- Title
-
The Glutamine Synthetases Are Required for Sensory Hair Cell Formation and Auditory Function in Zebrafish
- Authors
- Zhao, Y., Wang, Z., Xu, M., Qian, F., Wei, G., Liu, D.
- Source
- Full text @ Int. J. Mol. Sci.

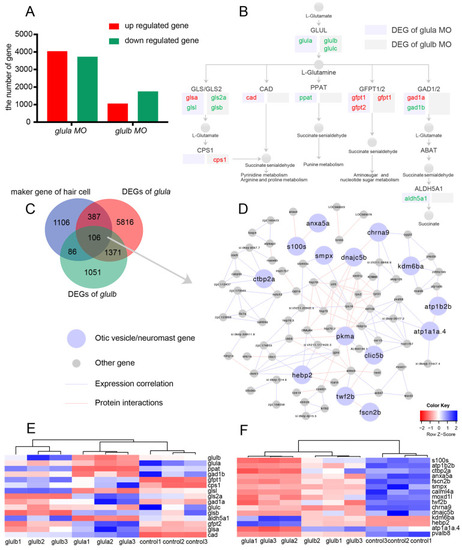
ZFIN is incorporating published figure images and captions as part of an ongoing project. Figures from some publications have not yet been curated, or are not available for display because of copyright restrictions. PHENOTYPE:
|

ZFIN is incorporating published figure images and captions as part of an ongoing project. Figures from some publications have not yet been curated, or are not available for display because of copyright restrictions. EXPRESSION / LABELING:
PHENOTYPE:
|
|
Phylogenetic analysis of |
|
The downregulation of EXPRESSION / LABELING:
PHENOTYPE:
|
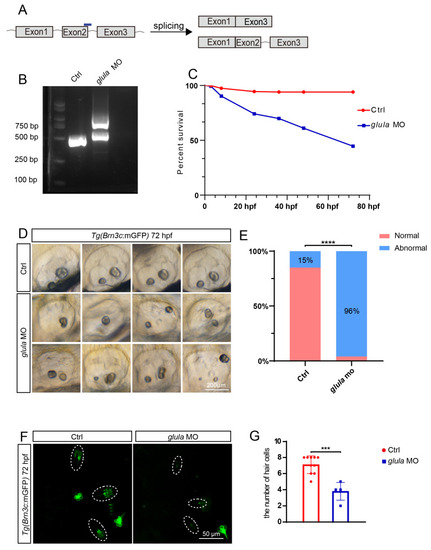
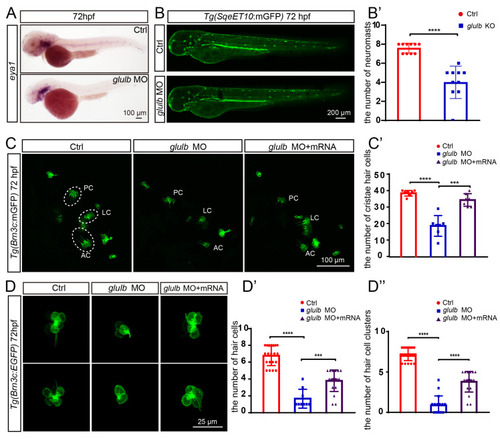
|
The knockdown of |
|
The zebrafish that were injected with EXPRESSION / LABELING:
PHENOTYPE:
|
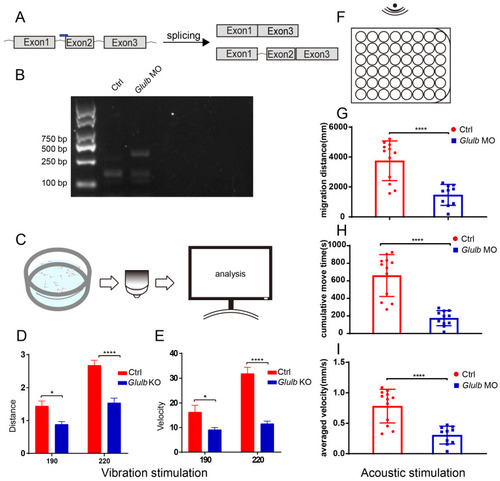
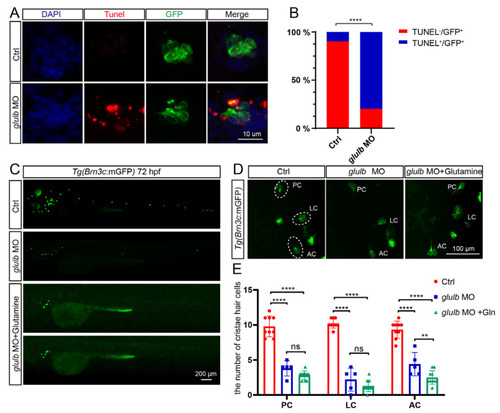
|
Knockdown of EXPRESSION / LABELING:
PHENOTYPE:
|
|
Transcriptomic analysis of zebrafish with |