- Title
-
Modulating expression level of secreted Wnt3 influences cerebellum development in zebrafish transgenics
- Authors
- Teh, C., Sun, G., Shen, H., Korzh, V., Wohland, T.
- Source
- Full text @ Development
|
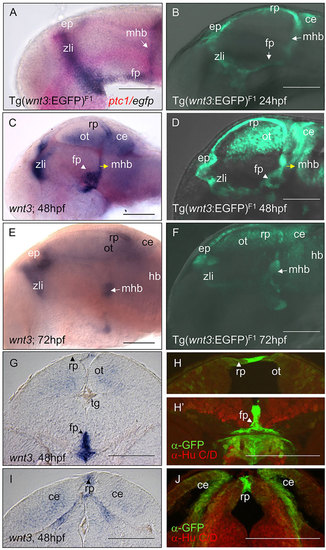
EGFP and Wnt3 expression in transgenics under control of the wnt3 promoter. A stable zebrafish transgenic line containing the 4kb wnt3 promoter drives EGFP expression in a wnt3-like manner with spatio-temporal correlation with endogenous transcripts. (A) egfp transcript expression (purple) in 4kb wnt3 promoter transgenic line Tg(-4.0wnt3:EGFP)F1 colocalized with ptc1 (pink) at the zona limitans intrathalamica (zli) at 24hpf. (B) An in vivo image of 24hpf Tg(-4.0wnt3:EGFP)F1 showing EGFP+ domains highlighted by the transgenic line. (C,E) wnt3 transcripts (purple) are detected in the cerebellum (ce), epithalamus (ep), floor plate (fp), optic tectum (ot), roof plate (rp), midbrain hindbrain boundary (mhb) and hindbrain (hb) of larvae at 48hpf (C) and 72hpf (E). (D,F) In vivo images of Tg(-4.0wnt3:EGFP)F1 at 48hpf (D) and 72hpf (F) showing similar EGFP+ domains. (G-J) Comparison of midbrain (G,H) and cerebellum (I,J) cross sections of wnt3 expression in a 48hpf zebrafish brain (G,I) with corresponding cross sections of Tg(-4.0wnt3:EGFP)F1 (H-J) that detected EGFP expression in similar regions. wnt3-positive domains include the optic tectum, floor plate and cerebellum. Co-immunohistochemical detection of EGFP and Hu-positive neurons showed that EGFP+ domains in the floor plate, floor plate and cerebellum are flanked by neurons (H,H′,J). Scale bars: 100�m. |
|
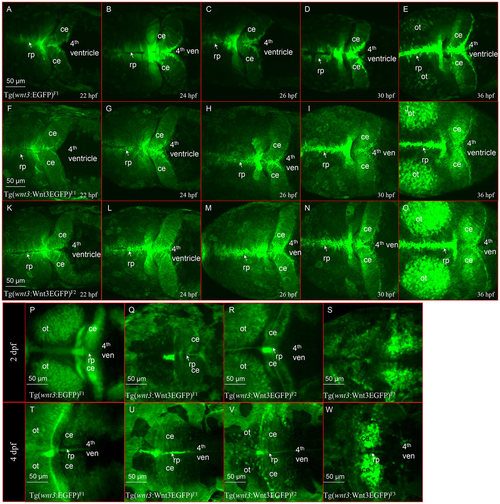
Spatio-temporal expression of wnt3 promoter-driven EGFP/Wnt3EGFP. Spatio-temporal expression of 4kb wnt3 promoter-driven EGFP/Wnt3EGFP influenced by genomic insertion of a transgene varies in strength. Tg(-4.0wnt3:EGFP)F1, Tg(-4.0wnt3:Wnt3EGFP)F1, Tg(-4.0wnt3:Wnt3EGFP)F2 and Tg(-4.0wnt3:Wnt3EGFP)F3 are abbreviated in the figure as Tg(wnt3:EGFP)F1, Tg(wnt3:Wnt3EGFP)F1, Tg(wnt3:Wnt3EGFP)F2 and Tg(wnt3:Wnt3EGFP)F3, respectively. (A-O) Initial spatio-temporal similarity of expression of EGFP and Wnt3EGFP is maintained in three independent transgenic lines. (P-W) Tg(-4.0wnt3:Wnt3EGFP)F3 maintains strong Wnt3EGFP expression in the cerebellum beyond 48hpf. Transgene expression in the optic tectum and cerebellum decreases from 48hpf in all other Wnt3 transgenics. 4th ven, fourth ventricle; ce, cerebellum; ot, optic tectum; rp, roof plate. |
|
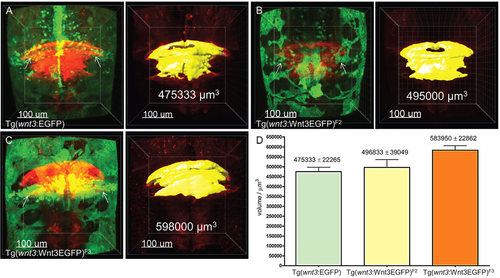
The size of the MHB and cerebellum segmental volume correlates with the level of Wnt3EGFP expression. (A-C) In vivo comparison of segmental volume in Tg(memKR15-16) in EGFP or different Wnt3EGFP transgenics background at 4dpf. Tg(memKR15-16)/EGFP (A), Tg(memKR15-16)/Wnt3EGFP [Tg(wnt3:Wnt3EGFP)F2] (B) and Tg(memKR15-16)/Wnt3EGFP [Tg(wnt3:Wnt3EGFP)F3] (C). KillerRed expression in the MHB and cerebellum of Tg(memKR15-16) is in red; EGFP or Wnt3EGFP expression in wnt3 transgenics is in green; and yellow represents a 3D view of the computed volume. (D) The increase in segmental volume in Tg(memKR15-16) is significant according to unpaired t-test comparing Tg(wnt3:EGFP)F1 vs Tg(wnt3:wnt3EGFP)F3 (P=0.0022). Arrows show expression of EGFP/Wnt3EGFP in the cerebellum. |
|
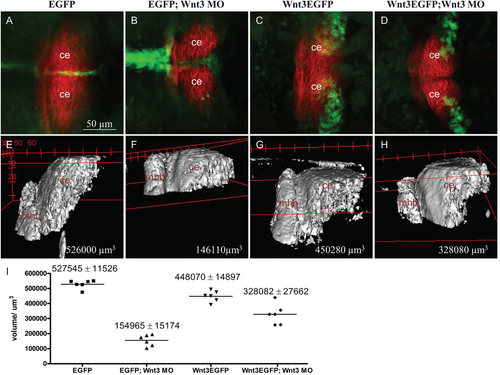
Expression of Wnt3EGFP in the cerebellum of Tg(wnt3:Wnt3EGFP)F3 partially compensates cerebellum growth in MO1-injected Wnt3 morphants. (A-D) Dorsal view of double transgenic Tg(memKR15-16) larvae co-expressing KillerRed with EGFP from Tg(wnt3:EGFP)F1 (A,B) or Wnt3EGFP from Tg(wnt3:Wnt3EGFP)F3 (C,D). KillerRed is expressed in the MHB and cerebellum. Reduction in KillerRed-positive cerebellum is partially compensated in 3dpf Tg(wnt3:Wnt3EGFP)F3 MO1-morphants (D). (E-H) 3D lateral view of KillerRed-positive MHB and cerebellum in Tg(wnt3:EGFP)F1 (E), MO1-injected Tg(wnt3:EGFP)F1 (F), Tg(wnt3:Wnt3EGFP)F3 (G) and MO1-injected Tg(wnt3:Wnt3EGFP)F3 at 3dpf (H). (I) Scatter plot of computed volumes. The decrease in segmental volume in Tg(memKR15-16) is significant between MO1-morphants and un-injected siblings. Unpaired t-test comparing Tg(wnt3:EGFP)F1 vs MO1-morphant Tg(wnt3:EGFP)F1, P<0.0001; Tg(wnt3:wnt3EGFP)F3 vs MO1-morphants Tg(wnt3:Wnt3EGFP)F3, P=0.0034. Segmental volume in MO1-morphant Tg(wnt3:Wnt3EGFP)F3 remains significantly higher than in MO1-morphant Tg(wnt3:EGFP)F1 (P=0.0003). ce, cerebellum; mhb, midbrain hindbrain boundary. |
|
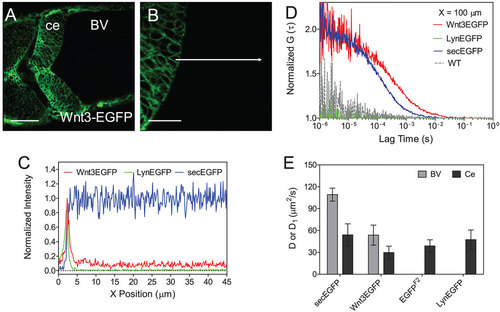
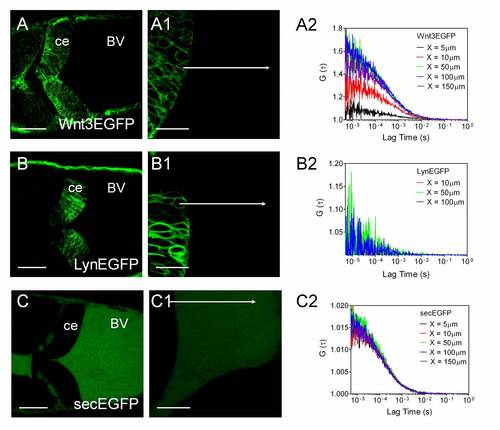
Wnt3EGFP, LynEGFP and secEGFP in the fourth brain ventricle. (A) Confocal image of zebrafish cerebellum expressing Wnt3EGFP at 34hpf. Scale bar: 50�m. (B) Zoom of A at 3� magnification with focus on the cerebellum boundary and flanking brain ventricle. Scale bar: 20�m. Images were taken in dorsal view. BV, brain ventricle; ce, cerebellum. The images were modified using Imaris to increase the contrast. (C) Normalized fluorescence intensity from the cerebellum boundary cell to the brain ventricle along the white arrow in B of Wnt3EGFP (red), LynEGFP (green) and secEGFP (blue). Data are the average of three scans of three embryos for each type to the highest point. (D) Normalized ACF curves taken within a ventricle at 100�m from the cerebellum boundary. Color-coding is the same as in C together with wild type (WT, dotted gray). The results show the free diffusion of Wnt3EGFP and secEGFP in the brain ventricle, whereas no fluorescence can be detected either for LynEGFP or for WT. (E) Diffusion coefficients extracted from fit for different types of EGFP-labeled proteins in both the cerebellum and the brain ventricle. secEGFP serves as an intercellular indicator of protein mobility in multicellular tissue and extracellular indicator in the brain ventricle. EGFP reporter Tg(-4.0wnt3:EGFP)F2 and LynEGFP transgenics Tg(-8.0cldnB:lynEGFP) serve as an indicator of intracellular protein mobility. Data are mean�s.d. Light gray bar, brain ventricle (BV); dark gray bar, cerebellum (ce). See also Fig. S2 and Tables S2, S3. |
|
Wnt3EGFP secretion is affected by the block of Porcupine. (A,B) Confocal images of zebrafish cerebellum expressing Wnt3EGFP and LynEGFP after C59 treatment. The embryos were treated with 5�M C59 10-28hpf (for details, see Materials and Methods). The samples were briefly soaked in 1� egg water before imaging in dorsal view, and FCS measurement. Scale bars: 50�m. (C-E) Diffusion coefficients (D1, D2) and protein membrane distribution (F2) extracted from fit at different conditions for both Wnt3EGFP and LynEGFP. (F) The results show that FCS signatures remain unchanged for LynEGFP, indicating that properties of the plasma membrane are not influenced by treatment or the drug function. Data are mean�s.d. Red bar, Wnt3EGFP; green bar, LynEGFP. Significance level, two-way t-test, *P<0.001. See also Table S3. |
|
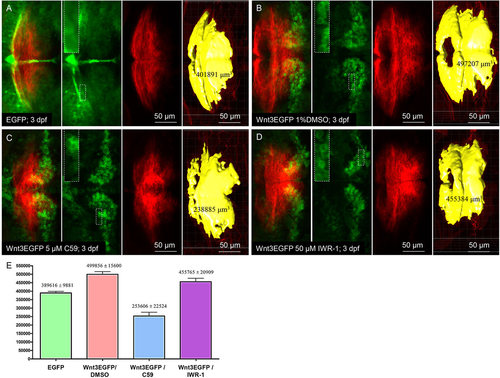
Exposure of Tg(wnt3:Wnt3EGFP)F3 larvae to Wnt inhibitor C59 decreased the segmental volume of cerebellum in Tg(memKR15-16). (A-D) In vivo images of 3dpf double transgenic larvae: control Tg(wnt3:EGFP)F1/Tg(memKR15-16) (A), Tg(wnt3:Wnt3EGFP)F3/Tg(memKR15-16) (B-D). All Tg(wnt3:wnt3EGFP)F3 double transgenic larvae were exposed to either 1% DMSO (B), 5�M C59 (C) or 50�M IWR-1 (D) at 36hpf-stage for 36h before assaying expression of fluorescent reporter at 72hpf. High-magnification view of representative EGFP/Wnt3EGFP-expressing cells from different treatment groups (insets). KillerRed expression in the MHB and cerebellum of Tg(memKR15-16) is in red; EGFP or Wnt3EGFP expression in wnt3 transgenics is in green; and yellow represents a 3D view of the computed volume. (E) Comparison of the resultant KillerRed-positive brain segment in Tg(memKR15-16). A significant difference was observed between segmental volumes in 1% DMSO-treated Tg(wnt3:Wnt3EGFP)F3 vs C59-treated Tg(wnt3:Wnt3EGFP)F3 (unpaired t-test, P=0.0001). |
|
Functional overexpression of recombinant Wnt3. The recombinant Wnt3 zebrafish fusion protein with EGFP at the C-terminal (Wnt3EGFP) is functional as overexpression in zebrafish produces a posteriorized brain phenotype typical of overt Wnt activity. (A) Bright field image of a malformed embryo with a posteriorized brain upon overt Wnt3EGFP expression under the ubiquitous CMV promoter. Injection of the Wnt3EGFP expression construct at blastula stage resulted in the deformed phenotype. (B) Tg(memKR15-16) expressed membrane localized KillerRed in the midbrain hindbrain boundary (mhb) was used to characterize the deformed brain phenotype. (C) The posteriorized brain phenotype is accompanied by the loss of the midbrain hindbrain boundary observed in Tg(memKR15-16) upon overt Wnt3EGFP expression. |
|
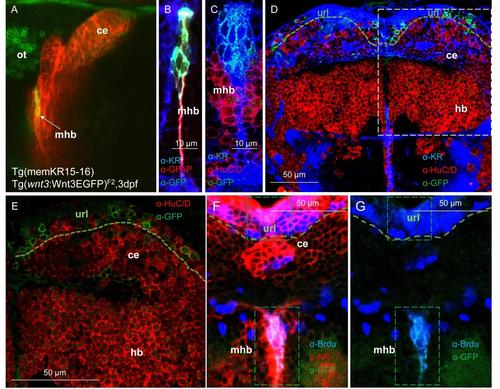
Cells expressing Wnt3EGFP in the midbrain hindbrain boundary and cerebellum are located in proliferation niches. (A) An in vivo image of a double transgenic larvae co-expressing the membrane localized KillerRed in the midbrain hindbrain boundary (mhb) and cerebellum of Tg(memKR15-16) and C terminal EGFP Wnt3 fusion proteins from Tg(wnt3:Wnt3EGFP)F2. Double transgenic larvae confirmed Wnt3EGFP expression in the mhb and the cerebellum. wnt3:Wnt3EGFP+ cells in the cerebellum are predominantly localized to the upper rhombic lip (url). (B-E)Wnt3EGFP cells at midbrain hindbrain boundary (mhb) are glial cells (B) as it stained positively for GFAP (glial fibrilary acidic protein) and are negative for the neuronal marker HuC/D (C-E). (F-G) Midline restricted Wnt3EGFP cells at midbrain hindbrain boundary (demarcated by membrane KR expression in this domain) are proliferating cells since they stained positively for Brdu. |
|
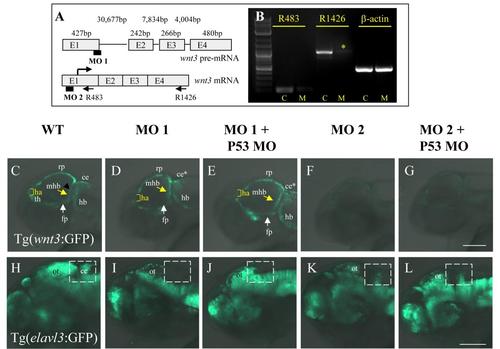
Interference with Wnt3 function disrupts cerebellar differentiation. (A) Organization of the wnt3 pre-mRNA and regions targeted by wnt3 MOs, MO 1 (targets wnt3 exon1-intron 1 boundary) and MO 2 (targets wnt3 5′UTR). The length of each exon and intervening intron is indicated. (B) RT-PCR analysis of wnt3 in wild type and morphants (MO1) at 30 hpf. Wnt3 knockdown upon microinjection of MO1 interfere with formation of wild type wnt3 transcripts highlighted by (*). Abbreviation: control (C); MO1 injected morphants (M). (C-G) Only microinjection of translation blocking MO (MO2) whose recognition site is present in the wnt3 regulatory element successfully interferes with GFP expression in Tg(wnt3:EGFP)F1. (H-L) Wnt3 deficiency through either microinjection of MO1 or MO2 with or without p53 MO consistently reduces GFP-positive neurons in the cerebellum (ce) of 72hpf Tg(elav:GFP) embryos. Insert scale bar is 50 �m. |
|
Wnt3EGFP fractions are in a stable equilibrium. (A) Confocal images of zebrafish cerebellum (with optic tectum) expressing Wnt3EGFP at 28 hpf (A1), 34 hpf (A2), 48hpf (A3). The images show that Wnt3EGFP expression level differs in different regions of the cerebellum: lateral side (LS) and dorsal midline (DM). Images were taken in dorsal view. Scale bar, 200 �m. (B) Confocal image of zebrafish cerebellum (with optic tectum) expressing H2A-EGFP at 36 hpf shows that at this stage the nucleus occupies most of the inner space of the cell. Image was taken in dorsal view. Scale bar, 200 �m. (C, D and E) Diffusion coefficients (D1, D2) and fraction of slow moving component (F2) extracted from fit at different development stages and regions in the cerebellum (LS and DM). The expression level in DM at 48 hpf was too low to be measured, therefore only data from LS were acquired. Data are mean � SD. Light grey bar, LS. Dark grey bar, DM. (F) Fraction of slow moving component (F2) does not change with the protein expressing level, which is indicated by the normalized intensity count rate, for both Wnt3EGFP (red dot) and LynEGFP (green dot). Solid lines represent the mean and areas represent the SD respectively. Abbreviations: Ce: cerebellum; BV: brain ventricle. |
|
Secreted Wnt3EGFP in the brain ventricle. (A, B, C) Confocal images of zebrafish cerebellum expressing Wnt3EGFP, LynEGFP and secEGFP at 34 hpf. Scale bar, 50 �m. Images were taken in dorsal view. A is the same as Fig. 6A. (A1, B1, C1) Three times zoom in of (A, B, C) with focus on the boundary of the cerebellum and brain ventricle. Scale bar, 20 �m. X-scanning was performed along the white arrow. Data were analysed using ImageJ. Results are shown in Fig. 6C in the main text. (A2, B2, C2) ACF curves of FCS measurements along the arrows at different distance from the cell boundary: 5�m (black); 10�m (red); 50�m (green); 100�m (blue); 150�m (purple). FCS data at 100 �m were normalized and plotted in Fig. 6D in the main text for comparison. Abbreviations: Ce: cerebellum; BV: brain ventricle. |
|
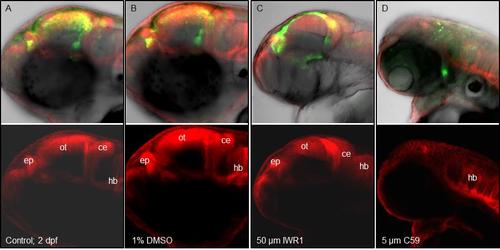
Effect of C59 Wnt inhibitor on brain patterning. Exposure of Tg(wnt3:Wnt3EGFP)F2 to C59 Wnt inhibitor suppresses Wnt3EGFP expression and interferes with brain patterning. Dorsal domains of the brain are highlighted by the expression of membrane tethered KillerRed in the epithalamus, optic tectum, cerebellum and hindbrain of Tg (memKR15-8). Double transgenic larvae were treated with 5 �M C59; 50 �M IWR-1 or 1% DMSO from 10 hpf to 48 hpf. Successful interference of Wnt function by C59 results in decreased Wnt3EGFP expression that correlated with defective brain patterning indicated by the absence of epithalamus (ep), optic tectum (ot) and cerebellum (ce) in C59 treated larvae (D). |