- Title
-
rnaset2 mutant zebrafish model familial cystic leukoencephalopathy and reveal a role for RNase T2 in degrading ribosomal RNA
- Authors
- Haud, N., Kara, F., Diekmann, S., Henneke, M., Willer, J.R., Hillwig, M.S., Gregg, R.G., Macintosh, G.C., G�rtner, J., Alia, A., and Hurlstone, A.F.
- Source
- Full text @ Proc. Natl. Acad. Sci. USA
|
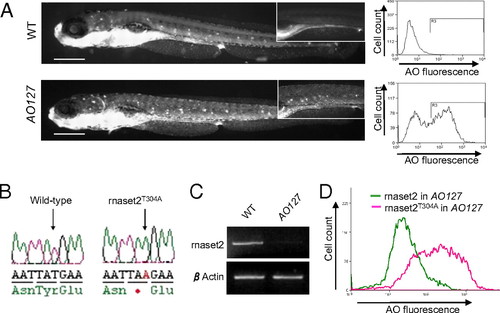
AO127 mutant animals are deficient in rnaset2. (A) AO127 mutants displayed a higher uptake of AO compared with WT embryos, quantifiable by flow cytometry. (Note the intense fluorescence common to both animals is a result of AO within the lumen of the pharynx and intestine, autofluorescence of residual yolk, and ready uptake of AO by olfactory neurons and hair cells within neuromasts.) (Scale bars: 200 μm.) (B) Sequencing of the gene zgc:113369, coding for the zebrafish orthologue of human RNASET2, identified a T-to-A transversion in the mutant sample. (C) RT-PCR indicates a dramatic down-regulation of rnaset2 mRNA in AO127 mutants compared with WT siblings. β-Actin serves as a control for RNA integrity and equal input cDNA. (D) Suppression of excess AO staining in AO127 mutants by injection of WT rnaset2 mRNA but not mutant mRNA. |
|
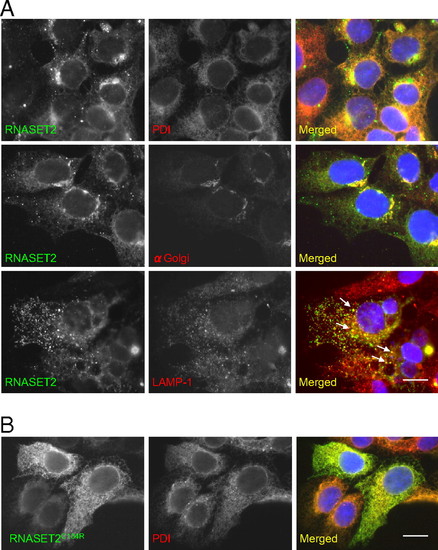
Subcellular localization of RNASET2. HEK293 cells were transfected with myc-epitope tagged WT RNASET2 (A) and the mutated version, RNASET2 C184R (B). Immunofluorescence for endoplasmic reticulum (PDI), Golgi, and late endosome (LAMP-1) markers show partial colocalization with WT RNASET2, suggesting that RNASET2 enters the secretory pathway. The mutated version colocalized completely with the PDI, suggesting ER retention. (Scale bar: 10 μm.) |
|
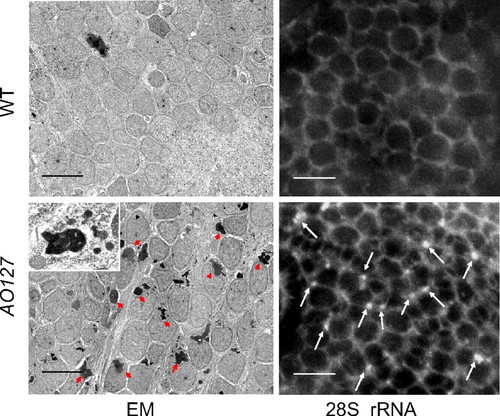
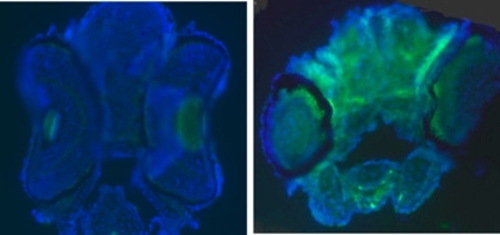
Brain neurons of AO127 mutants contain lysosomes engorged with rRNA. Brain transverse sections from embryos 5 d after fertilization. EM revealed abundant electron-dense vesicles in the neurons of AO127 mutants (some of these are indicated by red arrowheads; Inset, electron-dense material is bounded by a limiting membrane). (Scale bars: 5 μm). In situ hybridization revealed accumulation of rRNA aggregates (white arrows) in the cytoplasm of AO127 mutant neurons. (Scale bars: 10 μm.) PHENOTYPE:
|
|
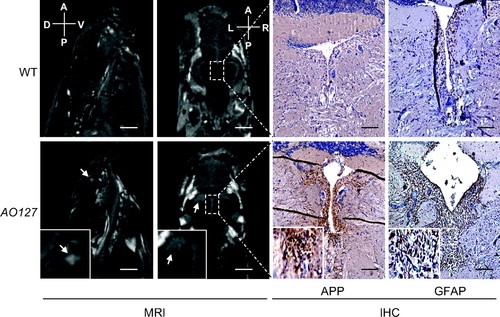
White matter anomalies in AO127 mutants. T2-weighted MR images of the brain of WT and mutant AO127 adult zebrafish. Sagittal images indicate the presence of a white matter anomaly (white arrow) in telencephalon of mutant, which was not seen in WT. Coronal images show the presence of lesions adjacent to the ventricles (white arrow). IHC was carried out on the same samples used for MRI. Axonal disruptions around ventricles were seen using APP as a marker, as well as a higher number of astrocytes using GFAP as a marker, suggesting the presence of damaged neurons and inflammation around the lesions. (Scale bars: 1 mm for MR sagittal images, 500 μm for coronal images, 100 μm for immunohistochemistry.) PHENOTYPE:
|
|
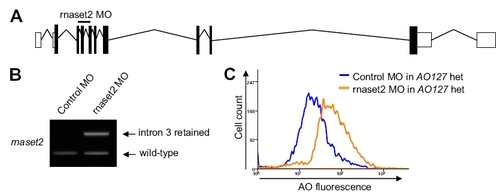
Confirmation of the AO127 mutation by knockdown. (A) Schematic of the zebrafish rnaset2 gene mutated in AO127 mutants showing exon?intron structure and the position of a MO designed to interrupt splicing of coding exon 3. (B) RT-PCR revealed the presence of mRNA retaining intron 3 from animals injected with the rnaset2 MO. Residual WT mRNA is presumably maternal. β-Actin serves as a control for RNA integrity and equal input cDNA. (C) Injection of rnaset2 MO, but not control MO, in animals heterozygous for rnaset2 resulted in an increase in AO staining. |
|
Specificity of the 28S rRNA probe. Frozen sections of WT embryos at 5 d after fertilization hybridized with a control sense 28S rRNA probe or antisense probe (green). Nuclei were counterstained with DAPI (blue). Note green autofluorescence in lens of section stained with sense probe. (Scale bar: 100 μm). |
|
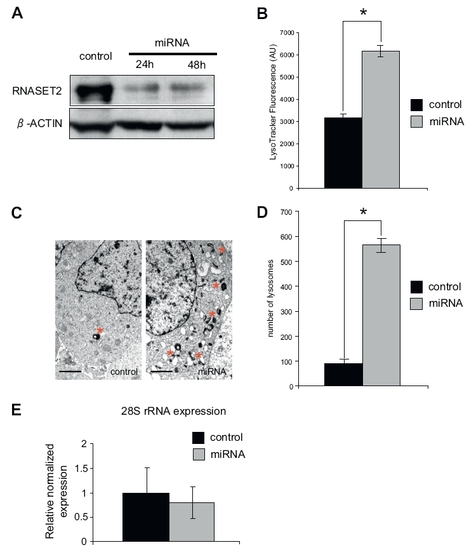
RNASET2 depletion in HEK 293 cells results in increased lysosome biogenesis without rRNA accumulation. (A) RNASET2 protein levels in HEK 293 cells transfected with control construct (control) or specific miRNA targeting RNASET2 detected by immunoblotting. (B) Cells transfected as in A for 48 h were stained with LysoTracker and fluorescence intensity measured in mCherry-positive cells using a flow cytometer (n = 3 independent experiments; error bars represent SEM). (C) EM examination of mCherry-positive cells transfected as in A for 48 h revealed more numerous lysosomes following RNASET2 knockdown. (Scale bar: 1 μm.) (D) Quantification of lysosome numbers from C. Mean number of lysosomes per EM grid quadrant measuring 500 μm2 were enumerated (n = 3 independent experiments; error bars represent SEM; *P < 0.01, independent-samples t test). (E) Quantitative PCR was performed for 28S RNA: histogram depicts relative fold change in expression normalized to GAPDH ± SEM (n = 3). |
|
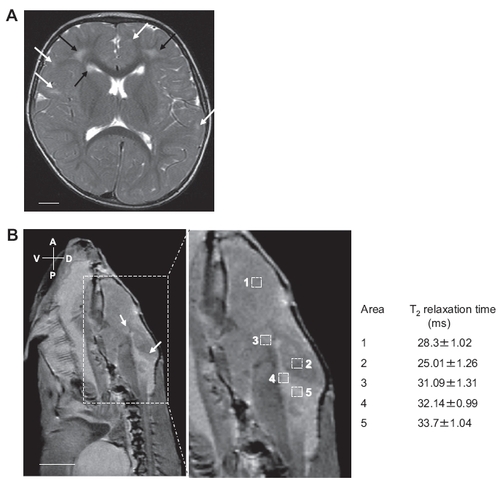
Comparable MR presentations of RNase T2-deficient human and zebrafish. (A) Axial T2-weighted cerebral MR image of a patient with RNASET2-deficient cystic leukoencephalopathy. The image indicates multifocal white matter abnormalities adjacent to the ventricles (black arrows) and in deep white matter (white arrows). (Scale bar: 1 cm). (B) T2 relaxation time measurement of the brain of a mutant zebrafish containing nullizygous mutation in rnaset2 gene. Sagittal MR image of the brain of an adult AO127 mutant zebrafish indicate the presence of lesions adjacent to the ventricles (white arrows) and were of similar intensity as that of CSF present in ventricles. (Boxed area, enlarged at right.) Areas were selected for T2 relaxation time measurements within healthy brain region (1, 2), in ventricles (4), and in lesion outside (3) or adjacent to ventricle (5). The lesions adjacent to ventricles have similar T2 relaxation time as that of ventricles, suggesting that these lesions may be filled with solution similar to CSF. PHENOTYPE:
|
|
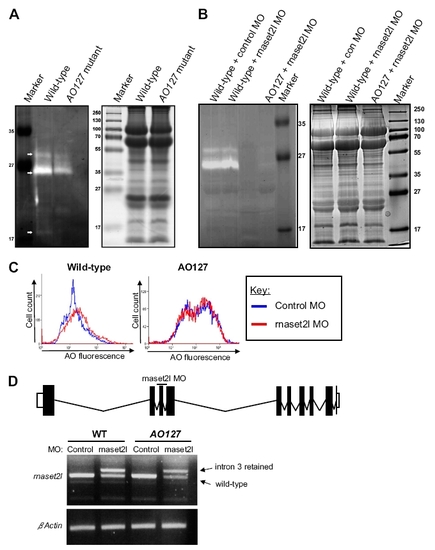
Decreased RNase T2 activity in AO127 mutants and redundancy with rnaset2l. (A) Left: In-gel activity assay was performed at pH 6.0 to analyze RNase T2 activity in protein extracts from WT and AO127 mutant embryos at 3 d after fertilization. White arrows indicate species present in only WT or greatly diminished in AO127 mutants. Note the different molecular weight species result from glycosylation (1) and/or possibly from proteolytic processing of RNase T2 (2). Right: SDS/PAGE gels run in parallel and stained with Coomassie blue to verify loading amounts and protein quality. (B) As in A, only samples prepared from embryos injected with a standard control oligonucleotide (control MO) or antisense oligonucleotide targeting rnaset2l (rnaset2l MO) are shown. (C) Embryos at 3 d after fertilization injected with control MO or rnaset2l MO were stained with AO, disaggregated, and subjected to flow cytometry. (D) Depiction of exon?intron structure of the rnaset2l gene and the position of a MO designed to interrupt splicing of exon 3. RT-PCR revealed the presence of mRNA retaining intron 3 from animals injected with the rnaset2l MO. Residual WT mRNA is presumably maternal. β-Actin serves as a control for RNA integrity and equal input cDNA. |