- Title
-
A screen for mutations in zebrafish that affect myelin gene expression in Schwann cells and oligodendrocytes
- Authors
- Kazakova, N., Li, H., Mora, A., Jessen, K.R., Mirsky, R., Richardson, W.D., and Smith, H.K.
- Source
- Full text @ Dev. Biol.
|
Myelination and neuronal differentiation in the PLL. (A–F) show whole mount preparations of 2.5–5 dpf zebrafish. (A) Mbp expression begins around 2.5 dpf in myelinating glial cells (arrow) immediately posterior (arrowhead) to the posterior lateral line ganglion (pllg). (B) Expression is activated in progressively more posteriorly situated cells as time progresses. By 3 dpf mbp-expressing cells (arrow) extend 5–7 somites posterior to the pllg. (C) By 5 dpf mbp (arrow) is expressed all along the PLL. (D) Mbp expression in the anterior lateral line and the hindbrain at 5 dpf. c—commissure. ht—hindbrain tract. (E) Plp1b expression in the PLL (arrow) begins around 4 dpf. (F) PLL axons (arrow) at 5 dpf labeled with anti-acetylated tubulin. Higher power view of inset shows axons extending ventrally to innervate individual neuromeres (arrowheads). Panels G, H are electron micrographs of the PLL of a 5 dpf larva showing a transverse section through the PLL. (H) High power view of inset in panel G. Note loose wraps (arrowheads) around the large diameter axon. Scale bars in panels A, B and inset in panel F 5 μm; in panel C 20 μm; in panels D–F 10 μm; in panel G 1 μm; in panel H 200 nm. |
|
PLL phenotype of doc and igu mutants. (A, B) Mbp expression (arrow) in 5 dpf larvae homozygous mutant for doc and igu respectively. Expression is restricted to the anteriormost section of the PLL. (C, D) Anti-acetylated tubulin staining (arrow) of PLL axons in panel C doc and panel D igu mutants. A PLL nerve extends to the posterior segments (arrows) of the fish in both mutants. (E, F) Electronmicrographs showing a transverve section of the rostral (mbp expressing) PLL in doc mutants. Axons ensheathed by loosely wrapped myelin (arrows) are clearly present in the mutant nerve. Scale bars in panels A and C, 5 μm; in panels B and D, 10 μm; in panel E, 1 μm; in panel F, 200 nm. |
|
PLL phenotype of mot/ott mutants. (A–C) Myelin protein expression in ott and mot mutants. Both mutants show a complete lack of mbp and Plp1b expression. (D, E) Mpz expression in the CNS (arrows) of wild type (D) and ott (E) embryos at 72 hpf. Mpz is expressed at reduced levels in ott embryos. (F) Anti-acetylated tubulin staining of the PLL in ott mutants. The axons (arrow) are present but do not extend the whole length of the fish. (G–J) Early development of the PLL nerve and glia precursors. At 30 hpf, axons of the PLL nerve (arrows)of ott mutants (G) have extended less far than in the wild type. Arrowhead—caudal end of yolk sac (I). Prior to myelination, early glial expression (arrowheads) of foxD3 in both mutant (H) and wild type (J) fish. (K–N) Electronmicrographs of transverse sections through the PLL nerve of wild type (K, L) and ott mutant (M, N) fish 80 hpf. The larger diameter axons are ensheathed by loosely packed myelin (arrows) typical of this early stage of development. Scale bars in panels A–C 10 μm; in panels D–F, H and J 5 μm; in panels G and I 3 μm; in panel E 1 μm; in panels K and M; in panels L and N 200 nm. PHENOTYPE:
|
|
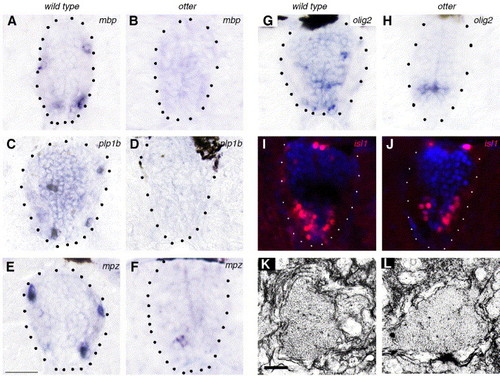
CNS phenotype of ott mutants. Transverse sections through the spinal cord of 80 hpf larvae. (A–F) Myelin protein expression in wild type (A, C, E) and ott (B, D, F) mutant fish. (A, B) mbp is not expressed in ott mutants. (C, D) Plp1b is not expressed in ott mutants. (E, F) Mpz is weakly present in some grey matter cells in ott mutants. (G, H) In wild type fish, olig2-expressing cells have migrated to the periphery by 80 hpf (G) whereas olig2 expression is still restricted to the ventral ventricular zone in ott mutants (H). (I, J) The distribution of cells expressing the motor neuron marker isl1 is similar in wild type (I) and ott mutant (J) fish. (K, L) Electronmicrographs of spinal cord axons ensheathed by loosely wrapped immature myelin in wild type (K) and ott mutant (L) fish. Scale bars in panels K and L, 200 nm. EXPRESSION / LABELING:
PHENOTYPE:
|
|
PLL phenotype of nls and nof mutants. (A) 5 dpf nls mutant fish. Note mbp expression in the anterior lateral line (arrows) but absent from the PLL. (B) PLL nerve (arrow) of nls mutant 5 dpf stained with anti-acetylated tubulin. In the wild type, axons branch off ventrally at intervals to innervate individual neuromeres (arrowhead) but no such branching is seen in the mutant. (C, D) 5 dpf nof mutant fish. The nof phenotype shows variable expressivity and in some mutants mpb is expressed on one side only (arrow) as shown in panel D. Note normal mbp expression in the PLL on the left side (arrow). (E, F) Plp1b expression in nls mutant (E) and wild type (F) fish. Plp1b is detectable on both sides in the wild type and on one side of the nls mutant (arrow). (G. H) Electron micrographs of the PLL in nls mutant fish. Note loosely wrapped ensheathment by myelin (arrows) of the large diameter axons. Scale bars in panels B, E and F, 10 μm; in inset in panel B, 5 μm; in panel G 1 μm; in panel H, 200 nm. |
|
(A–C) CNS phenotype of nls mutants at 4 dpf (A) Mbp expression in wild type spinal cord at 4 dpf. Differentiating oligodendrocytes are located peripherally in the white matter. (B) Mbp expression is absent in the great majority of nls mutant fish. (C) Sox10 expression in wild type spinal cord. Sox10 labels oligodendrocyte precursors in the grey matter (arrow) as well as differentiating oligodendrocytes in the periphery. (D) Sox10 expression appears normal in nls mutants. Note labeling of oligodendrocyte precursors in the grey matter (arrow). (E–H) Effects of timed application of DEAB (D–F) and RA (G) on wild type and nls mutant fish respectively. (E) Effects of DEAB treatment from 11 hpf on mbp expression. (F) Effects of DEAB treatment from 14 hpf on mbp expression. (G) Effects of DEAB treatment from 26 hpf on mbp expression. (H) Mbp expression is restored in the PLL (arrowheads) of nls mutants treated with exogenous RA between 10 and 16 hpf. EXPRESSION / LABELING:
|

Unillustrated author statements PHENOTYPE:
|
Reprinted from Developmental Biology, 297(1), Kazakova, N., Li, H., Mora, A., Jessen, K.R., Mirsky, R., Richardson, W.D., and Smith, H.K., A screen for mutations in zebrafish that affect myelin gene expression in Schwann cells and oligodendrocytes, 1-13, Copyright (2006) with permission from Elsevier. Full text @ Dev. Biol.