions. (E and L) Similarly, grip1 paralog grip2b is not visibly expressed in pharyngeal regions at 60 hpf (F), but is present by 72 hpf in the joint between the left and right Meckel’s cartilage (M, arrow). This suggests that genes other than grip1 and grip2b may process Fras1 in the face, or that earlier grip expression enables Fras1 processing. Two primer-sets failed to amplify grip2a cDNA at 3 dpf, so we did not assay facial expression of this gene. (G and N) npnt expression is most intense along the lateral edge of facial endoderm (asterisk), where the proposed Itga8-FPC interactions occur. All scale bars are 100 µm, applicable to their respective columns. EXPRESSION / LABELING:
|
|
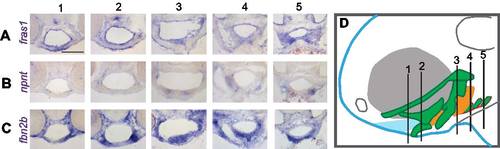
npnt is expressed in sub-regions of arch epithelia, and fbn2b is expressed in arch mesenchyme. In situ hybridization of sections from the head of 60 hpf wild-type embryos. (A) fras1 is expressed in epithelia. (B) npnt is expressed in a subset of these epithelia, while (C) fbn2b is expressed in a complementary pattern in arch mesenchyme. (D) Illustration of section level. Ectoderm is shown in blue; cartilages are green; anterior endoderm is orange; posterior endoderm is red. Anterior-posterior level for 1, 2, 3, 4 and 5 are shown with lines in D. Scale bar is 50 µm, applicable to A-C. |
|
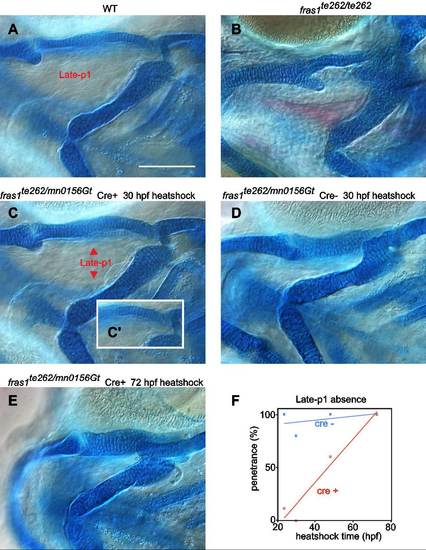
Cartilage and late-p1 phenotypes largely correlate in critical window experiments. (A–B) Differential interference contrast images of alcian/alizarin stained tissues of 6 dpf WT (A) and fras1te262 mutant homozygotes (B). (A) In WT, skeleton is well formed, and a completely outpocketed late-p1 is apparent. (B) In fras1te262 homozygotes, cartilage defects are seen and late-p1 is absent, as evidenced by mesenchyme occupying the space between ceratohyal and more dorsal skeleton. (C–E) 6 dpf alcian/alizarin skeletal preparations of fras1 reversion experiments, from crosses described in Fig. 7. Briefly, embryos trans-heterozygous for fras1mn0156Gt and fras1te262d were heat-shocked at different stages to induce Cre recombinase expression in the half of the clutch that inherited the hsp70l:Cre transgene; this Cre induction restores fras1mn0156Gt function. (C) If Cre is activated before the fras1 critical window (30 hpf), larvae form normal endodermal and skeletal morphology. (C’) Inset is a different focal plane showing normal symplectic morphology. (D) At 30 hpf, however, heat shock of control Cre siblings does not result in phenotypic rescue. (E) When heat-shock of Cre restores fras1mn0156Gt function after the critical window for fras1 function (72 hpf), both endodermal and skeletal phenotypes resemble fras1te262 homozygotes. (F) Plot of late-p1 defect penetrance as a function of time of development, determined by DIC imaging on one side of each embryo (n′s range from 5 to 13 for each condition). In Cre+ fish, there is a significant correlation between heat shock time and late-p1 defect penetrance (R2=0.92, P<0.05), however there is no significant correlation in Cre- fish (R2=0.17, P=0.6). Scale bar is 100 µm, applicable to A-E. |

ZFIN is incorporating published figure images and captions as part of an ongoing project. Figures from some publications have not yet been curated, or are not available for display because of copyright restrictions. |
Reprinted from Developmental Biology, 416(1), Talbot, J.C., Nichols, J.T., Yan, Y.L., Leonard, I.F., BreMiller, R.A., Amacher, S.L., Postlethwait, J.H., Kimmel, C.B., Pharyngeal morphogenesis requires fras1-itga8- dependent epithelial-mesenchymal interaction, 136-48, Copyright (2016) with permission from Elsevier. Full text @ Dev. Biol.