- Title
-
Presynaptic Nrxn3 is essential for ribbon-synapse maturation in hair cells
- Authors
- Jukic, A., Lei, Z., Cebul, E.R., Pinter, K., Tadesse, Y., Jarysta, A., David, S., Mosqueda, N., Tarchini, B., Kindt, K.
- Source
- Full text @ Development
|
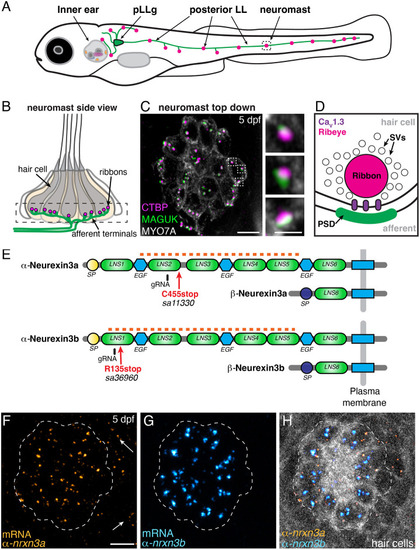
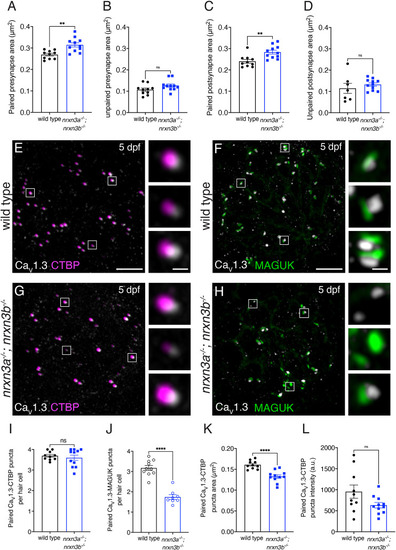
EXPRESSION / LABELING:
|
|
|
|
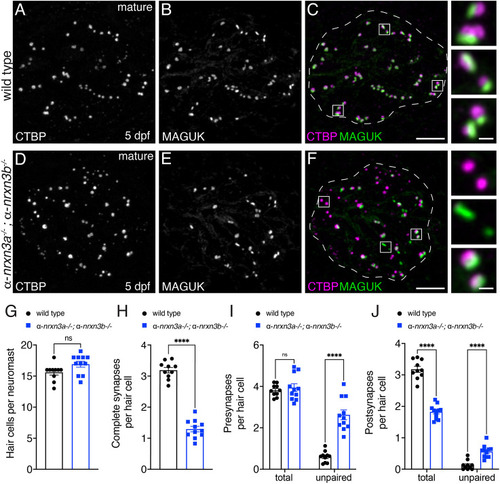
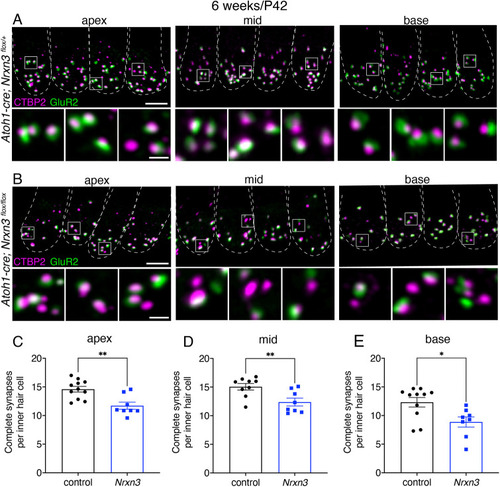
PHENOTYPE:
|
|
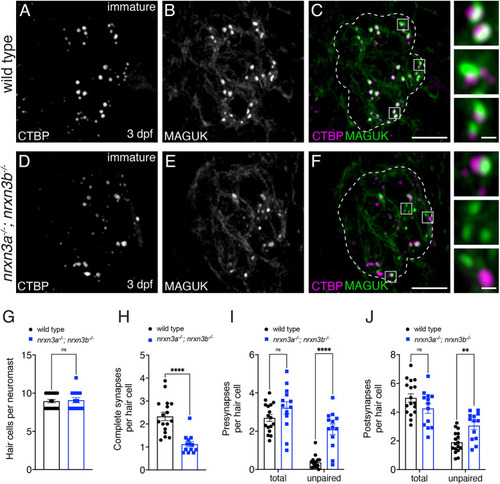
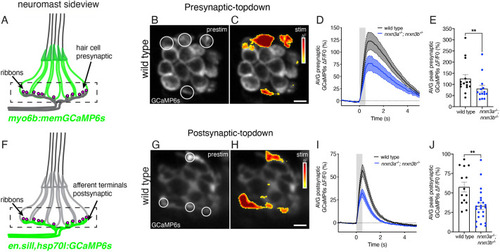
PHENOTYPE:
|
|
|
|
|
|
|
|
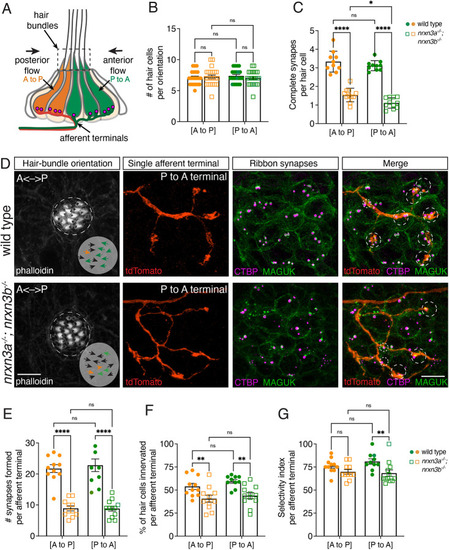
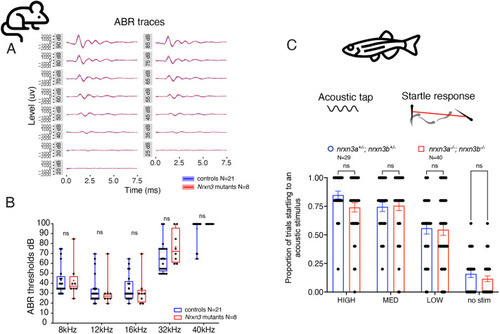
PHENOTYPE:
|