- Title
-
Novel RPL13 variants and variable clinical expressivity in a human ribosomopathy with spondyloepimetaphyseal dysplasia
- Authors
- Costantini, A., Alm, J.J., Tonelli, F., Valta, H., Huber, C., Tran, A.N., Daponte, V., Kirova, N., Kwon, Y.U., Bae, J.Y., Chung, W.Y., Tan, S., Sznajer, Y., Nishimura, G., N�reoja, T., Warren, A.J., Cormier-Daire, V., Kim, O.H., Forlino, A., Cho, T.J., M�kitie, O.
- Source
- Full text @ J. Bone Miner. Res.
|
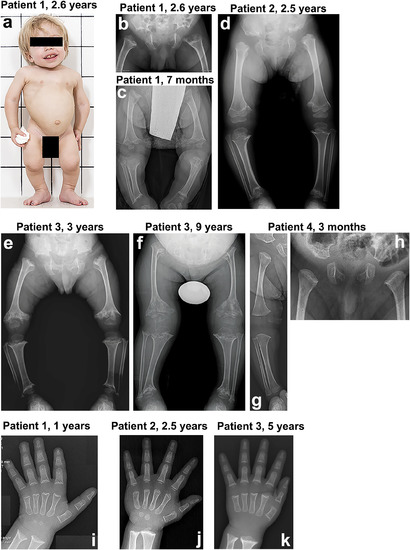
Clinical and radiological features of the four index patients with spondyloepimetaphyseal dysplasia. (A) Patient 1 at 2.6?years. The clinical features include mild coarseness of facial features with short nose, depressed nasal bridge and hypomineralization of the teeth, severe short stature with short neck, trunk, and limbs, narrow thorax, protruding abdomen, and flexion contractures of the hip and knee joints. Pelvis and lower limbs in patient 1 (B, C), patient 2 (D), patient 3 (E, F), and patient 4 (G, H). The iliac bones are short and broad with serrated iliac crests. The acetabulae are flat and wide with spur?like projections at the medial and lateral margins. The ischia and pubes are thick with serration at the lateral margin of the ischia. The femora are mildly bowed. The metaphyses of the long bones are wide, cupped, and severely irregular (arrows). The capital femoral epiphyses display significantly delayed ossification, while the other epiphyses of the long bones show normal maturation. The epimetaphyseal changes in patient 3 progressively worsened with age (E, F). Megaepiphyses are found at the knee and ankle. Wrist and hand in patient 1 (I), patient 2 (J), and patient 3 (K). Metaphyseal cupping and irregularity of the radius and ulna are apparent in patients 1 and 3, while those are mild in patient 2. Metaphyseal cupping and irregularity of the metacarpals and proximal phalanges are also found in patients 1 and 3. Delayed carpal bone age is detected in all three patients, but it is more severe in patients 1 and 3. No carpal ossifications are visible in patient 3 at 5?years. Brachydactyly is not apparent in all. |
|
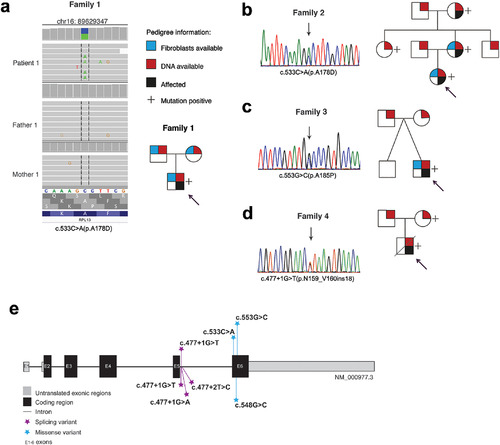
Genetic findings in the RPL13 gene. Genetic variants and pedigrees of the four families. RPL13 mutations were identified by WGS in family 1 (A), by Sanger sequencing in families 2 and 3 (B, C), and by WES followed by Sanger sequencing in family 4 (D). Schematic illustration of the RPL13 gene showing all the variants identified in this report (marked above the gene) and previously reported (below the gene) (E). |
|
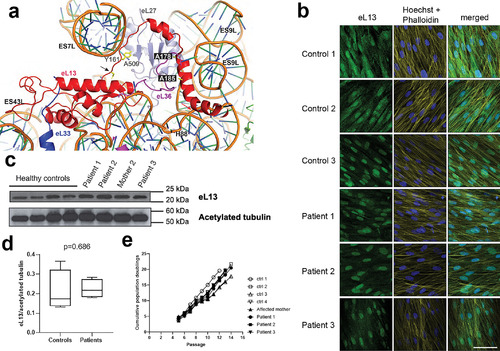
Expression of the eL13 protein in dermal fibroblasts from mutation?positive and control subjects. (A) Ribbon representation of eL13 bound to the human 60S subunit (based on pdb code 4V6X). Residues in eL13 targeted by the two missense mutations are highlighted in black boxes. The starting position of the affected region caused by the intronic mutation at N159 (p.N159_V160ins18) (black arrow) and the potentially disrupted interaction between Y161 and nucleotide A509 are highlighted. The start of the frameshift deletion harbored by our zebrafish model, p.L191Afs*32, which occurs at the point where the C?terminal ??helix of eL13 binds to the rRNA, is also marked (red arrow). Ribosomal proteins that interact with eL13 (red) include eL33 (blue), eL36 (purple), or eL27 (gray) are shown. Figure was generated using the PyMOL molecular graphics system, version 2.0.7 (Schr�dinger, LLC). (B) Representative immunofluorescence images of dermal fibroblasts from patients and healthy controls showing staining for eL13 (column 1), structural dyes Hoechst and Phalloidin (column 2), and merged (column 3). Scale bar = 30 ?m. (C) Western immunoblotting (WB) demonstrated expression of the eL13 protein in dermal fibroblasts from mutation?positive subjects at similar levels as in fibroblasts from control subjects. Acetylated tubulin was used as a loading control. (D) Densitometry analysis of the immunoblot confirmed the lack of significant differences?in eL13 expression (mean?�?SD, Student's t test). (E) Dermal fibroblasts from patients and controls showed similar growth kinetics over time in culture. Control 1 = male, age 29?years; control 2 = female age 30?years; control 3 = male age 17?years; control 4 = female age 31 years. |
|
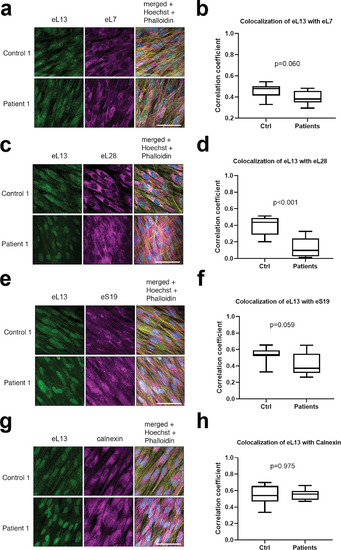
Comparison of colocalization of eL13 with other ribosomal proteins and with ER. Representative immunofluorescence images of dermal fibroblasts from patient 1 and one healthy control showing co?staining of eL13 with eL7 (A), eL28 (C), eS19 (E), and with calnexin as a marker of ER (G). Scale bars = 30??m. 3D colocalization of mutant and WT eL13 with the selected ribosomal markers was analyzed from entire z?stacks of three samples from patients 1 to 3 (n =?3) and each healthy control (n =?3) using the colocalization test plugin of ImageJ Fiji and is expressed as correlation coefficient (B, D, F, H). Box plots with median and range; p values from Mann?Whitney U test. |
|
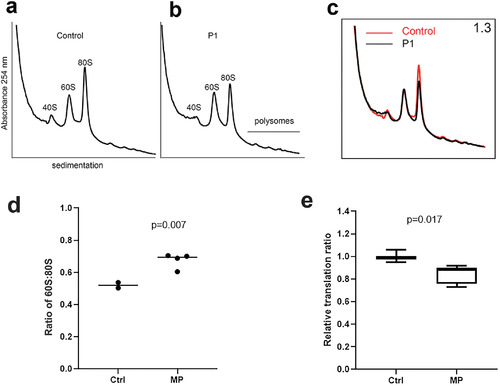
Sucrose profiles and global translation in patients 1, 2, and 3. Comparison of ribosomal fractions from fibroblasts of healthy control parent (A) and affected patient 1 (P1) heterozygous for the p.A178D RPL13 mutation (B). (C) Overlay of sucrose gradient profiles derived from healthy control parent (red line) and affected individual P1 (black line). Ribosomal subunit ratio (60S:80S) is indicated in the top right corner. (D) Box plot showing the results of the 60S:80S from fibroblasts of healthy controls (parents of patient 1) and mutation?positive (MP) subjects (patients 1 to 3 and mother of patient 2), each of whom is heterozygous for an RPL13 mutation. An increase median value for the 60S:80S ratio is observed in the MP group. The p value from one?sample t test comparing mutation?positive group against the median of the control samples. (E) Relative translation ratio from OP?puro translation assay. Global protein rate is reduced in the MP subjects (patients 1 to 3 and mother of patient 2) compared with controls (n =?2). Data are presented as mean and interquartile range. Experiments were performed twice, in duplicate or triplicate/donor/experiment, and analyzed using Mann?Whitney U test. |
|
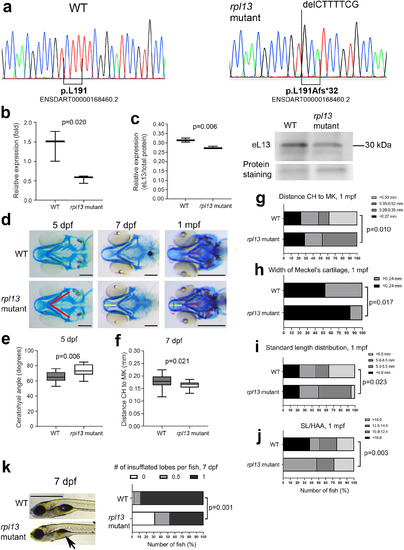
Evaluation of the rpl13L191Afs/L191Afs zebrafish. (A) Sanger sequencing confirmed the genotype of the homozygous rpl13L191Afs/L191Afs (rpl13 mutant) fish. (B) qPCR performed on RNA extracted from three WT and three rpl13L191Afs/L191Afs reveals a partial activation of nonsense?mediated mRNA decay in mutant zebrafish compared with WT. The p value from Student's t test. (C) WB performed on protein extracts from three WT and three rpl13L191Afs/L191Afs fish reveals reduced expression of eL13 in mutant fish compared with WT. The p value from Student's t test. (D) Alcian blue staining of control (WT) and rpl13L191Af/L191Afs zebrafish at 5 dpf, 7 dpf, and 1 mpf. Compared with WT, cartilage deformities in the head of the rpl13L191Afs/L191Afs are evident. Red lines indicate the ceratohyal (CH) angle, yellow line indicates the distance between the tip of CH and the tip of Meckel's (MK) cartilages, and green line indicates the MK width. Scale bar at 5 and 7 dpf = 200??m; scale bar at 1 mpf = 500??m. (E) At 5 dpf, rpl13L191Af/L191Afs show a significant increase of the CH angle compared with WT and (F) at 7 dpf a significant reduction in the distance between the CH and MK cartilages is evident in mutants compared with WT. The p values from Student's t test. (G) At 1 mpf, the frequency of fish with short distance (<0.53?mm) between the CH and MK cartilages and (H) the frequency of fish with narrower MK cartilage were significantly higher in mutants compared with WT. The p values from chi?square test. (I) Standard length (SL) distribution is significantly different in WT compared with rpl13 mutants at 1 mpf, with a higher number of long fish (>6.5?mm) in the WT population compared with mutants. The p value from chi?square test. (J) A higher frequency of fish with high SL/HAA (height at anterior margin of anal fin) ratio is found in the mutant group compared with WT group. Morphometric parameters (panels G?J) were categorized by global quartiles, presented as number of fish (%) in each category, and frequencies analyzed by chi?square test. (K) Swim bladder inflation in control (WT) and rpl13L191Af/L191Afs zebrafish at 7 dpf. Arrows indicate a partially inflated lobe in mutants compared with WT. Scale bar = 1?mm. Quantitative analysis shows a significant delay in the swim bladder inflation of the first lobe in rpl13L191Af/L191Afs compared with WT. The p values from crosstabs with chi?square test for frequencies. PHENOTYPE:
|