- Title
-
Functional constraints on SoxE proteins in neural crest development: the importance of differential expression for evolution of protein activity
- Authors
- Lee, E.M., Yuan, T., Ballim, R.D., Nguyen, K., Kelsh, R.N., Medeiros, D.M., McCauley, D.W.
- Source
- Full text @ Dev. Biol.
|
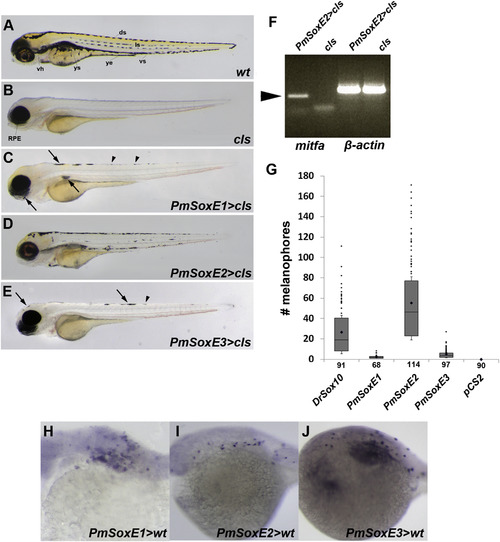
Melanogenesis in cls mutants resulting from expression of lamprey SoxE sequences inserted into pCS2+ vectors. (A-E) 96 hpf zebrafish larvae. (A) wildtype. (B) cls; RPE, retinal pigment epithelium. Expression of PmSoxE1 (C), PmSoxE2 (D), and PmSoxE3 (E) in cls mutants. (F) RT-PCR of PmSoxE2-injected cls mutants. mitfa expression in a cls embryo resulting from PmSoxE2 injection (lane 1) is absent in the control cls embryo (lane 2). β-actin internal control (lanes 3-4). (G) box and whisker plot indicating the number of melanophores in zebrafish injected with Drsox10 (positive control), lamprey PmSoxE1, PmSoxE2, and PmSoxE3, and cls embryos mock-injected with empty pCS2 vector as a negative control. Whiskers represent standard error. Each dot above the positive whiskers represents individual data points of the first quartile. Sample sizes are indicated under each bar along the X-axis. Comparison of means among samples all resulted in statistical significance (ANOVA, p-value<0.05). (H-J) in situ hybridization using lamprey SoxE probes in zebrafish wildtype embryos injected with either lamprey SoxE1 (H), SoxE2 (I), or SoxE3 (J) reveals similar mRNA expression, suggesting lamprey SoxE constructs injected into zebrafish embryos yields a comparable pattern and expression level irrespective of the lamprey SoxE gene sequence. Results shown are representative of 19/58 (PmSoxE1), 45/229 (PmSoxE2), and 90/356 (PmSoxE3) injections of lamprey constructs into early cleavage stage wildtype zebrafish embryos. vh, ventral horn; ys, yolk sac; ye, yolk extension; ds, dorsal stripe; ls, lateral stripe; vs, ventral stripe. Arrows point to melanophores, arrowheads point to iridophores. Orientation: anterior facing left. |
|
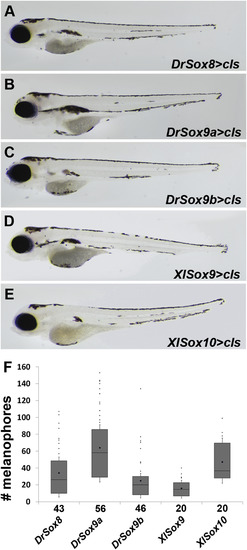
Gnathostome Sox8, Sox9, and Sox10 sequences inserted into Tol2 vectors can all promote melanogenesis in cls mutants. (A?E) 96 hpf zebrafish larvae. Expression of Drsox8 (A), Drsox9a (B), Drsox9b (C), XlSox9 (D), and XlSox10 (E) in cls mutants. (F) box and whisker plot indicating the number of melanophoresrescued. Whiskers represent standard error. Each dot above the positive whiskers represents individual data points of the first quartile. Sample sizes are indicated under each bar on the X-axis. |
|
Melanogenesis in cls mutants resulting from expression of PmSoxE1 and PmSoxE2 sequences inserted into Tol2 vectors. (A-H) 96 hpf zebrafish larvae. Expression of PmSoxE1 (A-D) and PmSoxE2 (E-G) and wildtype melanogenic phenotype (H). (I-V) pigmentation rescue phenotypes categorized as described in the text. Arrows indicate the absence of iridophores in cls mutant larvae (G) versus their presence in wildtype siblings (H). |
|
PmSoxE2 promotes differentiation of xanthophores in cls mutant zebrafish. (A-B) 76 hpf zebrafish embryos stained with methylene blue to detect presence of xanthophores. (A) cls mutant lacking differentiated xanthophores. (B) xanthophores in cls mutants injected with the pCS2-PmSoxE2 construct. (C) inset in ?B? ; higher magnification highlights the presence of xanthophores following PmSoxE2 injection. (D) morphology of xanthophores in a wildtype 76 hpf embryo. Orientation: anterior facing left. |
|
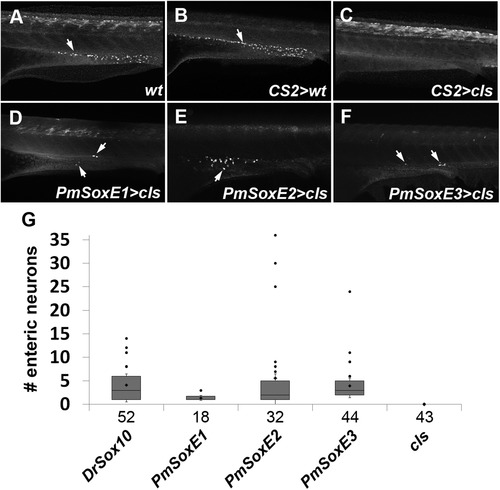
pCS2-PmSoxE heterospecific expression promotes differentiation of enteric neurons along the larval gut in cls mutants. (A-F) anti-HuC/D immunofluorescence in 96 hpf zebrafish. (A) wildtype. (B) control mock-injected wildtype. (C) cls embryo mock-injected with empty pCS2 vector. Note the absence of HuC/D-positive enteric neurons in cls embryos. Enteric neurons are present in cls mutants injected with lamprey PmSoxE1 (D), PmSoxE2 (E), and PmSoxE3 (F) constructs (arrows in D-F). (G) box and whisker plots indicate the number of enteric neurons present in zebrafish injected with Drsox10, PmSoxE1, PmSoxE2, and PmSoxE3 constructs. Whiskers represent standard error. Each dot above the positive whiskers represents individual data points of the first quartile. Sample sizes are indicated under each bar on the X-axis. Arrows indicate anti-HuC/D positive enteric neurons. Orientation: anterior facing left. |
|
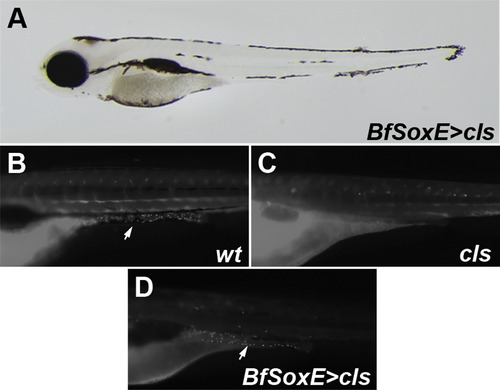
Amphioxus SoxE promotes differentiation of melanophores and enteric neurons. (A) differentiated melanophores in 96 hpf cls mutants injected with a Tol2:BfSoxE construct. (B-D) anti-HuC/D immunofluoresence in 96 hpf zebrafish indicates the presence of enteric neurons. (B) control wildtype. (C) cls mutants lack HuC/D-positive enteric neurons. (D) BfSoxE promotes differentiation of enteric neurons in cls mutants. Orientation: anterior facing left. |
|
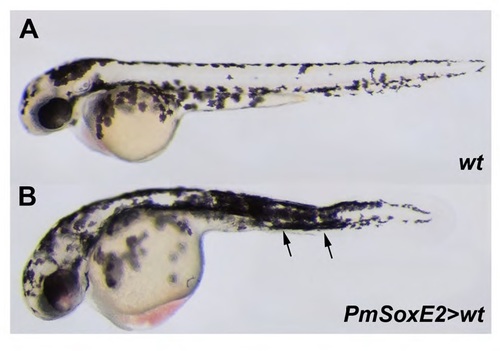
Lamprey SoxE2 expression in wildtype zebrafish causes excessive pigmentation. (A) pigmentation in a 56 hpf zebrafish embryo. (B) Excess pigmentation, especially in the trunk region, in a 56 hpf zebrafish embryo following injection with SoxE2. Orientation: anterior facing left. |
|
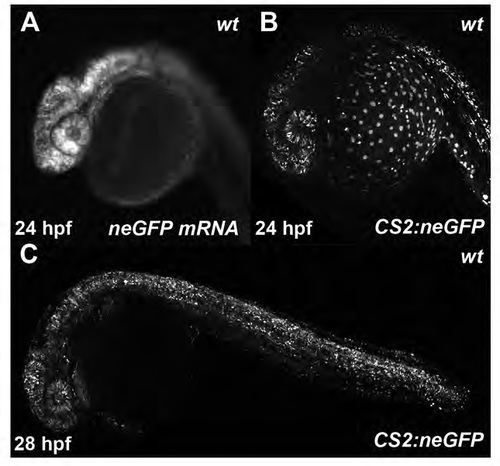
GFP expression in wildtype zebrafish embryos injected with either neGFP mRNA or CS2:neGFP plasmid. (A - C) 24 - 28hpf embryos show similar mosaic expression irrespective of mRNA or plasmid injection, highlighting the effectiveness of plasmid injection. |
Reprinted from Developmental Biology, 418(1), Lee, E.M., Yuan, T., Ballim, R.D., Nguyen, K., Kelsh, R.N., Medeiros, D.M., McCauley, D.W., Functional constraints on SoxE proteins in neural crest development: the importance of differential expression for evolution of protein activity, 166-78, Copyright (2016) with permission from Elsevier. Full text @ Dev. Biol.