- Title
-
A molecular basis for water motion detection by the mechanosensory lateral line of zebrafish
- Authors
- Chou, S.W., Chen, Z., Zhu, S., Davis, R.W., Hu, J., Liu, L., Fernando, C.A., Kindig, K., Brown, W.C., Stepanyan, R., McDermott, B.M.
- Source
- Full text @ Nat. Commun.
|
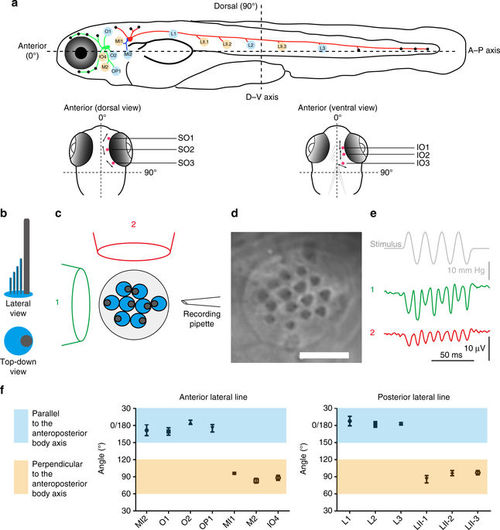
Axes of best sensitivity for anterior and posterior lateral line neuromasts. a Schematic of the zebrafish lateral line at ~6 dpf. (top) Side view. Neuromasts that have been scored are blue or tan and correspond to the graph below. ALL(green), PLL(red), and medial–lateral-line (blue) ganglia contact neuromasts. (bottom) Dorsal and ventral views of the head. Supraorbital (SO) neuromasts 1 to 3 and infraorbital (IO) neuromasts 1 to 3 are marked in red. The mean angle of the orientation of the axis of best sensitivity relative to the A–P body axis of each neuromast is presented as a double-headed arrow. b–e Evaluation of the axis of best sensitivity for a neuromast. b Schematics of hair bundles. Gray and blue protrusions are a kinocilium and stereocilia, respectively. c Experimental configuration: a top-down view of a neuromast with a fluid jet pipette that provides stimuli (1) parallel to the axis along which the bevel-shaped hair bundles face opposite directions or (2) orthogonal to that axis. The pipettes in the diagram are not to scale. d DIC image of hair bundles of a posterior neuromast from which stimulus-evoked microphonic potentials were recorded. Scale bar = 5 μm. e Recordings from an A–P oriented L3 neuromast with fluid jet pipettes stimulating, serially, from two different directions depicted in c. f Graphs of mean angle of the orientation of the axis of best sensitivity relative to the A–P body axis of each anterior and posterior neuromast position ± SEM (n = 4–7). Each axis with a range from 60° to 120° from the A–P body axis is defined perpendicular (tan). Each axis with a range from 0° to 30° or 150° to 180° from the A–P body axis is defined as parallel (blue). ALL nomenclature: opercular (OP), mandibular (M), infraorbital (IO), supraorbital (SO), middle (MI) and otic (O). PLL nomenclature: L neuromasts are A–P oriented; LII neuromasts are D–V oriented. These different neuromast types are derived from different migrating primordia during development |
|
Localization of Tmc2b and TALEN-mediated disruption of the tmc2b gene. a Schematic of the hair bundle. b Tmc2b localizes to the tips of stereocilia in a neuromast. Two lateral line hair cells expressing Tmc2b-GFP (green) demonstrate that the fusion protein localizes to the tips of β-actin-mCherry-labeled stereocilia (red). Scale bar = 0.5 μm. This pattern was observed in 130 of 381 hair cells of somatic transgenics. In transgenic hair cells with weak expression, localization to stereocilia was difficult to recognize. c Graphical representation of the tmc2b genomic locus in zebrafish. Putative exons and splice sites are displayed. Red arrow marks the targeted exon, exon 4. d Segment of exon 4 subjected to genome editing. Two differently engineered TALENs bind their corresponding half-sites to enable FokI dimerization and DNA cleavage. Mutagenesis deleted seven nucleotides. e Amino acid sequences of wild-type and mutant proteins. The alteration results in an opal mutation upstream of all putative transmembrane domains. f Sequencing results of mutagenized and control loci. Blue highlight and blue delta indicates deleted 7-nucleotide stretch absent in mutant. Red highlight denotes the opal mutation that was generated at the site of the TALEN targeting. g (left) Topographical representation of the Tmc2b protein. Red arrowhead indicates point of introduced mutation. Amino acids of putative transmembrane domains are labeled in blue and the TMC domain is in green. (right) Predicted truncated product of mutation EXPRESSION / LABELING:
|
|
Variable dependence on Tmc2b for mechanotransduction in posterior neuromast hair cells. a Extracellular recordings of microphonic potentials measured from posterior neuromasts with A-P orientations of 6-dpf zebrafish larvae are displayed. The responses in tmc2b −/− are often absent, bottom trace (n = 9/15); however, several responses (n = 6/15) are highly asymmetric, with weakened amplitude for one direction of stimulus and no response for the other direction, penultimate trace. Deflection anteriorly (A) or posteriorly (P). b, c Qualitative confocal images of hair cells labeled with FM1-43FX (red), to assess mechanotransduction function, and parvalbumin 3 (cyan)50, as a counter label, to visualize mature hair cells. b A posterior neuromast from a tmc2b +/− animal demonstrates co-labeling of all hair cells. c In a tmc2b −/− animal, a posterior neuromast contains hair cells that label with FM1-43FX and a subset that do not (label:no label; 2:5). Orange n, hair cells labeled with parvalbumin 3 antiserum. Blue n, hair cells that load with FM1-43FX. Green n, hair cells that do not load. Scale bar = 5 μm. d, e Percentages of hair cells within PLL neuromasts that label with 4-Di-2-ASP. d In neuromasts of tmc2b +/+ and tmc2b +/− animals, most hair cells take up fluorophore. Approximately 35% of hair cells in each PLL neuromast of tmc2b −/− fish take up 4-Di-2-ASP. In contrast, in cdh23 aj64a/aj64a mutants, all hair cells take up dye, though at much lower quantities than wild-type animals (see f and Supplementary Fig. 4b). **** Kruskal–Wallis test P value < 0.0001. n values ≥ 20. e In mutants, A–P-facing and D–V-facing posterior neuromasts have similar percentages of hair cells that gather fluorophore, ~38 and 32%, respectively. ****Student’s t-test P value < 0.0001. n values ≥ 23. f Mean fluorescence intensity of 4-Di-2-ASP uptake of the brightest cell per neuromast. n values ≥ 17. Kruskal–Wallis test ****P < 0.0001, ***P = 0.001, **P = 0.0215 |
|
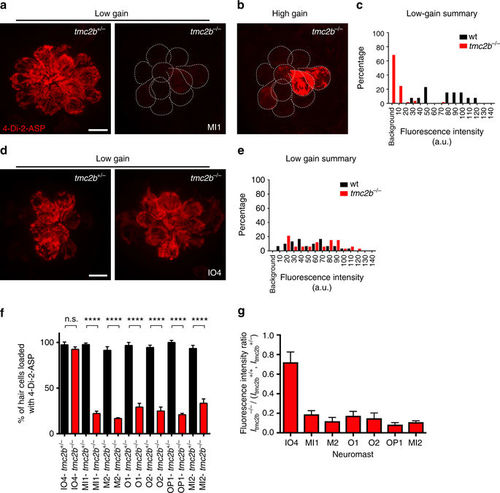
Spatial positioning of anterior neuromasts regulates diverse Tmc2b dependence. a Confocal micrographs of MI1 hair cells incubated with 4-Di-2-ASP from tmc2b +/− (left) or tmc2b −/− (right) animals at 6 dpf, viewed under low gain. b Hair cells from a, right, viewed under high gain. c Percentages of MI1 hair cells at different fluorescence intensities (n = 5). d Images and e percentages of hair cells at different fluorescence intensities from IO4 neuromasts (n = 5). f Percentages of hair cells of ALL neuromasts that take up 4-Di-2-ASP. ****One-way ANOVA with Holm-Sidak’s multiple comparisons test P < 0.0001. n values (het/homo): IO4 = 6/4, MI1 = 7/5, M2 = 5/3, O1 = 5/2, O2 = 5/4, OP1 = 5/3, MI2 = 5/5. g Mean whole-neuromasts normalized fluorescence intensity ratios, I tmc2b −/−/ (I tmc2b +/+, I tmc2b +/−). n values ≥ 5. Scale bar = 6 µm PHENOTYPE:
|
|
Hair cell PCP and neuromast position govern dependence of mechanotransduction channel function on Tmc2b. a–f Confocal images of hair cells from L1, LII.1, and IO4 neuromasts of tmc2b +/− or tmc2b −/− animals. FM1-43FX (red) uptake reveals functional channels, and phalloidin (cyan) shows hair bundle polarity. Qualitative maps (yellow) of micrographs from tmc2b −/− mutants show that subpopulations of posterior- and ventral-facing hair bundles preferentially function (red) in L1 and LII.1, respectively; in contrast, hair cell uptake in IO4 is non-biased. g In PLL neuromasts with A–P orientations of tmc2b −/− mutants, the vast majority (n = 21) of FM1-43FX uptake is by hair bundles that are posterior facing (P-facing). Similarly, in the A–P-oriented neuromasts of the ALL of tmc2b −/− (O1, O2, OP1, and MI2), the overwhelming majority of FM1-43FX uptake is by P-facing hair cells (n ≥ 5, except for O1, n = 4). Red bars represent tmc2b −/−, and black bars signify tmc2b +/+ and tmc2b +/−. h In the PLL neuromasts with D–V orientations of tmc2b −/− mutants, ventral-facing (V-facing) hair cells dominate the population that uptakes the fluorophore (n = 16). In ALL neuromasts with D–V orientations of tmc2b −/−, the patterns of hair cells that take up FM1-43FX are complex. For IO4, there is no significant difference between the numbers of D- and V-facing hair cells that loaded with FM1-43FX. The FM1-43FX loading percentages are 82.6 ± 11.1 % (n = 5) for V-facing hair cells and 83.0 ± 11.1 % (n = 5) for D-facing hair cells in tmc2b −/−, respectively. One-way ANOVA with Holm-Sidak’s multiple comparisons test, P = 0.9986. D–V-oriented neuromasts MI1 and M2 have opposite PCP-related loading preferences in mutants. In MI1 and M2 of tmc2b −/− animals, the percentages of V-facing hair cells that load with FM1-43FX are 36.1 ± 3.4% (n = 6) and 2.9 ± 2.9% (n = 5), respectively. Whereas, the percentages of D-facing hair cells from MI1 and M2 that load with FM1-43FX are 7.9 ± 3.6% (n = 6) and 44.9 ± 8.3% (n = 5), respectively (see Supplementary Table 1 for statistics). Corresponding images of MI1 and M2 displayed in Supplementary Fig. 7. Scale bar = 5 μm. Arrowheads, immature hair cells that do not take up FM1-43FX. *dying hair cell PHENOTYPE:
|
|
A mechanosensory map of Tmc2b dependence in the anterior and posterior lateral lines. In this graphic of a larval zebrafish tmc2b −/− mutant, red neuromasts have been characterized. Individual neuromasts with patterns of FM1-43FX uptake are displayed. Red hair cells take up FM1-43FX; gray hair cells do not. Below each schematized neuromast is the percentage of hair cells that load 4-Di-2-ASP (red text) in the tmc2b −/− mutant and the percentage of fluorescence intensity of hair cells of the mutant relative to hair cells of wild-type and heterozygous animals (black). Green arrows represent the directional sensitivity preserved in the mutant based on FM1-43FX uptake. Graphs of mean microphonic potentials from neuromasts directly above each plot are displayed. For IO4, tmc2b +/− = 9.2 ± 0.7 μV (n = 9), and tmc2b −/− = 8.9 ± 0.87 μV (n = 6). P value = 0.9546. For A–P oriented posterior neuromasts, tmc2b +/+, tmc2b +/− = 8.3 ± 0.37 μV (n = 20), and tmc2b −/− = 2.1 ± 0.8 μV (n = 15). ****P value < 0.0001. For D–V oriented posterior neuromasts, tmc2b +/− = 6 ± 0.49 μV (n = 4) and tmc2b −/− = 0 ± 0 μV (n = 6). ****P value = 0.0048. P values were obtained from the Mann–Whitney test. For 4-Di-2-ASP uptake assays of SO and IO neuromasts, n ≥ 5. n values for other neuromasts are listed in Figs. 3, 4 legends PHENOTYPE:
|
|
Tmc2a coordinates with Tmc2b to enable mechanotransduction in lateral line hair cells. a Stimulus-evoked microphonic potentials measured from posterior neuromasts with A–P-oriented hair cells from 6-dpf zebrafish larvae. The response in tmc2a −/− /tmc2b −/− is absent, bottom trace. b Graph of mean microphonic potentials from posterior neuromasts with hair cells with A–P orientations (n = 6). **Mann–Whitney test P = 0.0043. c, d Confocal images of hair cells from LII.1 and IO4 neuromasts of tmc2a +/− /tmc2b +/− or tmc2a −/− /tmc2b −/− animals labeled with FM1-43FX (red) and phalloidin (cyan). No dye was observed in LII.1 or IO4 of tmc2a −/− /tmc2b −/− animals. Scale bar = 6 μm PHENOTYPE:
|
|
Blueprint of the larval zebrafish’s lateral line axes of best sensitivity. a) Schematic of the axes of best sensitivity of neuromasts of the ALL and PLL of larval zebrafish. These neuromasts lay approximately on single planes or sheets on each of the lateral sides of the fish. Posterior neuromasts are slightly offset to show axes of best sensitivity. (b) Overall result of perpendicularly set axes of best sensitivity. |
|
Consequences of the tmc2b mutation on hair bundle morphology, hair cell presence, and ear and neuromast function. (a) Scanning electron micrograph of hair bundles of heterozygous animals with stereocilia (shorter, thinner) and kinocilia (longer, thicker). The stereocilia are linked at their tips by tip link filaments (red arrow head). (b) In the tmc2b-/- mutant, the hair bundle is morphologically normal at the ultrastructural level. Confocal micrographs of the L2 neuromasts of heterozygous (c) and tmc2b-/- mutant (d) zebrafish labeled with phalloidin (red) at 7 dpf. Planer cell polarity is normal in the mutant. Scale bar = 2.5 μm. (e) Mean hair cell numbers ± SEM in posterior neuromasts in tmc2b-/-, cdh23aj64a/aj64a, and controls of 6-dpf zebrafish. cdh23+/aj64a = 12.3 ± 0.6 (n = 32); cdh23aj64a/aj64a = 9.5 ± 0.4 (n = 39); tmc2b+/+, tmc2b+/- = 14.4 ± 0.6 (n = 31); tmc2b-/- = 9.0 ± 0.3 (n = 45). **** equal to P value < 0.0001 and *** represents P = 0.0067, Kruskal-Wallis analysis with a Dunn’s multi-comparison test. Representative extracellular recordings of microphonic potentials measured from ears (f,g) and neuromasts (h,i,j) are displayed (for comparison i is the same as in Fig. 3a). Ears from controls (67.95 ± 9.52 μV, n = 8) and mutant (69.3 ± 7.86 μV, n = 6) larvae at 8-9 dpf have robust responses at twice the stimulus frequency (P = 0.8518, Mann-Whitney test). Microphonic potentials from neuromast IO4 were not impacted by the tmc2b mutation. The responses of posterior A-P and D-V oriented neuromasts are most frequently absent (9 of 15 for A-P oriented and 6 of 6 for D-V oriented neuromasts); however, in some cases in A-P oriented, 6 of 15, responses were highly asymmetric, with a weakened amplitude for one direction of stimulus and no response for the other direction. Therefore, the 2f response became a 1f response. Red letters represent deflection dorsally (D), ventrally (V), anteriorly (A), and posteriorly (P). |
|
Variation of 4-Di-2-ASP uptake in hair cells within a single posterior neuromast of a tmc2b-/- mutant. (a and b) Micrographs of L1 neuromast hair cells exposed to 4-Di-2-ASP (top), histograms of individual hair cell uptake (middle), and quantitative maps of hair cell uptake (bottom) under low gain (a) and high gain (b). Scale bar = 6 μm. (a) Every posterior neuromast hair cell in tmc2b+/- larvae take up 4-Di-2-ASP, ranging from 30-110 fluorescence intensity units. In mutants, under low gain, hair cell fluorescence intensity values are negligible (<1 unit) for six cells and very low (1-20 units) for two cells. Background is defined as 0-1 unit. (b) Under high gain, 6 cells from a tmc2b-/- animal had negligible (<10 units) fluorescence intensities; however, two had increased intensities. In contrast, all hair cells of the cdh23aj64a/aj64a posterior neuromasts take up 4-Di-2-ASP. (Inset) Summary, percentages of hair cells at different fluorescence intensity units under low gain (n = 5). |
|
Roles of hair bundle polarity and neuromast position in the dependence of mechanotransduction channel function on Tmc2b for neuromasts MI1 and M2. Images of hair cells from neuromasts MI1 (a,b) and M2 (c,d) of tmc2b+/- and tmc2b-/- 6-dpf larvae. FM1-43FX (red) presence reveals functional channels, and phalloidin (cyan) shows hair bundle polarity. Qualitative maps (yellow) of micrographs indicate that MI1 hair cells with ventrally facing hair bundles and M2 hair cells with dorsally facing hair bundles preferentially function (red) in tmc2b-/- mutants. Scale bar = 5 μm. |