- Title
-
Zebrafish pitx3 is necessary for normal lens and retinal development
- Authors
- Shi, X., Bosenko, D.V., Zinkevich, N.S., Foley, S., Hyde, D.R., Semina, E.V., and Vihtelic, T.S.
- Source
- Full text @ Mech. Dev.
|
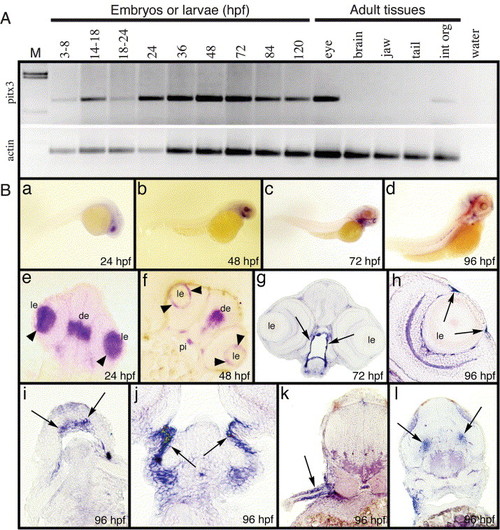
Temporal and spatial expression patterns of the pitx3 gene. (A) RT-PCR results from cDNA isolated from various staged embryos and larvae. Additionally, RT-PCR products resulting from different adult tissues are shown. Molecular weight markers and a control PCR reaction lacking DNA template are shown in the first and last lanes, respectively. The lower panel shows amplification of β-actin as a control. (B) The in situ hybridization of embryos and larvae using the full-length pitx3 riboprobe. Whole-mount embryos at 24, 48, 72 and 96 hpf (a?d). Frontal sections of 24, 48 and 72 hpf embryos at the level of the eye (e?g). The arrowheads in (e) and (f) point to lens, while expression surrounding the mouth is shown in (g). At 96 hpf, expression is shown within the iris (h), Meckel′s cartilage (i), branchial arch (j), pectoral fin (k) and lateral trunk (l). Abbreviations: de, diencephalon; le, lens; pi, pituitary. |
|
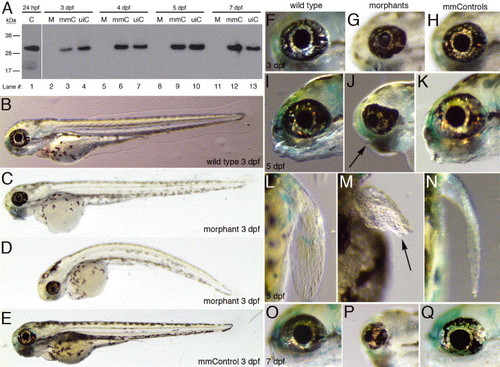
Morpholino knockdown of Pitx3 protein and the morphant phenotypes. (A) Immunoblot analysis of protein extracts from Morph1-injected embryos (M), a five base mismatch control (mmC) and an uninjected control (uiC) at 3 dpf (lanes 2?4), 4 dpf (lanes 5?7), 5 dpf (lanes 8?10) and 7 dpf (lanes 11?13). Lane 1 reveals the 32 kDa Pitx3 protein in 24 hpf whole embryo extract using the anti-Pitx3 polyclonal serum. Pitx3 protein expression was reduced in the morphant extracts compared to the mismatch and uninjected controls. Immunodetection of actin revealed that equivalent amounts of protein were loaded for all three samples from each time point (not shown). (B?Q) The pitx3 morphant phenotypes. Lateral views of wild type, morphants and mismatch control fish at 3 dpf (B?E). The small morphant eyes are shown in lateral views relative to the wild type and mismatch controls at 3 and 5 dpf (F?H and I?K, respectively). At 5 dpf, the morphants lack normal lower jaws (J; arrow) and pectoral fins (M; arrow). At 7 dpf, the morphant eyes are further reduced in size compared to the wild type and mismatch control (O?Q). |
|
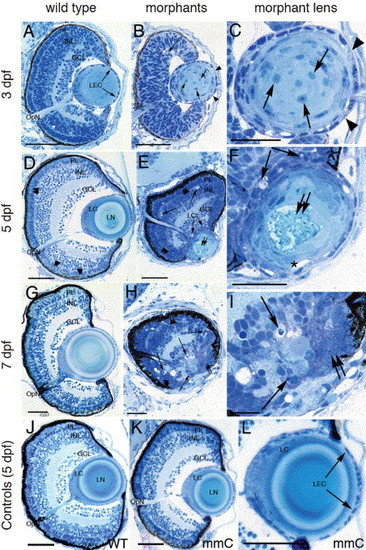
Histological analysis of the morphant lens and retina phenotypes. Stained eye sections of wild type (left column) and anti-pitx3 morphants at low and high magnification (center and right columns, respectively) at 3, 5 and 7 dpf. Sections of control eyes (wild type and mismatch control) are shown in the bottom row of panels (J?L). Retinal cell pyknosis (long arrows in B, E, F, H and I) and retention of the lens fiber cell nuclei (short arrows in B and C) are visible in the morphants, but absent in the controls. At 5 dpf, the epithelial cells in the morphant lens are irregularly shaped and overlie one another in the lens cortex (E, F). The lens epithelial cell layer contains gaps that lack nuclei or exhibits overlapping cells (B and C, arrowheads). Gaps exist between the lens epithelial cells and the underlying fiber cells in the distal lens (E and F, asterisks). Central lens fiber degeneration (double arrows) is observed in the morphant lens (E, F) at 5 dpf, but not in the wild-type lens (D). At 7 dpf, the retinal regression and lens degeneration in the morphant (H, I) are severe relative to wild type (G). The lens and retina of the mismatch control (K, L) appear identical to the wild type (J). Abbreviations: PL, photoreceptor layer; INL, inner nuclear layer; GCL, ganglion cell layer; OpN, optic nerve; LEC, lens epithelial cells; LN, lens nucleus; LC, lens cortex; IPL, inner plexiform layer. The scale bars represent 50 μm. |
|
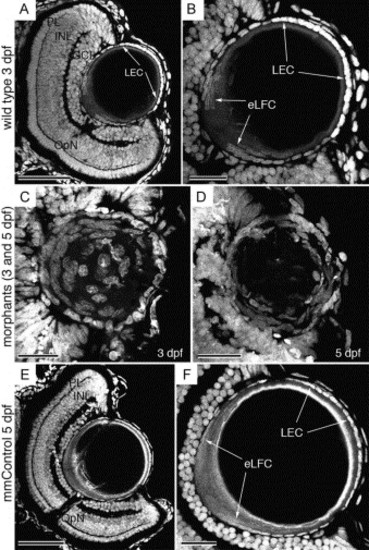
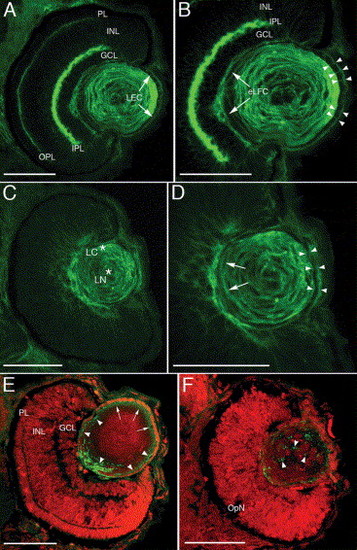
Morphology and spatial organization of the lens nuclei. Propidium iodide-stained wild type (A, B), morphant (C, D) and mismatch control (E, F) frozen eye sections. At 3 dpf, the normal retinal laminar arrangement (A) and lens epithelial cell organization (B) is shown. The epithelial cells covering the distal aspect of the lens (LEC) and the proximal elongating fiber cells (eLFC) are indicated by arrows (A, B). The 3 dpf morphant lens retains the central fiber cell nuclei (C) and the epithelial cell population exhibits enlarged and irregularly shaped nuclei and multilayering at 3 and 5 dpf (C, D). The mismatch control retina and lens (E, F) are indistinguishable from wild type (A, B). Abbreviations: PL, photoreceptor layer; INL, inner nuclear layer; GCL, ganglion cell layer; LEC, lens epithelial cells; eLFC, elongating lens fiber cells. Scale bars in A, B, E and F represent 50 μm, while the scale bars in C and D are 20 μm. |
|
Actin fails to localize properly in the morphant lens epithelial cells. Phalloidin-AlexaFluor 488 (A?D) was used to stain the filamentous actin in lenses and retinas of wild type (A, B) and morphants (C, D) at 3 dpf. In the control lens, actin is localized in a concentric pattern associated with the fiber cell membranes in both the nuclear and cortical regions (A, B). Lens epithelial cells (A, LEC) exhibit actin localization to both the distal (basal) and lateral aspects of the individual epithelial cells (B, arrowheads). Actin staining of the morphant lens reveals poorly defined elongating fiber cells (D, arrows) and a diffuse actin network in the lens epithelial cells (D, arrowheads). The sections also reveal intense actin staining within the retinal inner (IPL) and outer (OPL) plexiform layers (A, B), which is absent in the morphant (C). The lens-specific zl-1 monoclonal antibody labels the cortical region of the wild-type lens (E, arrowheads) and is localized near the apical?apical interface between the epithelial and fiber cells (E, arrows). In the morphant (F), the zl-1 protein is scattered throughout the lens and appears to be associated with the nucleated fiber cells (arrowheads). Abbreviations: PL, photoreceptor layer; INL, inner nuclear layer; GCL, ganglion cell layer; OPL, outer plexiform layer; IPL, inner plexiform layer; LEC, lens epithelial cells; eLFC, elongating lens fiber cells; OpN, optic nerve. Scale bars represent 50 μm. |
|
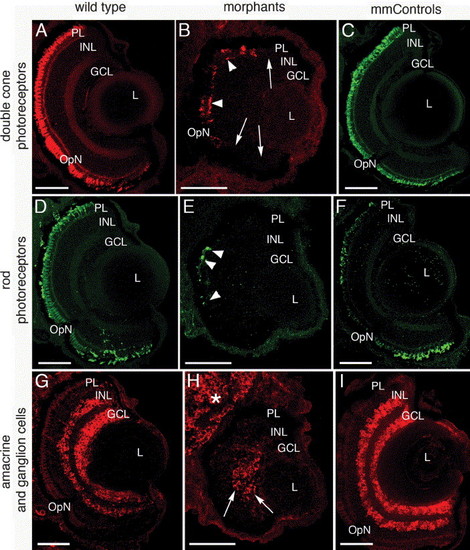
Reduction in retinal neurons revealed by immunohistochemistry at 5 dpf. Wild type (left column), morphant (center column) and mismatch control (right column) retinal cell types were identified by immunolocalization of zpr-1 (double cone photoreceptor cells, A?C), rhodopsin (rod photoreceptor outer segments, D?F), and HuC/D (amacrine and ganglion cells, G?I) in frozen eye sections. While some double cone cells are identified in the morphant (B, arrowheads), large regions of the retina lack this cell type (B, arrows). Similarly, only small numbers of rhodopsin-positive photoreceptors were detected in the morphants (E, arrowheads) compared to the wild type (D) and mismatch control retinas (F). The amacrine and ganglion cells (G?I) were also reduced in the morphants and were characterized by an absence of immunolabeling towards the retinal margins (compare H to G and I). Abbreviations: PL, photoreceptor layer; INL, inner nuclear layer; GCL, ganglion cell layer; OpN, optic nerve; L, lens. The scale bars represent 50 μm. EXPRESSION / LABELING:
|

Unillustrated author statements |
Reprinted from Mechanisms of Development, 122(4), Shi, X., Bosenko, D.V., Zinkevich, N.S., Foley, S., Hyde, D.R., Semina, E.V., and Vihtelic, T.S., Zebrafish pitx3 is necessary for normal lens and retinal development, 513-527, Copyright (2005) with permission from Elsevier. Full text @ Mech. Dev.