- Title
-
A direct role for Sox10 in specification of neural crest-derived sensory neurons
- Authors
- Carney, T.J., Dutton, K.A., Greenhill, E., Delfino-Machin, M., Dufourcq, P., Blader, P., and Kelsh, R.N.
- Source
- Full text @ Development
|
Generation and characterisation of sox10:egfp transgenic fish. (A) Restriction maps of the sox10 genomic region. Exons are boxed, sox10 untranslated sequences are indicated in light blue and coding region in dark blue. Lower panel shows a schematic of the construct used to generate transgenic fish. Yellow box shows the polyadenylation signal. Sizes in kb, N=NotI, S=SalI, RV=EcoRV, B=BamHI, Sp=SpeI, E=EcoRI, Nc=NcoI. (B-H) GFP in germline sox10:eGFP transgenic fish is seen in premigratory and migratory NCCs, in time and manner recapitulating the expression of sox10. Premigratory cranial NC at 11 hpf (B; fluorescence (left panel), DIC (middle) and combined (right)) and 17 hpf (C). (D) Cranial NCCs at 15 hpf to show resolution of fine cell morphology. (E) Premigratory (arrowhead) and migrating NCCs in head and on medial pathway in anterior trunk (arrows) at 24 hpf. (F) At 24 hpf, cranial GFP-expressing cells include migrating NC (mc), NC in branchial arches (a), glial precursors surrounding the cranial ganglia (g), and otic epithelium (o). (G) Double immunofluorescence imaging of GFP (green) and neurons (Hu, red) at 48 hpf. There is continued expression in otic epithelium and arches. (H) Tail of 40 hpf embryo to show nascent DRG and migrating NCCs. (I-K) GFP expression persists in peripheral glia, including DRG satellite glia (J), Schwann cells [posterior lateral line (I) and spinal nerves (J,K)], here at 5 dpf. (J) Double immunofluorescence demonstrates complementary expression of GFP (green) and Hu (red) in DRGs. (K) GFP+ Schwann cells are intimately associated with spinal nerve axons (DM-GRASP, red). All are confocal images except (B) and are of live embryos except (G), (J) and (K). In this and all subsequent figures, embryos are shown in lateral view unless stated otherwise. Scale bars: 100 μm in B,C,E; 20 μm in D,F,I,J,K; 50 μm in G,H. |
|
Glial differentiation fails in sox10 embryos. (A,B) Double immunofluorescent detection of trunk sensory neurons (Hu, red) and prospective glial cells (GFP, green) in anterior trunk in 5 dpf wild-type (A) and sox10 mutant (B) embryos carrying the sox10:egfp transgene. Although GFP+ cells occupy similar positions in sox10 mutant and wild-type embryos (arrows), the elongated morphology of Schwann cells and the compact clustered arrangement of satellite glial cells in DRGs (arrowheads) of wild-type embryos (A) are absent in sox10 mutants (B). Neurons are often displaced ventrally in sox10 mutant embryos, but sensory neuron number may be approximately normal at this axial position (B). (C,D) In situ hybridisation with mbp reveals absence of differentiated Schwann cells in sox10 embryos (D) compared with wild-type siblings (arrowhead, C). Likewise, oligodendrocyte differentiation (*, C) fails in sox10 mutants (D). Scale bars: 20 μm in A; 50 μm in B. |
|
sox10 mutants display a DRG neuron phenotype. Immunofluorescent detection of Hu at 5 dpf (A,B), DM-GRASP (zn-5 antibody) at 8 dpf (D,E) and Isl-1 at 60 hpf (F,G) in wild-type (A,D,F) and sox10 (B,E,G) embryos demonstrates the reduction in DRG sensory neurons (arrowheads) in the mutant. Quantitation (mean+s.d.) of Hu (C) and Isl-1 (H) phenotypes shows increasing severity posteriorly. Significant reduction in sensory neurons is noted at all stages (** P<0.01, ***P<0.0001; Student's t-test). n=12 embryos for each of wild-type and sox10 mutants in (C) and n=15 (wild type) and n=13 (sox10) in (H). There is an absence of enteric neurons in sox10 mutants (arrow, B). However, DM-GRASP expression in secondary motorneuron processes (*,D) is unchanged in sox10 mutants (E). Likewise, Islet1 also labels ventral spinal cord motorneurons (*,F,G). Quantification (I) of these cells at 5 dpf per somite segment equivalent shows no significant difference between wild-type and sox10 embryos (P=0.609; Student's t-test). Three consecutive segment-lengths in the tail were counted in seven embryos of each genotype. Scale bars: 200 μm in A,B; 50 μm in D-G. |
|
DRG sensory neurons persist in sox10 embryos without differentiated glial support. Double immunofluorescent detection of DRG neurons (Hu, red) and glia (GFP, green) in 5 dpf embryos showing two examples of sox10 mutant DRG neuron clusters (arrowheads) lacking intimate association with GFP+ cells (A,B), contrasting with consistently intimate arrangement with satellite cells in wild types (C). Scale bar: 20 μm. |
|
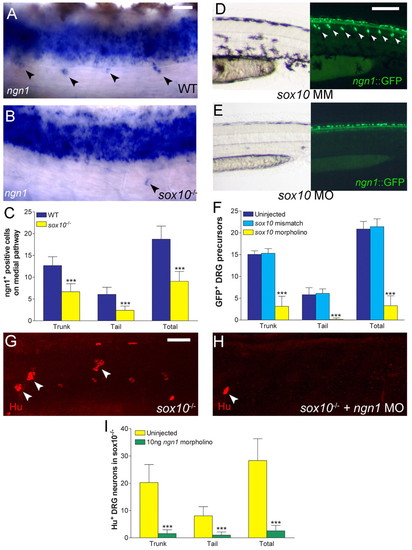
Specification of a subset of DRG neurons fails in sox10 mutants. (A-C) Whole mount in situ hybridisation of 36 hpf wild-type (A) and sox10 mutant (B) embryos for ngn1. Quantitation (mean+s.d., C) showed highly significant reduction in mutants (*** P<0.0001; Student's t-test; n=15 for both data sets). (D-F) Injection of sox10 morpholino (MO; E), but not 3 bp-mismatch morpholino (MM; D), into ngn1:gfp transgenic fish results in reduction of both melanophores (left panels) and DRG sensory neuron precursors (arrowheads, right panels) at 48 hpf. Mismatch morpholino-injected embryos are indistinguishable from uninjected embryos. Quantification (mean+s.d., F) showed highly significant effect in sox10 morphants (*** P<0.0001; ANOVA with Tukey's Post Test; n=22 for sox10 MO, n=20 for both sox10 MM and uninjected data sets). Numbers of GFP+ cells (F) are slightly greater than numbers of ngn1+ cells (C) consistent with the later stage examined and perdurance of GFP in cells in which the promoter is no longer active. (G-I) Ngn1 knock down using a previously characterised morpholino in sox10 mutant embryos results in near-complete loss of escaper DRG sensory neurons, even in the trunk (H), compared with uninjected sox10 mutants (G). This reduction was highly significant (I) (*** P<0.0001; Student's t-test) (n=16 for each data set). Scale bars: 20 μm in A,B; 50 μm in G,H; 100 μm in D,E. |
|
Functional redundancy between sox9b and sox10 in DRG neuron development. sox9b morpholino knock-down results in decreased Hu+ sensory neuron number in 5 dpf wild-type (A,B) and sox10 mutant (C,D) embryos. Sox9b morpholino injection into wild type (B) disrupts the regular DRG pattern of uninjected wild-type siblings (A), resulting in reduced numbers of DRGs (arrows), fewer DRG neurons and disorganised patterning of DRGs. These features are also typical of sox10 mutant embryos (C), although the latter show a more severe phenotype. Injection of sox9b morpholino into sox10 mutants results in a dramatic reduction in DRG sensory neurons (D). (A-D) Lateral views of trunk. (E) Quantitation (mean+s.d.) of total number of trunk Hu+ sensory neurons in 5 dpf wild-type and sox10 mutant embryos after injection with sox9b morpholinos as shown (n=10 for all conditions). Asterisk indicates statistically significant differences from respective controls (P<0.05; Student's t-test). Scale bar: 50 μm. PHENOTYPE:
|
|
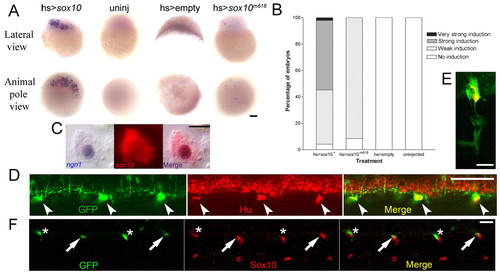
Cell-autonomous induction of ngn1 by Sox10. (A) In situ hybridisation reveals robust ngn1 induction by 4 hours after injection of heatshock construct driving wild-type sox10 (hs>sox10) into one-cell stage wild-type embryos followed by 2 hour heat shock at 37°C from 2 hpf (A). Heat-shocked uninjected embryos and embryos injected with an empty heat-shock construct, never contained ngn1+ cells at this stage. Injection of a heat-shock vector driving expression of the Sox10m618 mutant protein (hs>sox10m618(L142Q)) gave only very weak induction of ngn1. (B) Relative proportions of embryos showing very strong, strong, weak or no induction of ngn1 after each treatment. (C) Double in situ hybridisation of injected embryos with sox10 (red/fluorescence) and ngn1 (blue) showed that ngn1+ cells had detectable sox10 message (99/103 cells scored in ten embryos). Nuclear localisation of ngn1 message suggests that nuclear export of these transcripts was limited within the timeframe of this experiment. (D,E) Double immunofluorescence confocal images (single focal plane) of 48 hpf wild-type sox10:eGFP embryos showed perduring GFP protein (green, left panel) in Hu+ (red, middle panel) nascent DRG neurons (merged, right panel). At this stage, 54/71 (76%) DRG neurons in five wild-type embryos were detectably GFP+; colocalisation has been confirmed in an independent sox10:eGFP line (data not shown). Hu-/GFP+ glial precursors (green) surrounding the Hu+/GFP+ neurons (yellow) were revealed using a larger confocal pinhole size (E). (F) Double immunofluorescence confocal images (single focal plane) of 40 hpf wild-type ngn1:egfp embryo showing overlap of weak (arrowed) GFP (green, left panel) with Sox10+ (red, middle) nascent DRG neurons (merged, right panel). Cells expressing higher levels of GFP, likely to be more mature neurons, lack Sox10 expression (asterisks). Scale bars: 100 μm in A; 10 μm in C; 50 μm in D; 20 μm in E,F. EXPRESSION / LABELING:
|
|
sox10baz1 mutants show a unique, neurogenic DRG phenotype. (A-H) 5 dpf sox10baz1/baz1 showed severe reductions in NC-derived melanophores (B) and mbp+ Schwann cells of spinal nerves (D) compared with wild-type siblings (A,C). Note similar strength of these phenotypes to sox10m618/m618 (see Fig. 2C-D). In contrast, Hu+ DRG sensory neurons were more abundant in sox10baz1/baz1 embryos (E, low power; F, higher confocal magnification view of three segments of tail) than in wild type (H), and thus much more than in typical strong sox10 alleles (G). (I,J) sox10baz1/baz1 embryos similarly displayed an increased number of ngn1+ cells on the medial migration pathway at 36 hpf (J) compared with wild-type (I). (K,L) Quantitation of these Hu+ (K) and ngn1+ (L) DRG-associated cells in sox10baz1/baz1 and siblings (***P<0.0001, Student's t-test; n=10 and n=15 embryos counted for each data set, respectively). (M) Sequencing identified molecular lesion in sox10baz1 as a G to A transition at cDNA position 724 (upper panel), producing a Valine to Methionine substitution at amino acid 117 within the Sox10 protein (lower panel). DNA binding domain (red) and transactivation domain (blue) are indicated. Scale bars: 200 μm in A,B,E; 50 μm in C,D,F-J. |
|
Cell autonomy of sox10 action in DRG sensory neurons. Fluorescein dextran-labelled wild-type donor cells were transplanted into unlabelled sox10 mutant host embryo, and differentiated sensory neurons identified by anti-Hu immunofluorescence (red) at 3 dpf. Embryo is shown as a projected confocal stack. Transplanted cells (green) contributed to three adjacent DRGs, but whereas Hu+ neurons are found in two of them, the third (*) lacks sensory neurons despite having a large contribution of wild-type cells to glial component. Note that all other fluorescein+ cells lie within the CNS. Among our transplants, two separate individuals showed a result equivalent to that documented here. Thus, wild-type glia are insufficient for survival of DRG sensory neurons. |
|
Proliferating neurons in wild-type and sox10 mutant DRGs. (A) Double immunofluorescence confocal image (single focal plane) of 72 hpf wild-type embryo showing two adjacent DRGs in the anterior tail. Three Hu+ neurons (green) are visible, two expressing phospho-histone H3 (pH3, red). (B) Double immunofluorescence confocal image (single focal plane) of 72 hpf sox10 mutant embryo showing DRGs in the anterior tail. One Hu+ neuron is expressing pH3. An additional DRG is also visible out of focus to the right. Scale bar: 50 μm. |