- Title
-
E2f5 is a versatile transcriptional activator required for spermatogenesis and multiciliated cell differentiation in zebrafish
- Authors
- Xie, H., Kang, Y., Wang, S., Zheng, P., Chen, Z., Roy, S., Zhao, C.
- Source
- Full text @ PLoS Genet.
|
(A-B) Whole-mount EXPRESSION / LABELING:
PHENOTYPE:
|
|
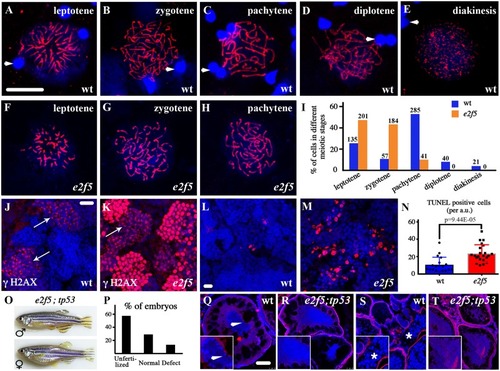
(A-H) Confocal images of primary spermatocytes at different prophase stages of meiosis I as indicated by anti-Sycp3 antibody staining (red). Arrowheads indicate spermatozoa nuclei from wild-type testis stained with DAPI in blue (A-E). (I) Bar graph showing the statistical results of the percentage of cells in different meiotic stages. The numbers of spermatocytes investigated are shown on top of each bar. (J-K) Staining of γH2AX (red) in the testes of wild-type and PHENOTYPE:
|
|
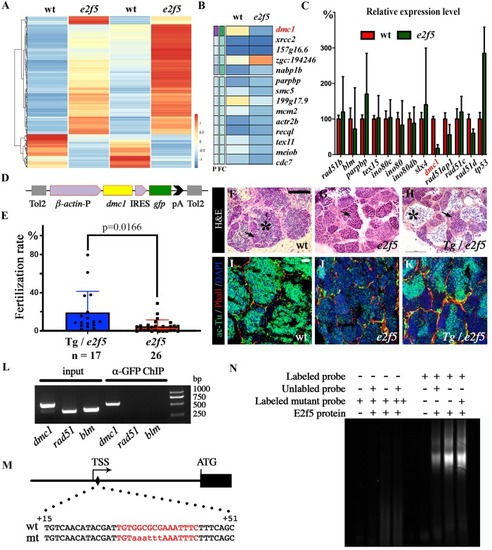
(A) Heat map of RNA-seq transcriptome analysis from two sets of wild-type and mutant testes. (B) Expression heat map of genes involved in homologous recombination in wild-type and |
|
(A-H) Confocal images showing cilia in the PST region of the pronephros of wild-type and mutant larvae at different stages as indicated. Cilia were visualized with anti-glycylated tubulin antibody in green. Nuclei were labeled with DAPI in blue. Red arrowheads indicate multicilia bundles and yellow arrowheads indicate single cilia. (I-J) Still images from PHENOTYPE:
|
|
(A-E) Double fluorescence EXPRESSION / LABELING:
|
|
(A-P) Whole-mount |

ZFIN is incorporating published figure images and captions as part of an ongoing project. Figures from some publications have not yet been curated, or are not available for display because of copyright restrictions. PHENOTYPE:
|

ZFIN is incorporating published figure images and captions as part of an ongoing project. Figures from some publications have not yet been curated, or are not available for display because of copyright restrictions. |