- Title
-
Epigenetic factors Dnmt1 and Uhrf1 coordinate intestinal development
- Authors
- Ganz, J., Melancon, E., Wilson, C., Amores, A., Batzel, P., Strader, M., Braasch, I., Diba, P., Kuhlman, J.A., Postlethwait, J.H., Eisen, J.S.
- Source
- Full text @ Dev. Biol.
|
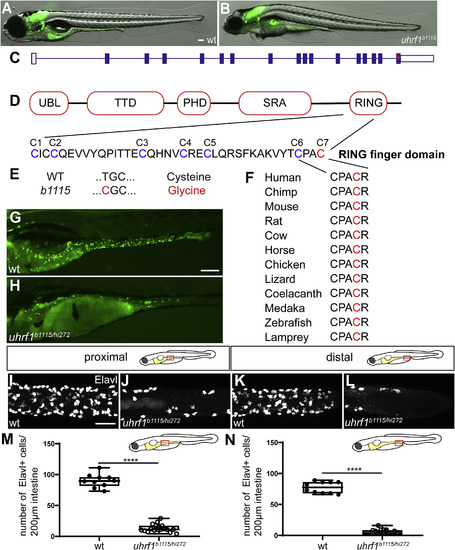
The zebrafish b1115 mutation has a non-synonymous single nucleotide polymorphism in an ultra-conserved amino acid of the RING domain of uhrf1. (A?B) Live images of 5 dpf wildtype (A) and uhrf1b1115 mutant (B) larvae show that mutants have smaller eyes and heart edema compared to wildtype siblings. (C) Exon 16 of uhrf1 contains the mutation (red asterisk) in the b1115 allele. (D) Schematic of important protein domains of uhrf1 with a close-up of the seven conserved cysteines (Tauber and Fischle, 2015). (E) A non-synonymous single nucleotide polymorphism (T=>C) mutates the conserved cysteine 7 (red) to a Glycine. (F) Cysteine 7 (red) is conserved among phylogenetically diverse vertebrate species. (G) Complementation cross between the b1115 allele and the hi272 allele of uhrf1 shows failure to complement in 5 dpf larvae as demonstrated by fewer phox2b:EGFP enteric neurons (green; H) compared to wildtype (green; G). In both proximal (J) and distal (L) intestine, uhrf1b1115/hi272 mutants have fewer enteric neurons than wildtype siblings at 5 dpf (proximal I; distal K) as quantified in M and N. (M,N) Quantification of Elavl positive cells per 200??m of proximal or distal intestine in wildtype or uhrf1b1115/hi272 mutant [wildtype n?=?10 (proximal), n?=?10 (distal); uhrf1b1115/hi272 n?=?19 (proximal), n?=?18 (distal)]. UBL, ubiquitin like; TTD, tandem tudor domain; PHD, plant homeodomain; SRA, SET and RING associated; PBR, polybasic region; RING, really interesting and new gene. A,B,G,H: side-views of whole-mount zebrafish larvae at 5 dpf. I?L: Confocal images of dissected intestines at 5 dpf. Scale bar?=?100??m in A,B,E,F; Scale bar?=?50??m in I-L. Unpaired t-test: **** ?= ?p ?< ?0.0001. PHENOTYPE:
|
|
The uhrf1 mutant ENS progenitor phenotype emerges around 72 hpf. Whole-mount side views of wildtype (A,C) and uhrf1 mutants (B,D). uhfr1 mutants have fewer phox2b:EGFP positive cells (green) than wildtype siblings at 72 hpf (A,B) and 96 hpf (C,D). Scale bar?=?100??m. PHENOTYPE:
|
|
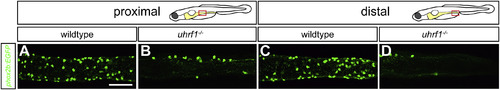
uhrf1 mutants have fewer neurons in both proximal and distal intestine. Confocal images of dissected intestines of wildtype (A,C) and uhrf1 mutants (B,D). uhfr1 mutants have fewer phox2b:EGFP positive enteric neurons (green) than wildtype siblings at 5 dpf (A,C) in both proximal (B) and distal (D) intestine. Scale bar?=?50??m. PHENOTYPE:
|
|
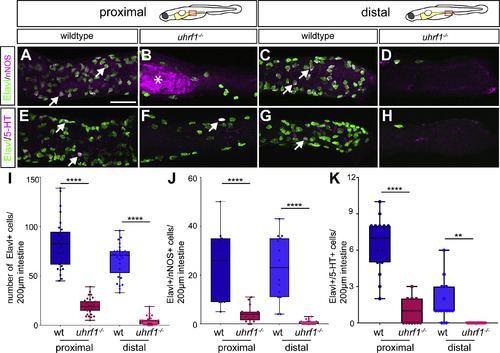
Both of the major ENS neuronal subtypes are reduced in uhrf1 mutants. Confocal images of dissected intestines from wildtypes (A,C,E,G) and uhrf1 mutants (B,D,F,H) stained for expression of the pan-neuronal marker Elavl, and either nNOS or 5-HT. In both proximal (B,F) and distal (D,H) intestine, uhrf1 mutants have fewer enteric neurons than wildtype siblings at 5 dpf (proximal A,E; distal C,G) as quantified in (I). Both nNOS and 5-HT expressing ENS neuronal subtypes show similar reductions at 5 dpf [Elavl (green), nNOS or 5-HT (magenta)] as quantified in (J,K). Arrows point to examples of nNOS or 5-HT positive neurons. (I) Quantification of Elavl positive cells per 200??m of proximal or distal intestine in wildtype (blue) or mutant (red) [wildtype n?=?30 (proximal), n?=?29 (distal); uhrf1?/? n?=?29 (proximal), n?=?30 (distal)]. (J?K) Quantification of Elavl and nNOS (J) and 5-HT (K) positive cells per 200??m proximal or distal intestine in wildtype (blue) and mutant (red) [nNOS: wildtype n?=?11 (proximal), n?=?15 (distal); uhrf1?/? n?=?17 (proximal), n?=?17 (distal); 5-HT: wildtype n?=?17 (proximal), n?=?14 (distal); uhrf1?/? n?=?15 (proximal), n?=?13 (distal)]. Asterisk in B indicates autofluorescent background common to uhrf1 mutant intestines. Scale bar?=?50??m in A-H. Unpaired t-test: **** ?= ?p ?< ?0.0001; ** ?= ?p ?< ?0.01. |
|
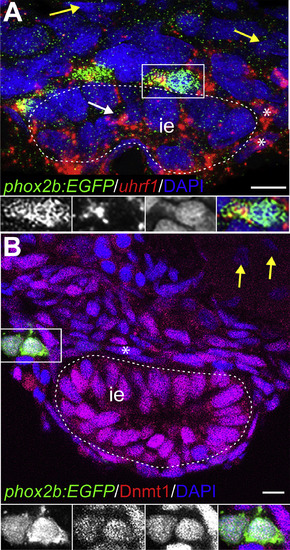
uhrf1 and Dnmt1 are expressed in enteric, epithelial, and smooth muscle progenitors during development. Transverse sections of stained embryos. (A) At 48 hpf, uhrf1 (red) is expressed in phox2b:EGFP (green) positive EPCs and also in DAPI-positive (blue) ISMPs (asterisks) and epithelial progenitors (white arrow). Yellow arrows point to uhrf1 negative cells outside of the nascent intestine. White dashed line indicates intestinal epithelium (ie). Insets show enlargement of cell (white box), GFP, uhrf1, DAPI, and overlay from left to right. Note that the GFP staining appears speckled due to the RNA in situ hybridization procedure. (B) At 48 hpf, Dnmt1 (red) is expressed in phox2b:EGFP (green) positive EPCs and in DAPI-positive (blue) ISMPs (asterisk). Insets show enlargements of outlined cell, GFP, Dnmt1, DAPI and overlay from left to right. Yellow arrow points to Dnmt1 negative cell outside of the intestine. Scale bar?=?10??m in A-B. EXPRESSION / LABELING:
|
|
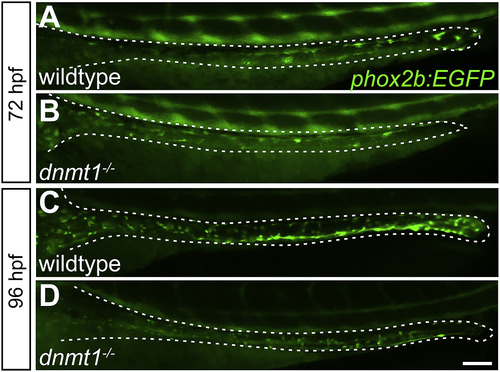
The dnmt1 mutant ENS progenitor phenotype emerges around 72 hpf. Whole-mount side views of wildtype (A,C) and dnmt1 mutants (B,D). dnmt1 mutants have fewer phox2b:EGFP positive cells (green) than wildtype siblings at 72 hpf (A,B) and 96 hpf (C,D). Scale bar?=?100??m. PHENOTYPE:
|

ZFIN is incorporating published figure images and captions as part of an ongoing project. Figures from some publications have not yet been curated, or are not available for display because of copyright restrictions. PHENOTYPE:
|
|
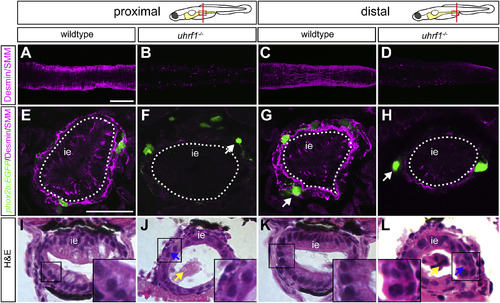
Disrupted smooth muscle development contributes to intestinal dysgenesis in uhrf1 mutants. Confocal images of 5 dpf intestines labeled with smooth muscle myosin (SMM) and desmin (magenta) antibodies. Dissected intestines (A?D) of wildtype (A,C) and uhrf1 mutants (B,D). In both proximal (B) and distal (D) intestine, uhrf1 mutants essentially lack smooth muscle cells, as revealed by whole-mount antibody staining. Transverse sections of wildtype (E,G) and uhrf1 mutants (F,H) showing loss of intestinal smooth muscle in mutants. White arrows point to phox2b:EGFP (green) positive ENS neurons that are further removed from the intestinal epithelium in mutants (F,H) than in wildtypes (G). Brightfield images of transverse sections of 5 dpf wildtypes (I,K) and uhrf1 mutants (J,L) stained with hematoxylin and eosin show disrupted proximal and distal intestinal development in mutants compared to wildtypes. Increased shedding of cells in uhrf1 mutants is indicated by a yellow arrow and disrupted intestinal epithelium with a blue arrow. Higher magnification insets of boxed areas indicate disrupted intestinal epithelium morphology in uhrf1 mutants. ie?=?intestinal epithelium. Scale bar?=?50??m in A-D, and 25??m in E-L. PHENOTYPE:
|
|
Disrupted smooth muscle development contributes to intestinal dysgenesis in dnmt1 mutants. Confocal images of 5 dpf intestines labeled with smooth muscle myosin (SMM) and desmin (magenta) antibodies. Dissected intestines (A?D) of wildtype (A,C) and dnmt1 mutants (B,D). In both proximal (B) and distal (D) intestine, dnmt1 mutants essentially lack smooth muscle cells, as revealed by whole-mount antibody staining. Transverse sections of wildtype (E,G) and dnmt1 mutants (F,H) showing loss of intestinal smooth muscle in mutants. White arrow points to phox2b:EGFP (green) positive ENS neuron that is further removed from the intestinal epithelium in mutants (H) than in wildtypes (G). ie?=?intestinal epithelium. Scale bar?=?50??m in A-D, and 25??m in E-H. PHENOTYPE:
|
|
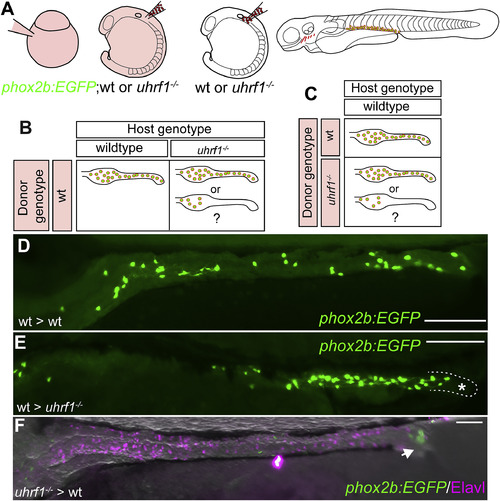
Genetic chimeras reveal that Uhrf1 functions cell-non-autonomously and cell-autonomously in ENS development. (A) Transplantation experiment diagram. Wildtype or uhrf1 mutant donor embryos that carry the phox2b:EGFP transgene were injected with rhodamine dextran. Vagal neural crest cells were transplanted from donors into unlabeled wildtype or uhrf1 mutant hosts. The contribution of transplanted cells was evaluated at 5 dpf. (B) Possible results for transplantation experiments to test cell-non-autonomous function of uhrf1 in ENS development. (C) Possible results for transplantation experiments to test cell-autonomous function of uhrf1 in ENS development. (D) phox2b:EGFP positive (green) wildtype cells transplanted into wildtype hosts can populate the entire intestine. Note that the density of these cells is less than in non-transplanted animals because they share the territory with unlabeled ENS cells of the host. (E) phox2b:EGFP positive (green) wildtype cells transplanted into uhrf1 mutant hosts can expand, but are consistently excluded from distal intestine (outlined and labeled with asterisk). (F) phox2b:EGFP positive (green) uhrf1 mutant cells transplanted into wildtype hosts can migrate to distal intestine in rare cases (1/5), but the population of transplanted mutant cells that successfully integrate into the ENS is smaller than that of the wildtype host cells as revealed by Elavl staining (magenta). (D?F) Lateral views of whole-mount zebrafish larvae at 5 dpf. Scale bar?=?100??m in D-F. |
Reprinted from Developmental Biology, 455, Ganz, J., Melancon, E., Wilson, C., Amores, A., Batzel, P., Strader, M., Braasch, I., Diba, P., Kuhlman, J.A., Postlethwait, J.H., Eisen, J.S., Epigenetic factors Dnmt1 and Uhrf1 coordinate intestinal development, 473-484, Copyright (2019) with permission from Elsevier. Full text @ Dev. Biol.