- Title
-
Tmem88a mediates GATA-dependent specification of cardiomyocyte progenitors by restricting WNT signaling
- Authors
- Novikov, N., and Evans, T.
- Source
- Full text @ Development
|
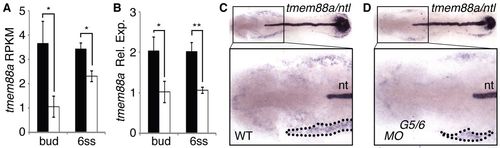
Confirmation of gata5/6-dependent tmem88a expression in the anterior lateral plate mesoderm. (A,B) RNA sequencing tmem88a expression in RPKM (A) and qPCR tmem88a relative expression (B) for wild-type (black) and gata5/6 morphants (white) at bud and 6-somite (6ss) stages of development. (C,D) Transcript localization of tmem88a and the regional notochord (nt) marker ntl, shown in flatmount in situ hybridization preparations of 6-somite stage control (C) and gata5/6 (D) morphants (dorsal views, anterior to the left). Insets are higher magnifications of the boxed regions. tmem88a expression in the anterior lateral plate mesoderm is outlined (dotted line). n>20 embryos analyzed per panel. *P<0.05, **P<0.01; error bars indicate s.d. EXPRESSION / LABELING:
PHENOTYPE:
|
|
tmem88a transcript localizes to cardiac progenitors. (A,B) Dorsal views (anterior to the left) of flatmount in situ hybridization of wild-type 8-somite stage zebrafish embryos stained for nkx2.5/ntl and tmem88a/ntl. Insets are higher magnifications of the boxed regions. (C,D) Laterals views of whole-mount in situ hybridization (WISH) preparations of wild-type 14-somite stage embryos stained for nkx2.5/ntl and tmem88a/ntl. Insets are higher magnification dorsal views of the boxed regions. (E,F) Dorsal views of cardiogenic mesoderm (anterior to the left) in flatmount dual fluorescent in situ hybridization preparations of wild-type embryos stained for nkx2.5/ntl or tmem88a at the 8- or 14-somite stage. Arrowheads indicate CPs. n>20 embryos analyzed per panel. EXPRESSION / LABELING:
|
|
Loss of Tmem88a results in profound cardiomyopathy. (A) Brightfield images of representative control (MOCtrl) and tmem88a (MOT88a) morphants at 48 hpf. Arrowhead indicates pericardial edema. (B,C) Higher magnification of craniocardiac tissues in control and tmem88a morphants. Brain (br), eye (e), yolk (y), heart (hrt, arrowheads) and blood (bl, arrow) are indicated. (D,E) Fluorescent images of control and tmem88a morphant hearts observed in the tg(myl7:eGFP) background. Ventricle (v) and atrium (a) are indicated. n>50 embryos analyzed per panel. Scale bars: 0.1 mm in B-E. EXPRESSION / LABELING:
PHENOTYPE:
|
|
Tmem88a-deficient hearts are small and hypocellular. (A,B) Representative confocal images of control and tmem88a morphant hearts dissected from 51 hpf zebrafish embryos. Morphants were generated in the tg(myl7:DsRed2-nuc) background. Left panels show immunohistochemistry for DsRed2 labeling of cardiomyocyte nuclei (CMs). Middle panels show S46 antibody labeling of atrial cells. Right panels show merge. Atrium (a) and ventricle (v) are indicated. (C) Atrial (a), ventricular (v) and total (T) cardiomyocyte numbers. (D) Atrial, ventricular and total cardiac chamber length. n=6 hearts quantified per condition. *P<0.05, **P<0.01; error bars indicate s.d. |
|
Tmem88a depletion reduces the expression of cardiomyocyte differentiation markers. Zebrafish embryos were analyzed at the 23-somite stage. (A-D) WISH for ventricular cardiomyocyte marker (vmhc) and atrial cardiomyocyte marker (amhc) in control (A,B) and tmem88a (C,D) morphants. n>20 embryos analyzed per condition. (E,F) Relative vmhc+ and amhc+ areas in control (Ctrl) and tmem88a (T88a) morphants. (G,H) Relative expression levels of vmhc and amhc assessed by qPCR. Measurements are normalized to control; n=10 embryos measured per condition. **P<0.01; error bars indicate s.d. EXPRESSION / LABELING:
PHENOTYPE:
|
|
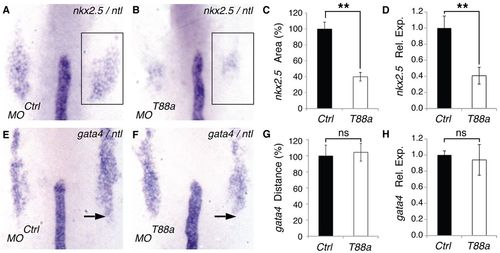
Loss of Tmem88a reduces the CP fields. Zebrafish embryos were analyzed at the 8-somite stage. (A,B) WISH for nkx2.5 (box) and ntl in representative control and tmem88a morphants. (C) Relative nkx2.5+ areas in control and tmem88a morphants. (D) nkx2.5 relative expression levels as assessed by qPCR. (E,F) WISH for gata4 and ntl in representative control and tmem88a morphants. Arrow indicates the posterior gata4+ domain boundary. (G) Relative distance between the notochord tip and the posterior gata4+ domain boundary. (H) gata4 relative expression levels by qPCR. (A,B,E,F) Dorsal views of flatmount embryos, with anterior to the top; n>20 embryos analyzed per condition. (C,G) Measurements are normalized to control; n=10 embryos measured per condition. **P<0.01; ns, not significant; error bars indicate s.d. EXPRESSION / LABELING:
PHENOTYPE:
|
|
Loss of CPs in tmem88a morphants is not caused by increased apoptosis, decreased cell proliferation or fate conversion to the hematovascular or forelimb lineages. Dorsal views of representative flatmount preparations of 8-somite (A-H) and 23-somite (I,J) control and tmem88a morphants, with anterior to the top (n>20 embryos analyzed per condition). (A,B) WISH for nkx2.5 and ntl, showing the cardiogenic mesoderm (arrowheads). (C,D) Apoptosis detection by TMR-Red. (E,F) Phospho-Histone H3 antibody staining for cell proliferation. (G,H) WISH for the hematovascular marker scl and for ntl. Arrow indicates the boundary of the anterior scl expression domain. (I,J) WISH for the forelimb marker tbx5. EXPRESSION / LABELING:
PHENOTYPE:
|
|
WNT target gene expression negatively correlates with tmem88a levels. Relative expression levels assessed by qPCR of (A,B) the WNT sensor TOPdGFP and (C,D) the WNT target gene dkk1 in wild-type zebrafish embryos (WT), in embryos injected with control (Ctrl) or tmem88a (T88a) MO, and in embryos injected with mutated tmem88a mRNA (Δt88a) or wild-type tmem88a mRNA (t88a). *P<0.05, **P<0.01; ns, not significant; error bars indicate s.d. EXPRESSION / LABELING:
PHENOTYPE:
|
|
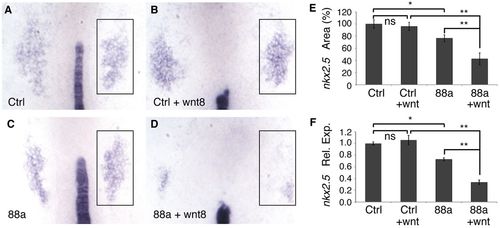
CP formation is repressed by forced expression of wild-type but not mutant tmem88a, and this phenotype is rescued by enhancing early WNT signaling. (A-E) WISH for nkx2.5 (box) and ntl in representative 8-somite stage uninjected control zebrafish embryos (A, Ctrl), embryos injected with wild-type tmem88a mRNA (B, 88a RNA), embryos injected with mutated tmem88a mRNA (C, Δ88a), heat-shocked uninjected control embryos (D, Ctrl+wnt8), and heat-shocked embryos that had been injected with wild-type tmem88a mRNA (E, 88a RNA+wnt8). Heat-shock treatment was administered to embryos at 3 hpf for 30 minutes. n>15 embryos analyzed per condition. (F) nkx2.5 relative expression levels assessed by qPCR. **P<0.01; error bars indicate s.d. |
|
Limited WNT signaling sensitizes cardiogenic mesoderm to subthreshold depletion of Tmem88a. (A-D) WISH for nkx2.5 (box) and ntl in representative 8-somite stage control morphants (Ctrl) and subthreshold tmem88a morphants (88a), without or with (+wnt8) low-level WNT activation. Retraction of the notochord and general widening of the mesodermal plate occur in heat-shocked embryos, but independently of MO injection. Heat-shock treatment was administered to 75% epiboly embryos for 30 minutes. n>20 embryos analyzed per panel. (E) nkx2.5+ area normalized to control values. n=10 embryos measured per condition. (F) nkx2.5 relative expression levels assessed by qPCR. *P<0.05, **P<0.01; ns, not significant; error bars indicate s.d. EXPRESSION / LABELING:
PHENOTYPE:
|
|
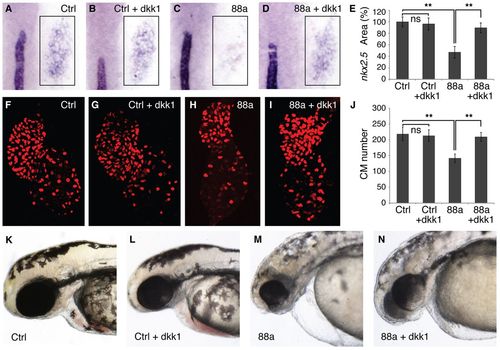
Late suppression of WNT signaling rescues CPs and cardiomyocyte numbers in tmem88a morphants. (A-D) WISH for nkx2.5 (box) and ntl in representative 8-somite stage control (Ctrl) and tmem88a (88a) morphants, without or with (+dkk1) WNT inhibition. Retraction of the notochord occurs in heat-shocked embryos, but independently of MO injection. Heat-shock treatment was administered to 75% epiboly embryos for 1 hour. n>20 embryos analyzed per panel. (E) nkx2.5+ area normalized to control values. n=6 embryos measured per condition. (F-I) Cardiomyocyte nuclei in representative 48-hpf embryos from the tg(myl7:DsRed2-nuc) line. n>25 embryos analyzed per panel. (J) Quantification of total cardiomyocyte (CM) numbers. n=6 control hearts analyzed; n=9 hearts analyzed for all other conditions. (K-N) Brightfield images showing craniocardiac tissues in 48-hpf embryos. n>50 embryos analyzed per condition. **P<0.01; ns, not significant; error bars indicate s.d. EXPRESSION / LABELING:
PHENOTYPE:
|
|
Analysis of tmem88a expression in embryos lacking hematovascular progenitors. WISH for tmem88a and the notochord marker ntl in sibling (A) wild-type (WT) (n=17) and (B) cloche (n=6) embryos at the 10-somite stage. Shown are dorsal views of flatmounted embryos, with anterior to the left. Homozygous cloche mutants lack tmem88a expression in the hematovascular region of the posterior lateral plate mesoderm (arrowheads). By contrast, as confirmed in higher magnification views of boxed areas, tmem88a transcript patterns are normal in the ALPM of cloche mutants compared with sibling wild-type embryos (insets, dotted lines). EXPRESSION / LABELING:
PHENOTYPE:
|
|
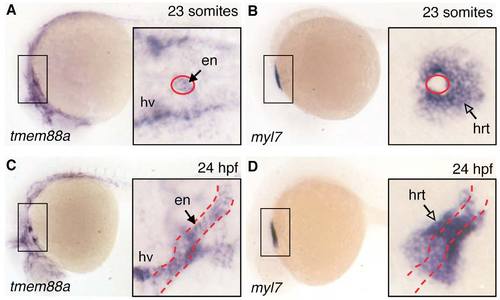
At later stages, tmem88a transcripts localize to endothelium. WISH in representative wild-type embryos for tmem88a and myl7 transcript localization. (A,B) Representative 23-somite stage and (C,D) 24-hpf embryos are shown with anterior to the left. Lateral views in each panel are supplemented with higher magnification dorsal views of the boxed regions (insets). Endothelial tissues, including endocardium (en, red dashed outline) and head vessels (hv) express the tmem88a transcript, whereas myl7 is restricted to the heart muscle (hrt, open arrow). All insets are shown at the same magnification. n>20 embryos analyzed per panel. EXPRESSION / LABELING:
|
|
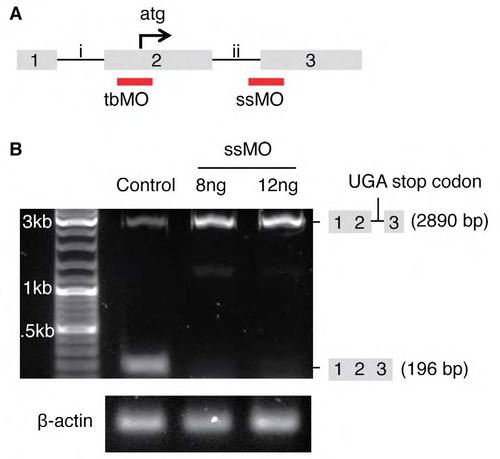
Design and validation of tmem88a morpholinos. (A) Morpholino design. A translation blocker (tbMO) was targeted to the translation start site (atg) of tmem88a mRNA. A splice-site blocker (ssMO) was targeted to the boundary between intron 2 (ii) and exon 3 (3) of the mRNA. (B) The efficacy of the ssMO was monitored by semi-quantitative reverse-transcription PCR. The abundance of the normal tmem88a transcript (196 bp) was reduced with optimal doses of ssMO. An unspliced transcript (2890 bp) accumulated that is predicted to encode a premature stop codon (UGA), confirmed by sequencing. Levels of β-actin were used as a loading control. |
|
Control and tmem88a morphants are morphologically similar at 28 hpf. Representative control (MOCtrl) and tmem88a (MOT88a) morphants shown at 28 hpf; lateral views, anterior to the left. n>50 embryos analyzed per panel. Scale bar: 0.5 mm. |
|
The cardiomyopathy caused by depletion of Tmem88a is p53 independent. Brightfield images of representative 48-hpf p53?/? embryos injected with (A) control (Ctrl MO) or (B) tmem88a (T88a MO) morpholino. Images on the right (insets) are higher magnifications of cardiac and cranial tissues in each embryo. All views are lateral, anterior to the left. In the main panels, blood flow is obvious in the p53 mutant (arrow in A), but absent in the tmem88a morphant, with pericardial edema (arrowhead in B). Brain (br), heart (hrt) and blood (bl) are indicated and a translucent heart-string is evident in the tmem88a morphant. n>50 embryos analyzed per panel. Scale bar: 0.1 mm in insets. PHENOTYPE:
|
|
Tmem88a depletion selectively affects lateral mesoderm derivatives including erythromyeloid progenitors. Flatmount preparations of representative 8- to 10-somite stage control (Ctrl) and tmem88a (88a) morphant embryos processed by WISH for (A,B) ntl+ axial mesoderm, (C,D) myod+ paraxial mesoderm, (E,F) pax2a+ intermediate mesoderm and (G,H) scl+ hematovascular domains in the context of ntl. (I,J) gata1+ hematopoietic progenitors. n>20 embryos per condition. (K) Relative expression levels by qPCR for various hematopoietic and vascular markers. **P<0.01. EXPRESSION / LABELING:
PHENOTYPE:
|
|
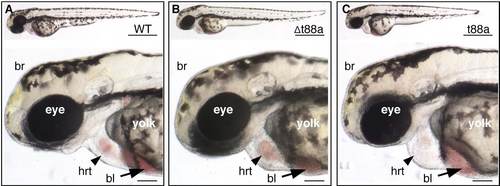
Tmem88a overexpression causes cardiomyopathy. Brightfield images of 48-hpf (A) wild-type (WT) embryos, (B) embryos injected with mutated tmem88a mRNA (Δt88a) and (C) embryos injected with wild-type tmem88a mRNA (t88a). Brain (br), heart (hrt) and blood (bl) are indicated. n>50 embryos analyzed per panel. Scale bars: 0.5 mm (top) and 0.1 mm (bottom). |