- Title
-
Dynamic Assembly of Brambleberry Mediates Nuclear Envelope Fusion during Early Development
- Authors
- Abrams, E.W., Zhang, H., Marlow, F.L., Kapp, L., Lu, S., and Mullins, M.C.
- Source
- Full text @ Cell
|
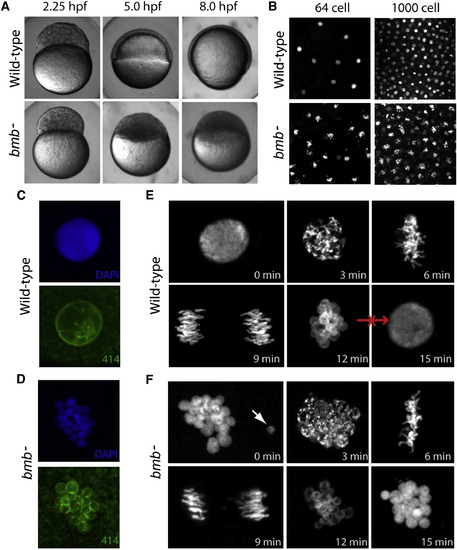
bmb Is Required for Early Development and Proper Nuclear Morphology(A) Prior to the MBT (2.25 hpf), embryos from bmb mutant females are morphologically similar to WT embryos. After the MBT at 5.0 and 8.0 hpf, bmb mutants fail to undergo cell movements associated with epiboly and gastrulation.(B) DAPI staining of interphase nuclei during cleavage in WT (top) and bmb (bottom) reveals multiple chromatin bodies associated with each nucleus.(C and D) (C) WT and (D) bmb interphase nuclei stained with mab414 (green) indicate that bmb nuclei are multimicronucleated compared to WT.(E and F) (E) WT and (F) bmb cell-cycle time course at the two- to four-cell transition (images are projections of multiple confocal Z-slices). Top left, interphase; top middle, prophase; top right, metaphase; bottom left, anaphase; bottom middle, telophase; bottom right, interphase. In (E) and (F) for each time point, n = 3.In this and subsequent figures, ?n? refers to number of embryos examined (unless otherwise noted). In each case, multiple nuclei or cells of each embryo were also examined. PHENOTYPE:
|
|
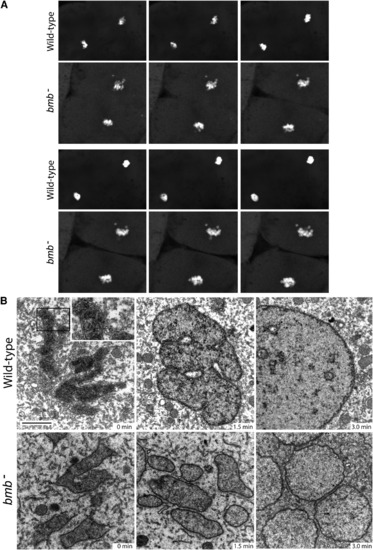
Nuclear Membrane Fusion Is Disrupted in bmb Mutants(A) Frames from time-lapse experiments at the telophase-interphase transition demonstrate that chromatin bodies normally coalesce in WT (top) but fail to do so in bmb (bottom). Note: six frames were selected (from Movies S1 and S2) to best align the sequence of events between WT and bmb at the telophase-interphase transition.(B) Electron microscopy of WT versus bmb at the telophase-interphase transition. Embryos at the 128-cell stage were fixed at 90 s intervals for transmission electron microscopy (TEM). Black bar, 2 microns. The inset in WT (0 min) is enlarged 2� to show the double-membrane nuclear envelope. For each time point, n = 2 embryos. Multiple cells from each embryo were examined in both WT and bmb. PHENOTYPE:
|
|
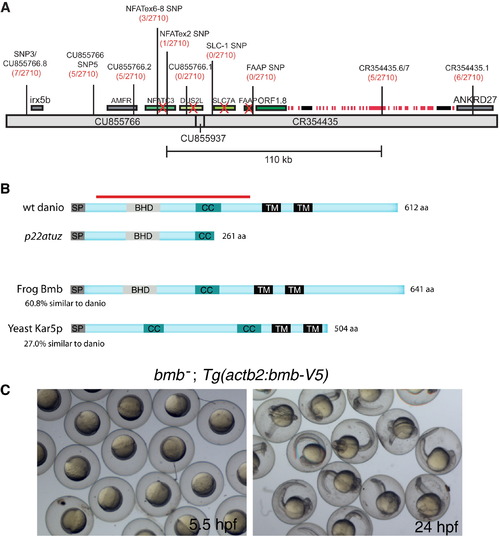
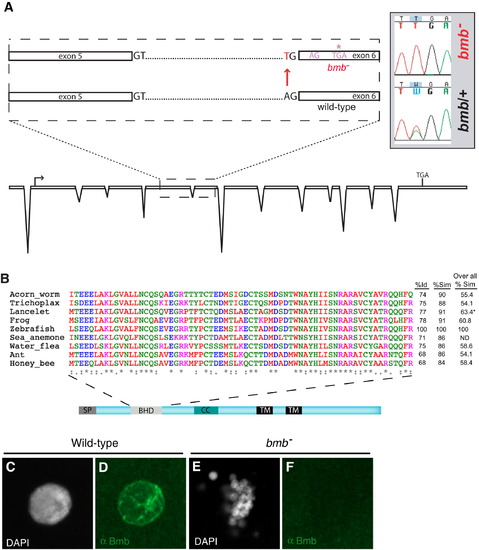
bmb Encodes a Conserved Predicted Transmembrane Protein(A) Recombinants over total meiotic events are indicated parenthetically in red below the marker name. Candidate genes eliminated based on their WT cDNA sequence are indicated with a red X. A histone gene cluster is indicated as red boxes. Two predicted transposable elements are indicated by black boxes.(B) ORF1.8 encodes a 612 residue protein with a predicted N-terminal signal peptide sequence (SP) followed by a BHD, a coiled-coil (CC), and two C-terminal transmembrane domains (TM) initiating at amino acid residues 363 and 415 (TMpred program) (Hofmann and Stoffel, 1993). Yeast Kar5p is 27% similar (excluding the C-terminal region) to the D. rerio homolog. The red bar corresponds to the recombinant protein used to generate Bmb antibodies.(C) Rescued bmb embryos at 50% epiboly (5.5 hpf) and 24 hpf.See also Figure S1. |
|
Bmb Protein Dynamics during Mitosis(A) Bmb protein localizes as distinct foci to the mitotic spindle region during metaphase (n = 4).(B) During anaphase, Bmb is interspersed between the separating chromosomes (inset; n = 3) and in the region of the mitotic spindle.(C) Later in anaphase, Bmb foci are decorating the chromosomes (inset; n = 3).(D) At the 2 min time point, Bmb surrounds the individual chromosomes, which are still separating (n = 5).(E) Schematic summarizing Bmb localization during metaphase to anaphase/telophase. Arrows in the right panel indicate that the karyomeres will continue to move to their final central position in the cell. 0 min corresponds to metaphase at the 32- to 64-cell transition. Bmb (green), microtubules (DM1a, red), and chromosomes (DAPI, white and blue in merge).Scale bar, 5 μm, and insets show 2� enlargements. See also Figure S2. EXPRESSION / LABELING:
|
|
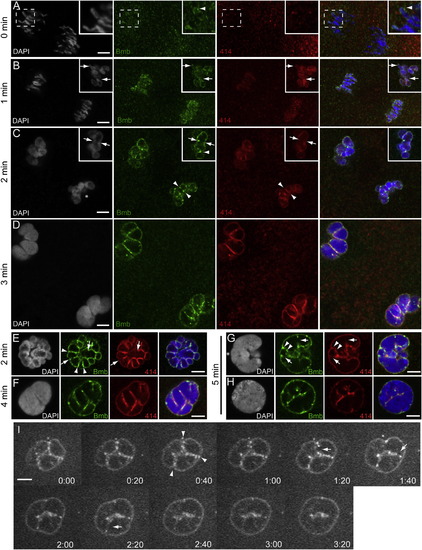
Bmb Foci Mark Sites Corresponding to Pre-, Post-, and Active Karyomere Fusion(A) At time point designated 0 min (midanaphase) at the 32- to 64-cell transition, Bmb protein (green) is first seen accumulating at the leading edge of the separating chromosomes (inset, arrowhead) prior to the assembly of nucleoporins (marked by mab414, red).(B) As the chromosomes continue to separate, nuclear membrane (marked by mab414) and Bmb surround individual chromosomes, forming karyomeres. Insets in (B) show a more advanced example from the same time point (1 min).(C) Karyomeres become increasingly more spherical, and prominent Bmb foci are more apparent at karyomere-karyomere interfaces (arrowheads). In another example, Bmb foci span a former interface, which now lacks nuclear membrane (inset, arrows), and a prominent Bmb aggregate interfaces adjacent karyomeres (inset, arrowhead).(D) At the 3 min time point, larger secondary karyomeres are present.(E) Another 2 min example at a 90� view compared to (C) (an asterisk in [C] marks reference point). Bmb foci at karyomere-karyomere interfaces, where membrane is still present (arrowheads) or partially/completely absent (arrows).(F) Secondary karyomere formation at a 45� view.(G and H) Bmb foci (Bmb, arrowheads) flank presumptive karyomere prefusion site (membrane still present, 414, arrowheads), and Bmb foci can be found at presumptive karyomere postfusion sites (lack of membrane, 414, arrows). Note that (G)?(H) show mononucleus formation (5 min) from a lateral side view (G) and (H) another sample from a 90� vantage point (reference point marked with asterisk in [G]). All panels correspond to individual confocal Z-slices. Scale bar, 5 μm, and inset in (A) shows a 2� enlargement. For each time point, n e 3.(I) Time-lapse imaging of Bmb-venus. Bmb foci are seen at the karyomere-karyomere interfaces (arrowheads) and are ultimately eliminated (arrows) as the mononucleus is formed.See also Movie S3 and Figure S3 for details. EXPRESSION / LABELING:
|
|
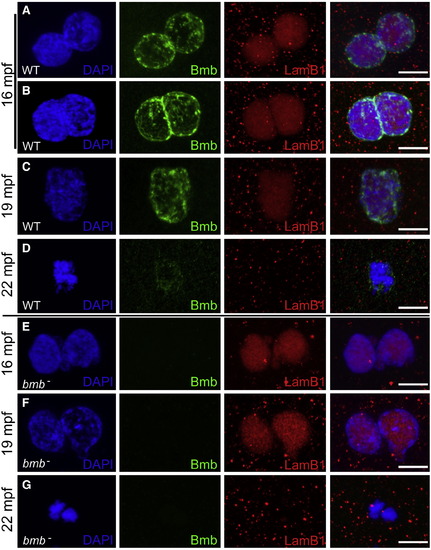
Bmb Is Required for Pronuclear Fusion(A) At 16 mpf, WT pronuclei are fully congressed.(B) Some WT pronuclei begin to fuse at 16 mpf.(C) At 19 mpf, WT pronuclear fusion is complete.(D) At 22 mpf, in WT embryos a single condensed DNA mass is detected at the first prometaphase.(E) At 16 mpf, bmb pronuclei are fully congressed.(F) At 19 mpf, bmb pronuclei fail to fuse as chromatin begins to condense.(G) At 22 min, two separate condensed DNA masses are detected in bmb embryos at the first prometaphase. Scale bars, 10 μm. At each time point, n e 4. Images correspond to Z-projections of individual confocal Z-planes. WT TL males were crossed to female bmb mutants in (E)?(G). |
|
Cloning, Alignment of the BHD from Potential Homologs, and Anti-Bmb Staining of Interphase Nuclei, Related to Figure 3(A) Our annotation of non-gene encoding genomic regions in the interval revealed a gene corresponding to a novel ORF, ORF1.8. In this gene in the p22atuz mutant allele, we identified an A to T base change at the splice acceptor site upstream of exon 6.(B) A 69-residue domain spanning residues 91 to 159 of zebrafish Bmb is compared with a diverse set of potential homologs (first column- % identity (Id), second column- % similarity (Sim) with in the BHD. The third column is a comparison of overall similarity among the potential Bmb homologs. * Only the available residues of the lancelet protein are aligned. A significant portion of the sea anemone protein is not available so overall similarity was not determined (ND).(C?F) (C and D) WT and bmb (E and F) interphase selected embryos were stained with Dapi (C and E) and anti-Bmb (D and F). Embryos are at the 32-cell stage (n = 3 WT; n = 6 bmb). Images correspond to a Z-projection from a confocal Z-series. |
|
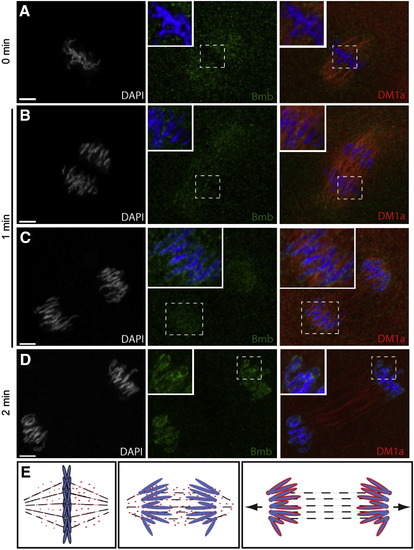
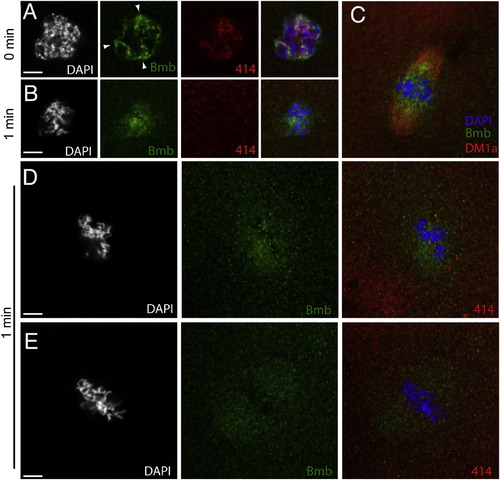
Bmb Localization at Prophase through Metaphase, Related to Figure 4(A) Embryos fixed at one-minute time points were stained with anti-Bmb (green), mab414 (red) and DAPI (white/blue). During prophase (A) (0 min), Bmb signal can be seen concentrated in regions of the nucleus as the chromatin condenses (n = 5). (B) At prometaphase (B) (1 min), concentrated Bmb begins to disperse (n = 5). (C) (C) is an analogous time point to (B) taken from an independent experiment staining for Bmb (green) and microtubules (red, DM1a) indicating that Bmb protein becomes more concentrated in the region of the spindle closer to the chromosomes (n = 5). (D) A different sample from the same time point (1 min) reveals Bmb signal becoming more dispersed in a punctate arrangement. At this point as in (B) mab414 is completely dispersed (n = 3). (E) Once the chromosomes are aligned at the metaphase plate, Bmb becomes more evenly distributed within the spindle region (n = 5). 0 min corresponds to early prophase at the 32-to-64 cell transition. Images correspond to Z-projections of individual confocal Z-planes. |
|
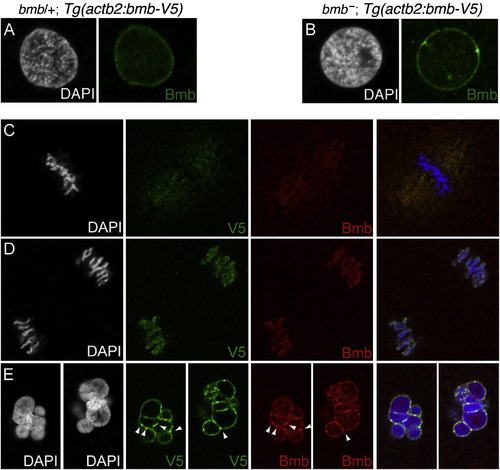
Transgenic Rescue of Bmb Multimicronuclei and Bmb-V5 Transgene Colocalizes with Endogenous Bmb in WT Embryos, Related to Figure 5(A and B) (A) A bmb-V5 transgene (corresponding to the bmb ORF) in WT and (B) bmb backgrounds.(C?E) (C) WT bmb-V5 transgenic embryos at metaphase, (D) anaphase, and (E) telophase (karyomere stage). These images correspond to single confocal slices at the 32-to-64 cell transition. Note colocalization of endogenous Bmb (red) and Bmb-V5 (green) foci (arrowheads). |
|
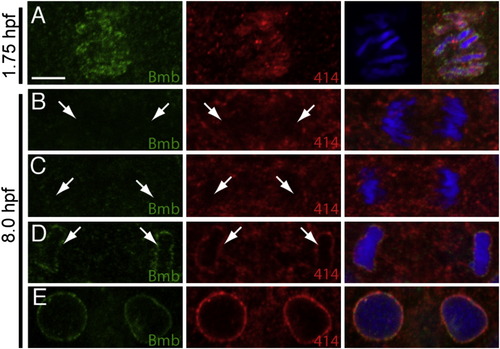
Nuclear Envelope Assembly during Midgastrulation, Related to Figure 6(A) One set of separating chromosomes (single Z-slice) at 1.75 hpf (comparable time point to 5B) stained with Bmb (green) and mab414 (red).(B?E) Anaphase-interphase transition at 8.0 hpf. In (B) and (C) Bmb (left, green) and mab414 (middle, red) are not detected on the separating chromosomes (arrows). In (D) Bmb and mab414 are now detected on the single chromatin mass and the fully formed nucleus (E). Scale bar = 5 μm. Panels only show a single Z-slice to illustrate individual chromosomes versus a single chromatin mass. Each time point n e 3. |
Reprinted from Cell, 150(3), Abrams, E.W., Zhang, H., Marlow, F.L., Kapp, L., Lu, S., and Mullins, M.C., Dynamic Assembly of Brambleberry Mediates Nuclear Envelope Fusion during Early Development, 521-532, Copyright (2012) with permission from Elsevier. Full text @ Cell