- Title
-
Comparative functional genomics revealed conservation and diversification of three enhancers of the isl1 gene for motor and sensory neuron-specific expression
- Authors
- Uemura, O., Okada, Y., Ando, H., Guedj, M., Higashijima, S., Shimazaki, T., Chino, N., Okano, H., and Okamoto, H.
- Source
- Full text @ Dev. Biol.
|
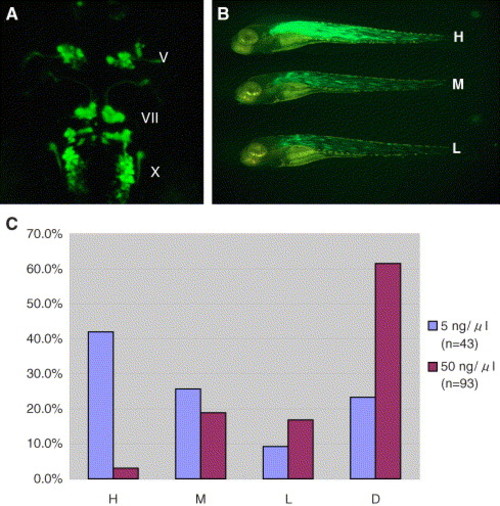
Effectiveness of transient transgene expression by injection of circular plasmid at a very low concentration. (A) Dorsal view of the 48-hpf embryo injected with the CM-isl1:GFP plasmid at a very low concentration. Note that most of the cranial motor neurons transiently expressed GFP at a level comparable to that of the embryo stably transgenic for the same DNA construct (Higashijima et al., 2000). V, trigeminal motor neurons; VII, facial motor neurons; X, vagus motor neurons. (B and C) The embryos injected with the α-actin:GFP plasmid were subdivided into four groups according to the degree of contribution of GFP-positive cells to the whole embryo, i.e., to groups H, M, and L, which represent embryos that express GFP in most muscles, scattered muscle cells plus some clusters of muscle cells, and in scattered separate muscle cells alone, respectively, and group D for dead embryos. (B) Representative embryos from each group are shown. Note that even in the embryo from group H, which expressed high levels of GFP in trunk muscles, craniofacial and fin muscles rarely expressed GFP. (C) The bar graph shows the percentage of embryos belonging to each group among all the embryos injected with the α-actin:GFP plasmid. Injection was performed at a concentration of 5 ng/μl (blue bar) or 50 ng/μl (red bar). |
|
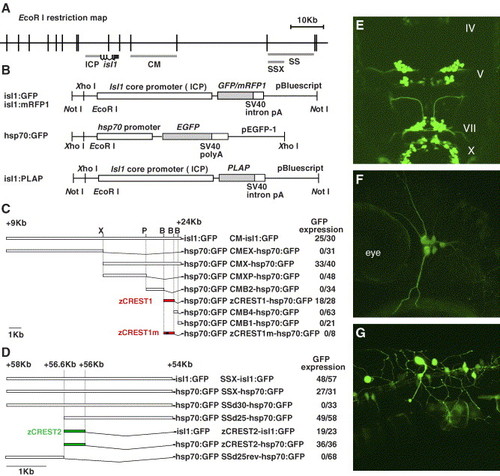
Genomic structure, plasmid constructs, and results of the transient transgene expression assay. (A) EcoRI restriction map of the genomic region flanking the zebrafish isl1 gene. The lines under the map indicate the regulatory regions of the isl1 gene. ICP, the isl1 gene core promoter; CM, an enhancer for the cranial motor neurons; SS and SSX, enhancers for the primary sensory neurons and the fin motor neurons. The thick line, isl1, indicates the transcribed unit of the isl1 gene. Note that the isl1 gene is composed of six exons divided by five introns. (B) Maps of plasmids used in this study. Each enhancer fragment was inserted immediately upstream of the promoter sequences. PLAP, human placental alkaline phosphatase gene. (C and D) Each plasmid construct shown here was injected into one-cell stage zebrafish embryos to examine its enhancer activity. The number on the right side of the name of each construct indicates the number of embryos exhibiting expression of GFP in cranial motor neurons (C) or primary sensory neurons (D) among all injected embryos. X, XhoI; P, PstI; B, BstXI. (C) Note that nucleotide conversion in TAAT to GGGG (termed zCREST1m, asterisk in Fig. 5A) completely inactivated its enhancer activity. (E) Dorsal view of a 48-hpf embryo injected with the zCREST1-hsp70:GFP plasmid and expressing GFP in the majority of cranial motor neurons IV, trochear motor neurons. (F and G) Lateral views of 32-hpf embryos injected with the zCREST2-hsp70:GFP plasmid and expressing GFP in the trigeminal ganglion neurons (F) and Rohon-Beard neurons (G). |
|
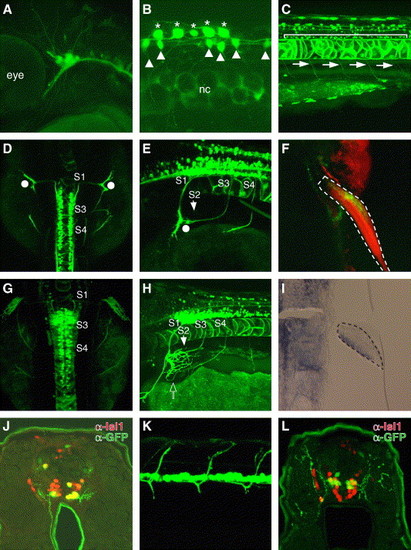
Sensory and motor neuron-specific GFP expression driven by zCREST2 in stable transgenic zebrafish. zCREST2 drives GFP expression both in the primary sensory neurons and in subsets of motor neurons innervating the ventral trunk muscles and the abductor pectoral fin muscle in transgenic zebrafish embryos. (A?E, G, and H) Lateral (A?C, E, and H) and dorsal (D and G) views of Tg(zCREST2-hsp70:GFP)rw011 embryos. At 36 hpf, GFP expression was observed in the trigeminal ganglion neurons (A) and in Rohon-Beard neurons (asterisks in B). GFP expression was detected in other tissues, such as the notochord and some commissural interneurons in the spinal cord (arrowheads). nc, notochord. (C) GFP was expressed in the spinal motor neurons (bracket) that project their axons ventrally (arrows) by 72 hpf. (D, E, G, and H) The GFP-positive fin motor neurons were distributed from the spinal segment 1 (S1) through 4 (S4), as described for Medaka fish embryos (Okamoto and Kuwada, 1991). (D and E) At 54 hpf, the GFP-positive fin motor nerves from S1 to S3 had reached the base of the fin bud without entering (white dots). Meanwhile, the nerve from S4 was still extending toward the fin bud. (G and H) By 72 hpf, the GFP-positive nerves from S1 to S3 had completely converged and entered the pectoral fin at the dorsal end of its base, while the nerve from S4 had entered at the opposite end (open arrow). (F) Ventral view of the 120-hpf Tg(zCREST2-hsp70:GFP)rw011 embryo stained with rhodamine phalloidin. The GFP-positive fin motor nerves terminated in the abductor pectoral fin muscle. The pectoral fin is enclosed in a broken line. (I) Expression of the lmx1b gene in the adductor pectoral fin muscle of the wild-type 50-hpf embryo. The developing fin bud is enclosed with a broken line. (J) GFP-positive spinal motor neurons in 72-hpf Tg(zCREST2-hsp70:GFP)rw011 embryo were the ventral subgroup of the Isl1-positive spinal motor neurons. (K and L) In 72-hpf Isl1-GFP-transgenic zebrafish, GFP-positive spinal motor neurons projected their axons dorsally (K, lateral view). (L) GFP-positive spinal motor neurons in the 72-hpf Isl1-GFP-transgenic zebrafish embryo were the dorsal subgroup of Isl1-positive spinal motor neurons. EXPRESSION / LABELING:
|
|
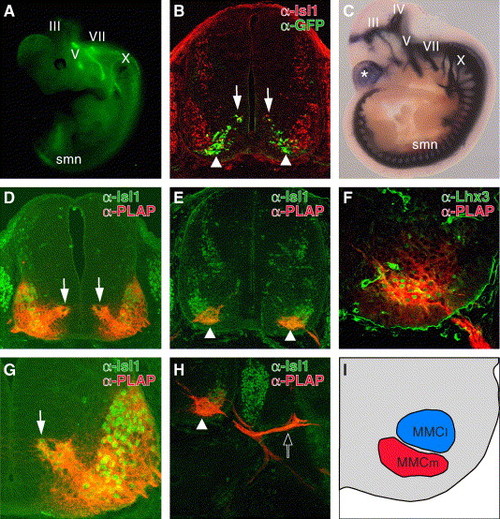
Functional conservation of CREST1 for motor neuron-specific gene expression in mouse. (A) Lateral view of E11.5 Isl1-GFP transgenic mice as seen through a dissecting fluorescence microscope. III, oculomotor nerve; V, trigeminal nerve; VII, facial nerve; X, vagus nerve. This transgenic embryo carries exactly the same transgene as Isl1-GFP transgenic zebrafish (Higashijima et al., 2000). (B) Cross-section of the E11.5 Isl1-GFP embryo at the thoracic level. GFP was expressed in migrating postmitotic motor neurons (arrows) and the ventromedial subset of motor neurons (arrowheads). (C) Lateral view of the E10.5 Tg(hCREST1/isl1-PLAP) embryo stained for alkaline phosphatase activity. Both somatic and visceral motor neurons expressed PLAP at this stage. The asterisk indicates PLAP expression in the telencephalon that was commonly observed in lines examined (data not shown). IV, trochlear nerve. (D?H) Cross-sections at the thoracic level of E11.5 (D and G) and E12.5 (E, F, and H) Tg(hCREST1/isl1-PLAP) embryos double labeled with anti-Isl1 (D, E, G, and H) anti-Lhx3 (F) antibody (green) and anti-PLAP antibody (red). At E11.5 (D and G), all Isl1-positive spinal motor neurons expressed PLAP. The dorsomedially located postmitotic-migrating motor neurons (arrows) also expressed PLAP at this stage. The weak expression of PLAP in MMCl cells (F and H) may reflect this early widespread activity of CREST1. By E12.5 (E and H), PLAP expression became restricted to the ventromedial subset of Isl1-positive motor neurons (arrowheads). Note that at this stage, PLAP-positive axons projected into dorsal trunk muscles (open arrow in H). (F) All Lhx3-positive spinal motor neurons also expressed PLAP at this stage, demonstrating that the PLAP-positive neurons were the MMCm motor neurons. (I) The schematic illustration of F. The regions containing PLAP-positive MMCm neurons and PLAP-negative MMCl neurons are indicated in red and blue, respectively. |
|
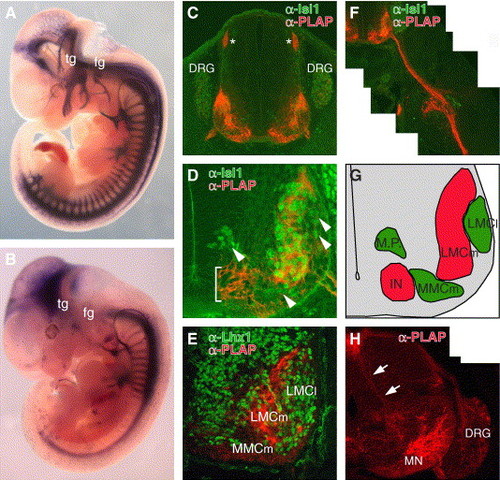
Functional conservation and diversification of CREST2 for motor and sensory neuron-specific gene expression in mouse. (A and B) Lateral views of the E11.5 Tg(zCREST2/isl1-PLAP) embryo (A) and Tg(hCREST2/isl1-PLAP) embryo (B). In both embryos, PLAP was expressed in spinal motor neurons that extend their axons ventrally, while PLAP expression in the sensory neurons, such as trigeminal ganglion neurons (tg), facial ganglion neurons (fg), and dorsal root ganglion neurons (DRG), was observed only in the Tg(zCREST2/isl1-PLAP) embryo (see also C and H). (C, D, and F) Cross-sections at the brachial level of E11.5 Tg(zCREST2/isl1-PLAP) embryos doubly labeled with anti-Isl1 antibody (green) and anti-PLAP antibody (red) showing PLAP expression in the subset of spinal motor neurons innervating the ventral muscles of the limb and in DRG neurons. Asterisks in C indicate the dorsal root entry zone (DREZ) where the central axons of the DRG neurons enter the spinal cord. Close-up view of the ventral spinal cord (D) showing expression of PLAP in the specific subset of spinal motor neurons, excluding the dorsomedially, ventromedially, and dorsolaterally (arrowheads) located Isl1-positive motor neurons. In this particular line, ectopic PLAP expression was also observed in the ventralmost region of the spinal cord (bracket). The specific projection of PLAP-positive motor axons into the ventral limb bud is shown in F. (E) Double labeling with anti-Lhx1 antibody (green) and anti-PLAP antibody (red) at the brachial level of the E11.5 Tg(zCREST2/isl1-PLAP) embryo revealed that PLAP-positive motor neurons were negative for Lhx1, a marker for LMCl motor neurons, supporting their identity as LMCm motor neurons. Lhx1 was also expressed in other interneurons (Tsuchida et al., 1994). (G) Schematic illustration of D. Regions containing PLAP-positive and PLAP-negative neurons are indicated in red and green, respectively. M.P., motor neuron precursors; IN, interneurons. (H) Brachial level of the E14.5 Tg(zCREST2/isl1-PLAP) embryo labeled with anti-PLAP antibody (red). The PLAP-positive Ia afferent fibers (arrows) were observed penetrating the spinal cord and projecting ventrally to the motor neurons (MN) in the ventral horn. |
|
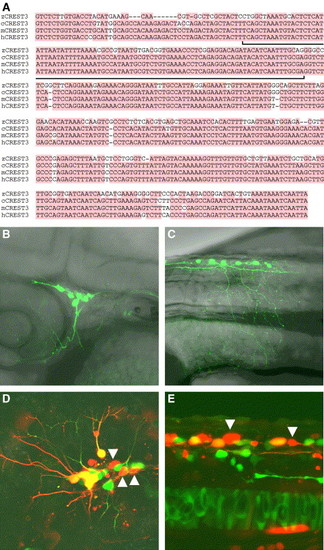
Structural and functional conservation of CREST3 during evolution. (A) Sequence comparisons between CREST3s. No similar sequences to CREST3 were found in the fugu genome. Only the most conserved regions among species are shown. The entire nucleotide sequences of zCREST3, cCREST3, mCREST3, and hCREST3 were deposited in DDBJ under accession numbers AB0158305, AB167734, AB158308, and AB158311, respectively. The nucleotides conserved in more than three of all species are shaded by the red color. The bracket indicates the conserved region between CREST3s and C3Rs (Fig. 9D). (B and C) Lateral views of 36-hpf embryos injected with the zCREST3-isl1:GFP plasmid and exhibiting expression of GFP in trigeminal ganglion neurons (B) and Rohon-Beard neurons (C). (D and E) Lateral views of 24-hpf Tg(zCREST2-hsp70:GFP)rw011 embryos injected with the zCREST3-isl1:mRFP1 plasmid and exhibiting mRFP1 expression in trigeminal ganglion neurons (D) and Rohon-Beard neurons (E). Note that there are some mRFP1 single-positive neurons in both trigeminal ganglion neurons and Rohon-Beard neurons (arrowheads). |
|
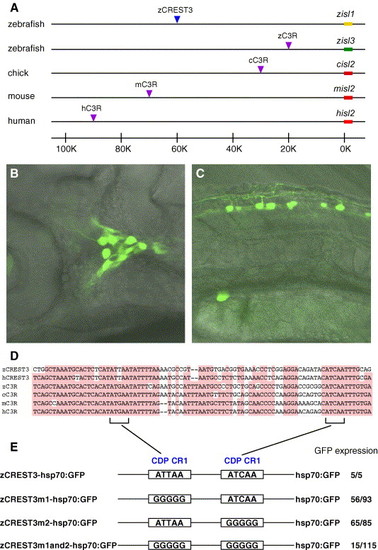
Structural and functional conservation of sensory neuron-specific enhancers CREST3 and C3R. (A) Comparison of the distribution of CREST3 and C3R in each species. Yellow and green bars indicate the coding regions of zebrafish isl1 and isl3, respectively. Red bars indicate the coding regions of isl2 of chick, mouse, and human. The blue arrowhead indicates zCREST3 locus. In contrast, purple arrowheads indicate the C3R loci. No similar sequences to C3R were found in the fugu genome. (B and C) Lateral views of 32-hpf embryos injected with the zC3R-hsp70:GFP plasmid and GFP expression in the trigeminal ganglion neurons (B) and Rohon-Beard neurons (C). (D) Sequence comparisons between CREST3 and C3R. Only the most conserved regions between CREST3 and C3R are shown. Nucleotides conserved in more than four of all are shaded by the red color. The entire nucleotide sequences of zC3R, cC3R mC3R, and hC3R were deposited in DDBJ under accession numbers AB179552, AB179553, AB179554, and AB179555, respectively. The highly conserved region between CREST3 and C3R contains two CDP CR1 recognition motifs (brackets). (E) Mutational analysis of zCREST3. Both or either of the CDP CR1 sites was converted to GGGGG. The number on the right side of the name of each plasmid construct indicates the number of embryos exhibiting expression of GFP in primary sensory neurons among all injected embryos. |
Reprinted from Developmental Biology, 278(2), Uemura, O., Okada, Y., Ando, H., Guedj, M., Higashijima, S., Shimazaki, T., Chino, N., Okano, H., and Okamoto, H., Comparative functional genomics revealed conservation and diversification of three enhancers of the isl1 gene for motor and sensory neuron-specific expression, 587-606, Copyright (2005) with permission from Elsevier. Full text @ Dev. Biol.