- Title
-
Zebrafish early cardiac connexin, Cx36.7/Ecx, regulates myofibril orientation and heart morphogenesis by establishing Nkx2.5 expression
- Authors
- Sultana, N., Nag, K., Hoshijima, K., Laird, D.W., Kawakami, A., and Hirose, S.
- Source
- Full text @ Proc. Natl. Acad. Sci. USA
|
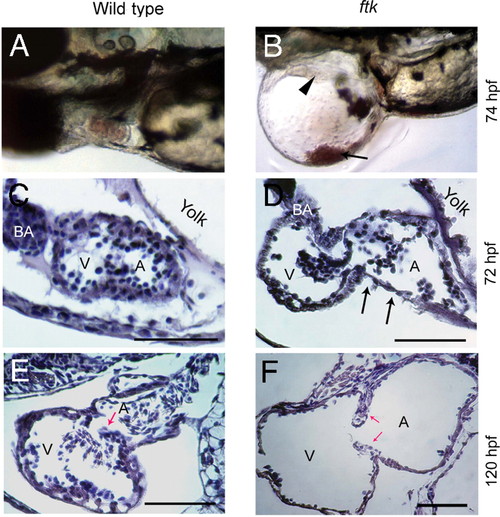
Progressive heart malformation in zebrafish ftk mutant. (A and B) Gross heart morphology of ftk mutant. Wild-type (A) and ftk mutant embryos (B) at 74 hpf. Arrowhead indicates heart perforations in the ftk mutant; arrow points to blood cells accumulated in the pericardiac cavity. (C?F) Sagittal sections of the heart. At 72 hpf (C and D), a dilated atrium (A) and ventricle (V) are evident in the ftk mutant. The cardiac muscles appear to be thinner (arrows in D), and the dilation of heart chambers is under way. At 120 hpf (E and F), the heart chamber dilation becomes more severe in ftk mutants with thinner ventricular and atrial walls. Red arrows indicate the valve leaflets. (Scale bars: 150 μm.) PHENOTYPE:
|
|
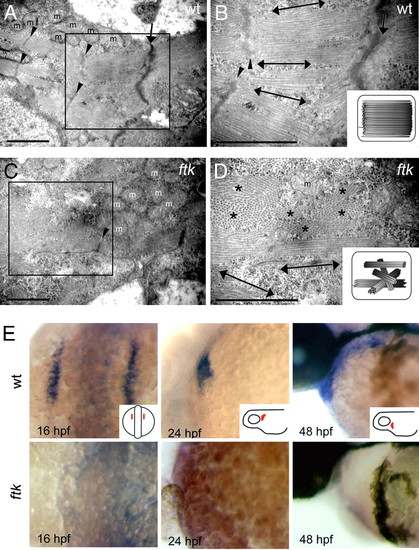
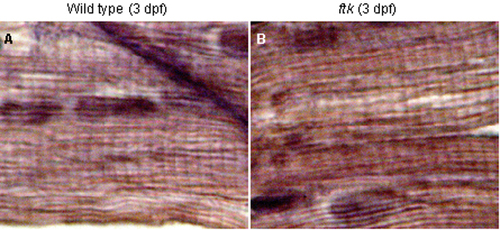
ftk mutation primarily affected the myofibril organization and nkx2.5 expression. (A?D) Myofibril organization examined by TEM analysis. In wild-type embryos at 58 hpf (A and B), myofibrils assemble as thick parallel bundles with orientation toward the longitudinal direction of the cardiomyocyte (double-headed arrows). In contrast, myofibrils in the ftk heart (C and D) are oriented in irregular directions. Double-headed arrows and asterisks indicate the myofibril bundles sectioned parallel and perpendicular to the long axis of the cells, respectively. Arrowheads, Z-lines; arrows, cell?cell contact sites; m, mitochondria. (Scale bars: 500 nm.) (B and D Insets) Schematic drawings of myofibril organization. (E) Whole-mount in situ hybridization analysis of nkx2.5 expression. In wild-type zebrafish embryos (Upper) The expression is detected in the developing heart; however, it is not detectable in ftk mutants (Lower). Mutants at early stages were genotyped by RFLP analysis. (Insets) Orientations of embryos. EXPRESSION / LABELING:
PHENOTYPE:
|
|
Molecular characterization of ftk gene. (A) Chromosome map of the ftk locus on LG 7 between markers z1206 and z14888. An enlarged map (brown line) and overlapping BAC clones (blue lines) are shown below. (B) Missense mutation in the zebrafish cx36.7 gene of the ftk mutant. (C and D) Rescue of the ftk phenotype by cx36.7 mRNA injection. Embryos obtained from crosses of ftk heterozygotes and injected with cx36.7 mRNA (30 pg) did not show the ftk phenotype at 36 hpf (C). (Inset) Sibling uninjected ftk mutant at 28 hpf, which displays the growing pericardiac edema (arrow). Genotyping of mRNA-injected embryos by RFLP analysis confirmed that two of eight embryos were genotypically ftk mutants (res1, res2) (D). (E) Whole-mount in situ hybridization analysis of cx36.7 expression during heart development. (Insets) Orientation of embryos. EXPRESSION / LABELING:
PHENOTYPE:
|
|
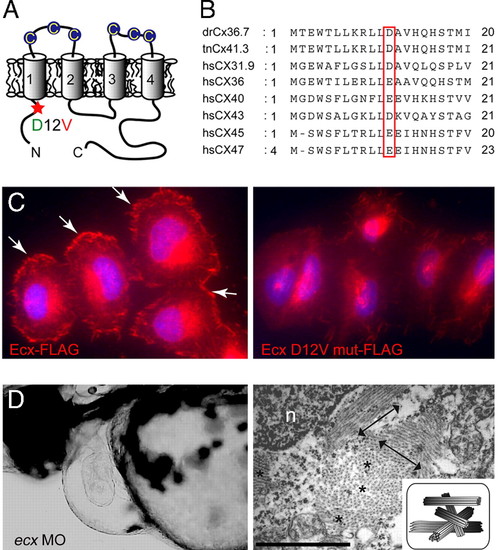
ftk is a loss-of-function mutation of ecx/cx36.7. (A) Putative topology of Ecx protein. Extracellular cysteine residues are indicated by circles. The D12V mutation located in the N-terminal intracellular region of Ecx is indicated with a red asterisk. (B) Alignment of the N-terminal domain of zebrafish Ecx with Tetraodon Cx41.3 and six human connexins. (C) Subcellular localization of FLAG-tagged Ecx proteins in HeLa cells. Wild-type Ecx is localized at the cell surface (Left, arrows), whereas the mutant protein is not recruited to the membrane surface (Right). Nuclei were stained with Hoechst 33342 (blue). (D) Production of ftk phenotype with the ecx MO. (Left) Severe pericardiac edema and dilated heart chambers are observed in larvae injected with ecx MO. Lateral view at 56 hpf is shown. (Right) Disorganized myofibrils as seen in ftk mutants were also caused by the ecx MO. Double-headed arrows and asterisks indicate the myofibril bundles sectioned parallel and perpendicular to the long axis of the cells, respectively. (Scale bar: 500 nm.) n, nucleus. (Inset) Schematic of myofibril organization. PHENOTYPE:
|
|
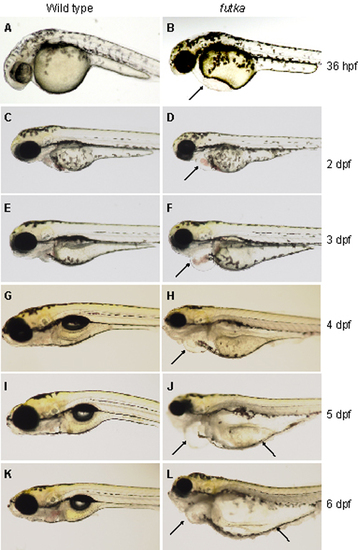
Progressive heart phenotype of ftk mutants. (A-L) Morphology of wild-type (A, C, E, G, I, and K) and ftk mutant (B, D, F, H, J, and L) embryos and larvae. The ftk mutant begins to develop heart edema at around 36 hours postfertilization (hpf) (arrow in B), and by 2 days postfertilization (dpf) it shows irregular heart beating and reverse blood flow (arrow in D). At 3 dpf, the area of pericardiac edema enlarges, and the heart chambers become dilated (arrow in F). ftk mutants become bloodless at ~4 dpf (arrow in H). Pericardiac edema expands to the yolk region (arrows in J and L). |
|
Formation of striated myofibers in the skeletal muscles. (A and B) Hematoxylin/eosin staining of sagittal sections of trunk skeletal muscles. Wild-type (A) and ftk mutant (B) are shown at 3 dpf. A typical striated muscle structure was observed in both samples, indicating that ftk mutation did not affect the myofibril organization in the skeletal muscle. PHENOTYPE:
|
|
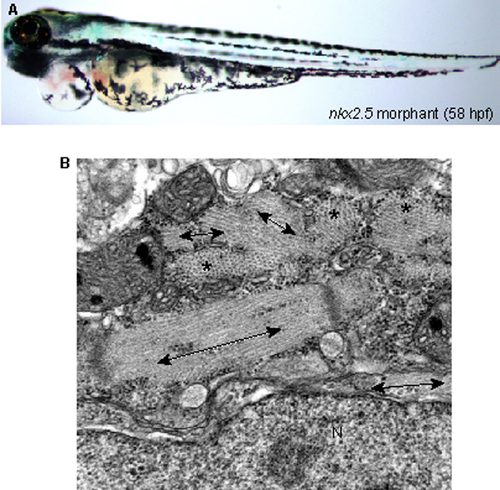
Phenotype of nkx2.5 morphant. (A) Morphological phenotype of nkx2.5 morphant. nkx2.5 knockdown developed a severe heart phenotype, which is indistinguishable from that of ftk mutants, but did not affect other parts of the body. (B) Abnormal myofibril organization in nkx2.5 morphants, which is similar to that of ftk mutants. N, nucleus. The myofibers that run in the plane of section is shown with double-headed arrows, and those run perpendicular to the plane of section are indicated with asterisks. PHENOTYPE:
|
|
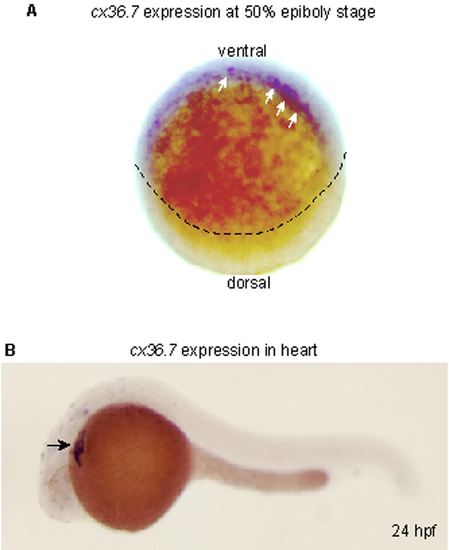
Expression of zebrafish cx36.7. (A) Onset of cx36.7 expression. Whole-mount in situ hybridization analysis of cx36.7 expression at 50% epiboly is shown. Arrows indicate Cx36.7 transcript-positive cells in the ventral half of the marginal zone, which is the region fated to the endoderm. Dotted line indicates the margin of epiboly. (B) Expression of cx36.7 at 24 hpf. Lateral views are shown anterior to the left. cx36.7 expression is observed exclusively in the developing heart (arrow). The expression of cx36.7 was also seen in the ftk mutants (data not shown). EXPRESSION / LABELING:
|
|
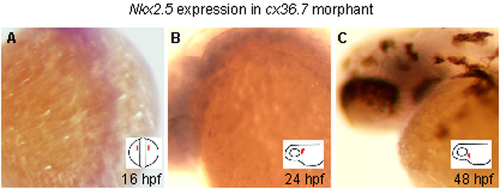
Down-regulation of nkx2.5 expression in ecx morphants. (A-C) Whole-mount in situ hybridization analysis of nkx2.5 expression in the ecx morphants at 16 hpf (A), 24 hpf (B), and 48 hpf (C). No expression of nkx2.5 was detected in all stages examined. (Insets) Orientations of the embryos. Position of the developing heart field is shown in red. EXPRESSION / LABELING:
|

Unillustrated author statements |