- Title
-
FKRP-dependent glycosylation of fibronectin regulates muscle pathology in muscular dystrophy
- Authors
- Wood, A.J., Lin, C.H., Li, M., Nishtala, K., Alaei, S., Rossello, F., Sonntag, C., Hersey, L., Miles, L.B., Krisp, C., Dudczig, S., Fulcher, A.J., Gibertini, S., Conroy, P.J., Siegel, A., Mora, M., Jusuf, P., Packer, N.H., Currie, P.D.
- Source
- Full text @ Nat. Commun.
|
PHENOTYPE:
|
|
|
|
|
|
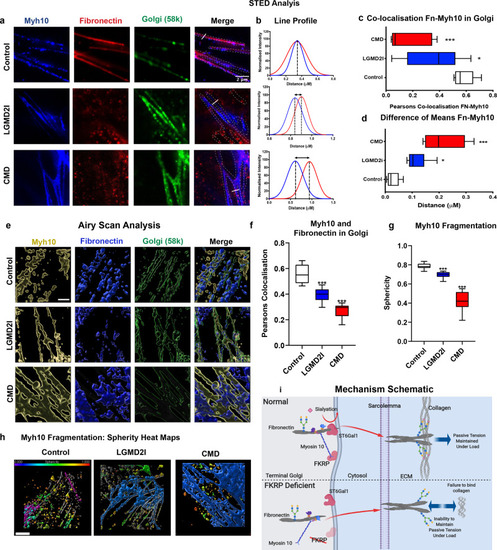
Fibronectin and myosin10 protein localisation within the trans-Golgi, as determined by STED microscopy and Gaussian distribution line profile analyses, images representative of a minimum of three repeats separated by a minimum of a week and three technical repeats. |

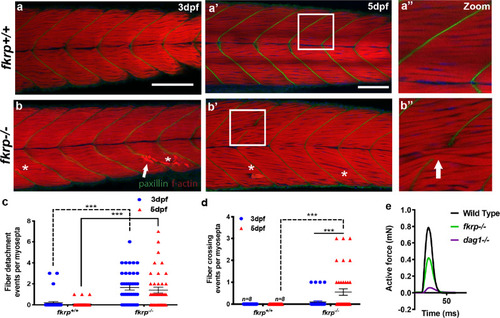
ZFIN is incorporating published figure images and captions as part of an ongoing project. Figures from some publications have not yet been curated, or are not available for display because of copyright restrictions. |

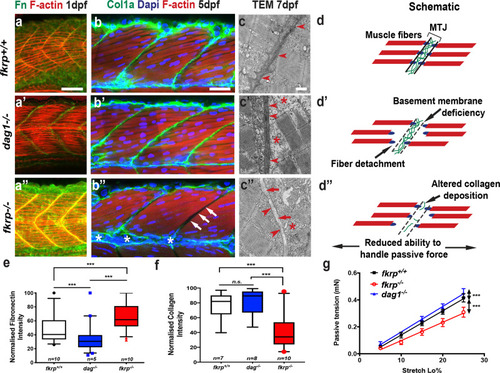
ZFIN is incorporating published figure images and captions as part of an ongoing project. Figures from some publications have not yet been curated, or are not available for display because of copyright restrictions. |

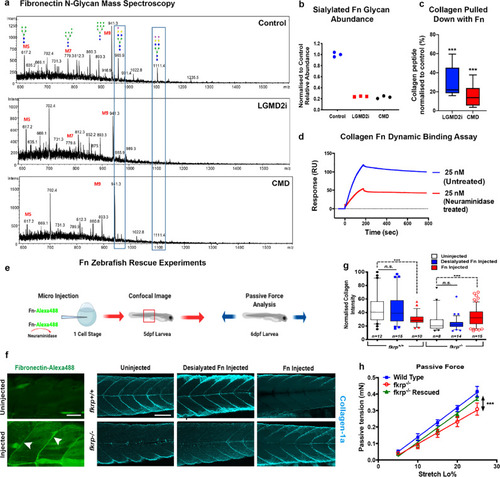
ZFIN is incorporating published figure images and captions as part of an ongoing project. Figures from some publications have not yet been curated, or are not available for display because of copyright restrictions. |