- Title
-
Ret isoform function and marker gene expression in the enteric nervous system is conserved across diverse vertebrate species
- Authors
- Heanue, T.A., and Pachnis, V.
- Source
- Full text @ Mech. Dev.
|
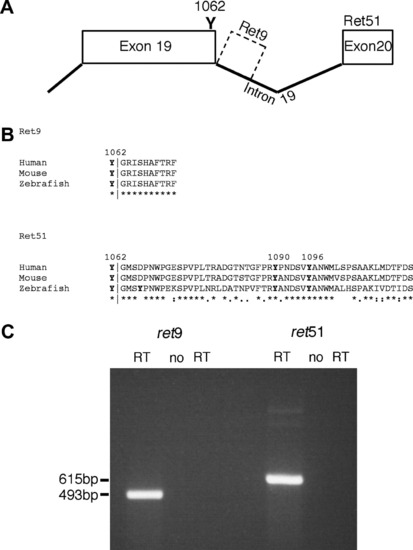
Zebrafish ret exists as ret9 and ret51 isoforms. (A) Analysis of genomic sequences reveals that in zebrafish, alternative splicing at exon 19 can lead to generation of two isoforms of ret. Splicing to exon 20 generates ret51, whereas an absence of splicing generates ret9 from intronic sequences. (B) Sequence alignment using ClustalW 1.83 shows that zebrafish Ret9 amino acid sequences are identical to mouse and human RET9, and that zebrafish Ret51 sequences show significant sequence homology to mouse and human RET51, with 67% amino acid identity to each, including tyrosines (Y) at positions 1090, and 1096. Y1062, which is present in both Ret9 and Ret51, is also conserved. In alignment, ?*? indicates identical amino acids, ?:? indicates conserved substitutions, and ?.? indicates semi-conserved substitutions (C) RT-PCR analysis of 24hpf zebrafish embryos using primers specific to ret9 and ret51 reveals that both isoforms are produced. These primers show no contaminating genomic DNA amplification in the ?no RT? controls. EXPRESSION / LABELING:
|
|
Ret regulates enteric neuron colonization of the gut in a dose-dependent manner. (A?D) Zebrafish injected with translation blocking antisense morpholino oligos against ret ret MO at the one- to two-cell stage are analyzed for the presence of Hu-expressing enteric neurons at 4dpf. Compared to control morpholino injected fish that possess enteric neurons along the full length of the gut tube (A), zebrafish injected with 6 ng retMO show an absence of enteric neurons throughout the majority of the gut tube (B). While injection of 1 ng retMO has no apparent effect on enteric neurons (C), injection of 3 ng of retMO leads to partial phenotypes, with enteric neurons absent from only the distal most gut regions (compare (D) to (B) and (C)). Asterisk indicates the end of gut tube. Arrows indicate distal most enteric neurons. All panels show lateral views. Images for comparison are photographed at the same magnification. |
|
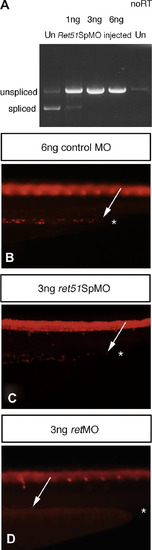
Enteric neurons colonize distal gut regions in the absence of Ret51. (A) RT-PCR analysis reveals that use of a splice blocking morpholino directed against ret51 sequences ret51SpMO on zebrafish embryos, leads to a dose-dependent block of splicing between exon 19 and exon 20, and therefore the loss of the spliced isoform of ret, ret51. RT-PCR on uninjected control fish Un, shows clearly the presence of spliced isoform of ret, ret51. Use of 1ng ret51SpMO leads to a significant reduction of splicing required to generate ret51, whereas use of 3 ng or 6 ng ret51SpMO results in the absence of splicing necessary to generate ret51, and leads to exclusive production of the unspliced form of ret, ret9. The no RT control shows low levels of amplification of contaminating genomic DNA, which as expected, amplifies a band identical in size to the unspliced RNA band. (B?D) Analysis of Hu positive enteric neurons at 4dpf reveals that zebrafish injected with ret51SpMO C show an equivalent distal extent of gut colonization relative to zebrafish injected with a control morpholino (B). In contrast, enteric neurons are absent from distal gut regions in retMO injected zebrafish (D). Asterisk indicates the end of gut tube. Arrows indicate distal most enteric neurons. Lateral views of distal gut are shown. Images for comparison are photographed at the same magnification. |
|
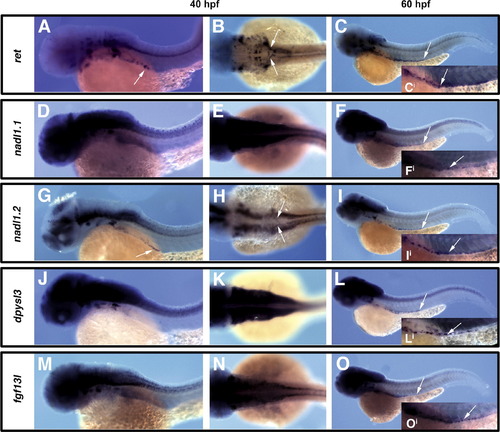
Orthologous zebrafish genes are expressed within the ENS and display similar profiles of expression to their mouse counterparts. Analysis of expression of ret (A?C), nadl1.1 (D?F), nadl1.2 (G?I), dpysl3 (J?L), and fgf13l (M?O), within enteric precursors and their derivatives at 40hpf lateral views (A, D, G, J and M) or dorsal views (B, E, H, K and N) and 60hpf lateral views (C, F, I, L and O). Like ret, nadl1.2 is expressed in migratory enteric precursors at earliest stages, as neural crest migrate in two streams towards the centrally located developing gut tube (arrows in A, B, G, H), and persists as enteric neurons are colonizing the gut tube (distal extent of expression within gut tube marked by arrow in C, I, and individual enteric neurons shown in higher magnification in insets Ci and Ii). In contrast, dpysl3, fgf13l, and nadl1.1 are not expressed in early migratory enteric precursors, but are expressed within enteric neurons colonizing the gut tube (see arrows marking distal extent of expression within gut tube in F, L, O, and marking individual enteric neurons in insets Fi, Li, and Oi). Images for comparison are photographed at the same magnification. |
|
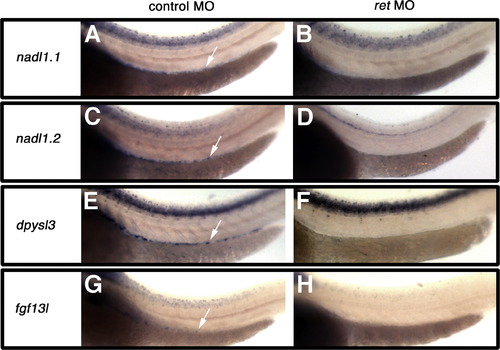
Loss of gene expression following retMO injection verifies that genes are expressed within the ENS. Injection of zebrafish with the retMO, which leads to an absence of enteric neurons in the gut, results in loss of nadl1.1, nadl1.2, dpysl3, and fgf13l expression in the gut of 3dpf embryos B, D, F, H as compared to control morpholino injected zebrafish expression within the gut denoted by arrows in A, C, E, G. fgf13l expression is clearly restricted to a subpopulation of the ENS G. Lateral views are shown. Images for comparison are photographed at the same magnification. EXPRESSION / LABELING:
PHENOTYPE:
|
|
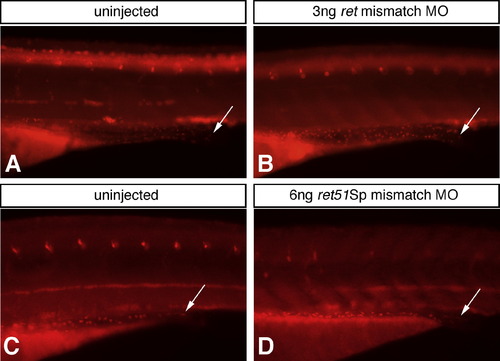
Sequence specificity for ret and ret51Sp MOs is assessed using control morpholinos containing five mismatched nucleotides along their length (mismatch MOs). Following injection of ret mismatch MO (B) or ret51Sp mismatch MO (D), fish are analyzed for the presence of Hu-expressing enteric neurons at 4dpf and compared to uninjected control fish (A, C). No difference in number or distribution of Hu-expressing enteric neurons is detected in these fish (A?D). This finding provides important control for the sequence specificity of ret and ret51Sp MOs used in Figs. 2 and 3, respectively. Critically, this result helps to demonstrate that the retMO effect on ENS number shown in Fig. 2 is sequence specific. Arrows indicate distal most enteric neurons. Images for comparison are photographed at the same magnification. |
|
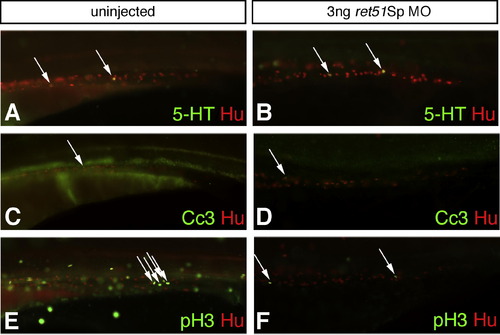
Fish injected with ret51Sp MO analyzed for effects on the serotonergic subpopulation of ENS, on cell death and cell proliferation. Fish injected with ret51Sp MO are compared to uninjected fish by analysis at 4dpf for the presence of Hu-expressing enteric neurons (red, A?F), serotonergic neurons (5-HT positive, green, A?B), cells undergoing cell death (cleaved caspase 3 (Cc3) positive, green, C?D), and proliferative cells (phoshpo-histone 3 (pH 3) positive, green E?F). Equivalent numbers of 5-HT expressing serotonergic neurons are observed in uninjected fish (A, arrows) and those injected with 3 ng ret51Sp MO (B, arrows). Only very small numbers of cells are observed to be undergoing cell death in uninjected (C, arrow) or 3 ng ret51Sp MO injected fish (D, arrow). Significant reductions in numbers of pH 3 positive cells are observed globally in 3 ng ret51Sp MO injected fish (F) as compared to uninjected counterparts (E), and the same trend is observed within the developing gut (E, F, arrows). This difference is cell proliferation likely to reflect the significant non-specific developmental delays observed in ret51Sp MO injected fish, which leads to low levels of survival of fish to stages showing the morphological criteria of a 4dpf fish (Kimmel et al., 1995). It is likely, therefore, that the observed reduction in cell proliferation is largely artefactual and not a reflection of a role for Ret51 in regulating cell proliferation. Images for comparison are photographed at the same magnification. |
Reprinted from Mechanisms of Development, 125(8), Heanue, T.A., and Pachnis, V., Ret isoform function and marker gene expression in the enteric nervous system is conserved across diverse vertebrate species, 687-699, Copyright (2008) with permission from Elsevier. Full text @ Mech. Dev.