- Title
-
Myogenin is an essential regulator of adult myofibre growth and muscle stem cell homeostasis
- Authors
- Ganassi, M., Badodi, S., Wanders, K., Zammit, P.S., Hughes, S.M.
- Source
- Full text @ Elife
|
( |
|
( |
|
( |
|
( |
|
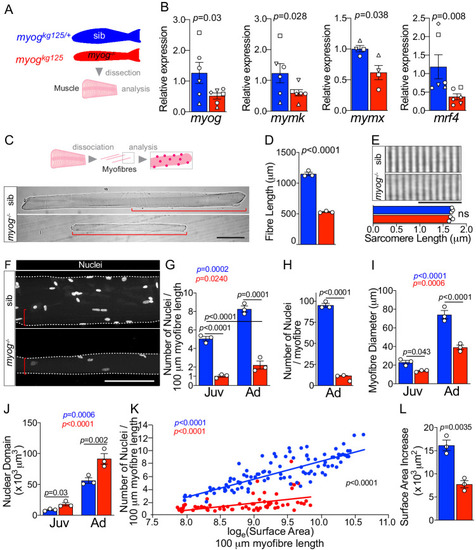
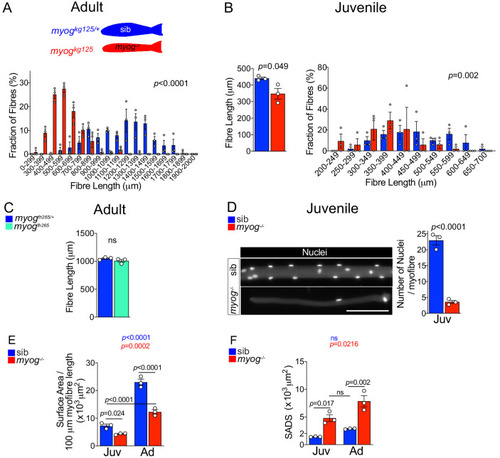
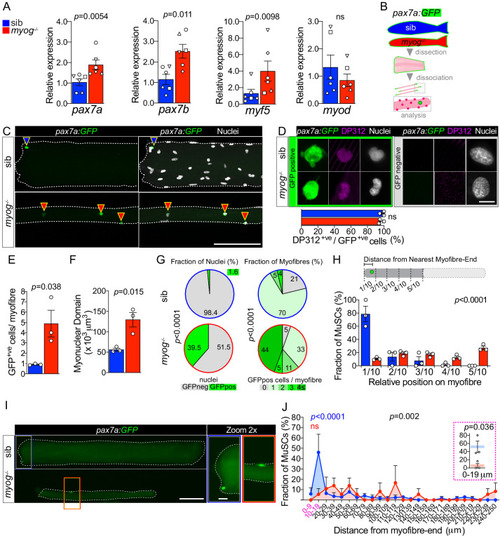
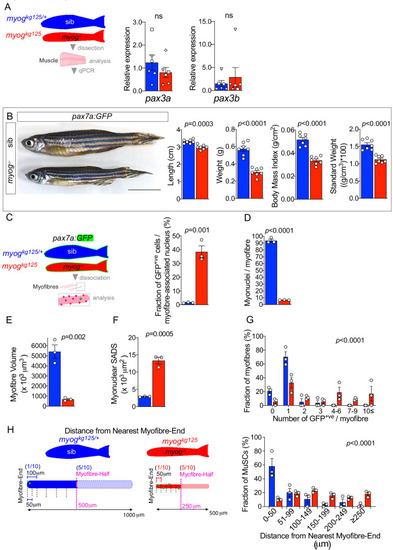
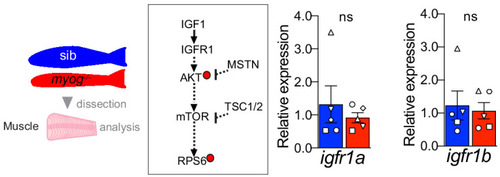
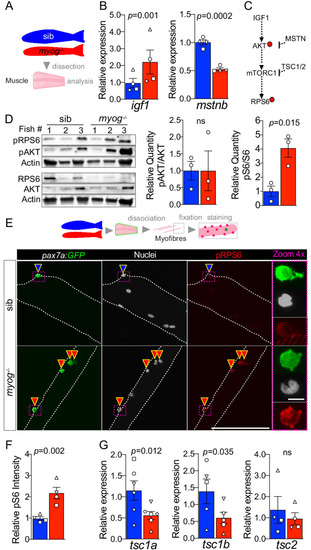
Schematic of adult trunk muscle processing for qPCR analysis (left). Summary of mTORC1 pathway with analysed members, positive (arrows) or negative (bars) effects are indicated (centre). Dashed lines indicate other molecules involved, plain arrow indicate direct effect, red dots represent phosphorylation. |
|
Schematic of adult trunk muscle processing for qPCR analysis (left). Summary of mTORC1 pathway with analysed members, positive (arrows) or negative (bars) effects are indicated (centre). Dashed lines indicate other molecules involved, plain arrow indicate direct effect, red dots represent phosphorylation. |
|
( |
|
( |