- Title
-
A zinc finger protein that regulates oligodendrocyte specification, migration, and myelination in zebrafish
- Authors
- Sidik, H., Talbot, W.S.
- Source
- Full text @ Development
|
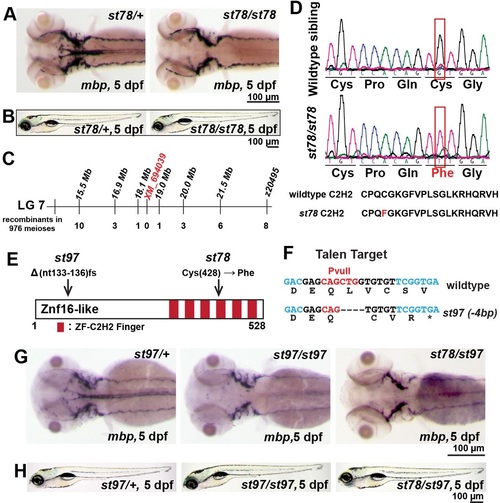
Mutations in znf16-like reduce mbp mRNA expression in the CNS. (A) At 5dpf, expression of mbp was specifically reduced in the CNS but not the PNS of st78 homozygote compared with the heterozygous sibling. (B) st78 mutant had normal gross morphology at 5dpf compared with the wild-type sibling. (C) Genetic mapping linked st78 locus to XM_694039 in linkage group 7. (D) Sequence chromatogram shows the lesion (red box) in the coding sequence of znf16-like (XM_694039). The point mutation in st78 changes a cysteine in a Cys2His2 zinc finger of Znf16l to a phenylalanine. (E) Schematic diagram depicting Znf16-like protein with all its zinc fingers, the location of the transversion in the st78 allele, and the 4bp deletion resulting in a frameshift in the st97 allele. (F) Nucleotide sequence of znf16l and the TALE nuclease targeted sequence (black) and diagnostic restriction enzyme site (red). Bottom line shows the st97 allele and the corresponding amino acid sequence resulting from the frameshift. (G) At 5dpf, expression of mbp was reduced in the CNS of st97 homozygote in comparison to the heterozygous sibling. st97 failed to complement st78, and the transheterozygous mutant lacked mbp in the CNS, similar to the homozygous mutants. The genotypes of all larvae shown in A,B,G,H were determined by PCR tests for their respective lesions. |
|
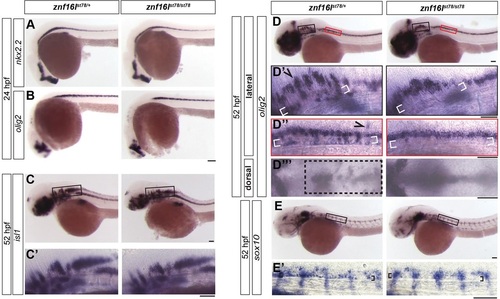
Developmental disruptions in znf16l mutants are specific to the oligodendrocyte lineage. (A,B) znf16l mutants displayed normal expression of nkx2.2 (A) and olig2 (B) at 24hpf. (C,C2) znf16l mutants also displayed normal expression of the motoneuron marker islet1 at 52hpf. (D,E) Defects in znf16l mutants were specific to cells in the oligodendrocyte lineage. (D-D4) olig2 is expressed by oligodendrocyte-lineage cells and a subset of motoneurons. The expression pattern of olig2 in wild-type siblings revealed groups of cells that had migrated dorsally (arrowhead), ventrally (white brackets) and laterally (box). These cell populations are missing or reduced in the mutants. D2 and D3 show regions in black and red boxes, respectively, in D. (E) sox10 expression is restricted to cells of the oligodendrocyte lineage in the CNS. Wild-type siblings displayed a group of cells (brackets in E2) immediately dorsal to the notochord that were missing in the mutants, confirming the disruption of oligodendrocyte-lineage cells in znf16l mutants. The genotypes of all embryos shown were determined by PCR tests for the st78 lesion. Scale bars: 100�m. |
|
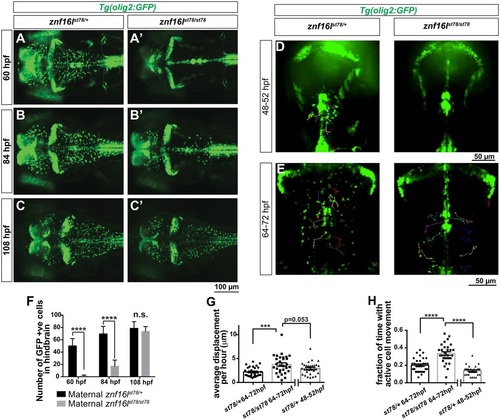
Disruption of OPC migration in znf16l mutants. (A-C) znf16lst78 mutants and wild-type siblings in Tg(olig2:GFP) background. The number of OPCs is greatly reduced in the mutants at 60hpf (A,A2) and 84hpf (B,B2), but recovers quickly by 108hpf (C,C2). (D,E) Dorsal view of time-lapse imaging from 48-52 and 64-72hpf, with trajectories of 10 individual OPCs as they migrated laterally out of the midline, shown using the MTrackJ cell-tracking tool (n=3 fish per genotype, per time point). Cells were tracked every 15min in real time (see Movies 1-4 for time-lapse images). Genotypes of all embryos were determined by PCR after imaging. (F) Quantification of the number of GFP-expressing cells in the hindbrain of Tg(olig2:GFP) znf16lst78 mutants and wild-type siblings (n=15 wild type, n=9 mutant). The numbers were significantly different at 60 and 84hpf, but not at 108hpf (two-tailed Student′s t-test, ****P<0.0001). (G) Quantification of average OPC displacement (�m) per hour of 30 tracked cells from each timepoint. Mutant OPCs traveled farther than wild-type siblings. (H) Quantification of the fraction of time for which each OPC was actively moving. Mutant OPCs spent more time moving compared with wild-type siblings at either stage. Error bars show s.e.m. significance with one-way ANOVA and post hoc comparisons. ***P=0.0003 in G; ****P<0.0001 in H. |
|
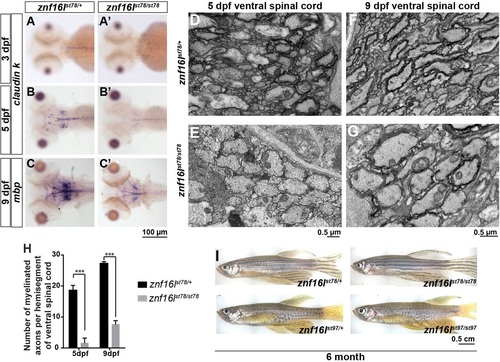
CNS myelin is reduced in znf16l mutants. (A-C) Expression of mature oligodendrocyte markers claudin k (A,B) and mbp (C) was reduced in znf16lst78 mutants between 3 and 9dpf. Expression of cldnk was reduced but detectable at 5dpf (B), and expression of mbp was reduced but detectable at 9dpf (C). (D,E) Transverse transmission electron microscopy images of ventral spinal cord showed that many wild-type axons were already myelinated in wild type (D) but not mutants (E). (F,G) At 9dpf, more axons were myelinated in the mutants (G) than at 5dpf, but the number was still much less than in wild-type siblings at 9dpf (F). (H) Quantification of the number of myelinated axons in ventral spinal cord at 5 and 9dpf. Error bars show s.d.; significance was determined with two-tailed Student′s t-test. ***P<0.001 in H. (I) Adult znf16l homozygous mutants are viable and fertile, with no gross morphological defects compared with their wild-type siblings for both of the alleles. Genotypes of all fish analyzed were determined by PCR assay. |
|
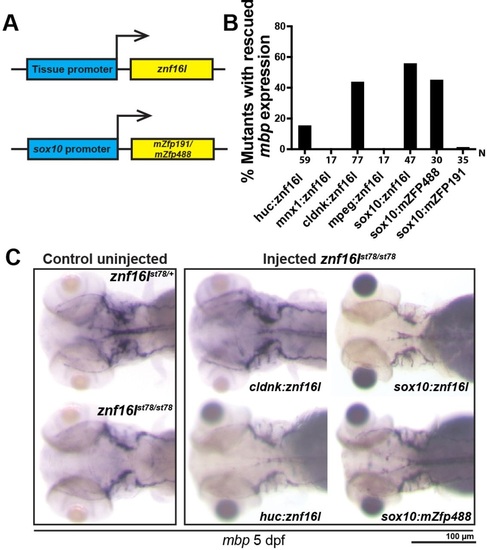
Znf16l functions cell autonomously in the oligodendrocyte lineage. (A) Diagram of the transgenic constructs driving expression of znf16l by tissue-specific gene promoters in neurons (huC), oligodendrocytes (cldnk, sox10) or macrophages (mpeg). Bottom panel shows another transgenic construct strategy to express mammalian zinc finger proteins, mZfp191 and mZfp488, in oligodendrocytes under the control of the sox10 promoter. (B) Plot quantifies the percentage of mbp rescue using different tissue promoters and different zinc finger proteins. (C) Representative images of mbp rescue in cldnk:znf16l-, sox10:znf16l-, huc:znf16l- and sox10:mZfp488-injected znf16l mutants. There is no visible difference in the level of rescues with the different constructs. Genotypes of all fish analyzed were determined by PCR assay. |
|
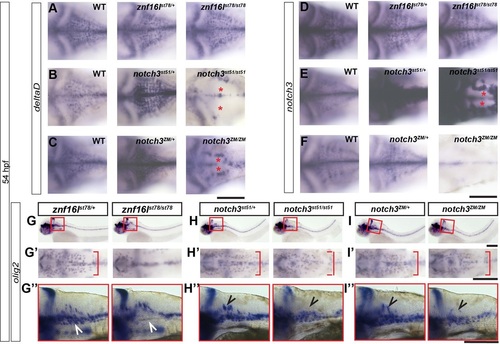
znf16l and notch3 mutants lack distinct populations of OPCs. In situ hybridization of znf16lst78, notch3st51 and notch3ZM embryos at 54hpf for deltaD (A-C), notch3 (D-F), and olig2 (G-I) mRNA expression. (A-F) znf16l homozygous mutants do not exhibit decreased deltaD (A) or notch3 (D) expression seen in notch3 homozygous mutants (asterisks in B,C,E). (G′-I′) znf16l mutants exhibit a delay in lateral migration of OPCs (red brackets in G′) that is not seen in either notch3 mutant (H′,I′). (G-I,G′′-I′′) Lateral view of the hindbrain shows that znf16l mutants are mostly lacking the ventral OPC population (white arrowheads), whereas notch3 mutants are mostly lacking the dorsal OPC population (black arrowheads). Genotypes of all fish analyzed were determined by PCR assay. Scale bars: 100�m. |
|
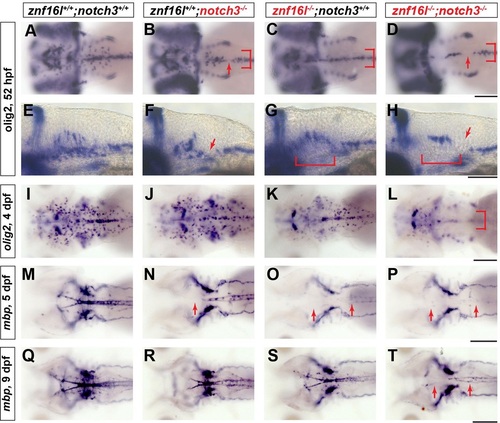
Severe defects in oligodendrocyte development in mutants lacking function of Znf16l and Notch3. In situ hybridization analyses of oligodendrocyte development and myelination in mutants lacking Notch3, Znf16l, or both. (A,E,I,M,Q) Wild-type oligodendrocyte precursors migrating laterally can be detected at 52hpf (A), and strong expression of mbp can be detected at 5dpf (M) continuing to 9dpf (Q). (B,F,J,N,R) notch3 mutants (st51) exhibit reduced lateral migration of OPCs at 52hpf and gaps in olig2 expression (red arrows in B,F and bracket in B). The number and distribution of OPCs recovers by 4dpf (J), although mbp expression is still reduced strongly in hindbrain of notch3 mutants (N). (R) By 9dpf mbp expression recovers, but remains distinguishably reduced compared with wild type. (C,G,K,O,S) znf16l single mutants (st97) show reduction of OPCs at an early stage (bracket in C,G) and a lack of mbp at 5dpf that recovers by 9dpf as shown before (O and S). (D,H,L,P,T) Mutants lacking both Znf16l and Notch3 have a more severe phenotype than either single mutant. Numbers of OPCs are strongly reduced and no OPCs migrate laterally similar to the znf16l mutant at 52hpf (red arrows and bracket in D,H). Numbers of OPCs are still reduced by 4dpf (L). mbp expression in double mutants is strongly reduced at 5dpf (P) and very slight expression is observed by 9dpf (T) compared with either single mutant (R and S). Genotypes of all fish analyzed were determined by PCR assay. Scale bars: 50�m. |
|
huC regulatory sequences drive expression in some OPCs. Tg:olig2:GFP stably transgenic embryos were injected with a construct encoding huC:mCherry-CAAX, which labeled cells expressing huC regulatory sequences with membrane-tethered mCherry (B, red). (A-C) Representative embryo showing (A) GFP in oligodendrocytes and motor neurons, (B) membrane tethered mCherry, and (C) merged image. OPCs and motor neurons were distinguished by morphology and position. In 3 of 38 injected embryos, some OPCs co-expressed GFP and membrane-tethered mCherry (arrow), indicating that the HuC construct may be expressed in a subset of cells of the oligodendrocyte lineage. (A′-C′) These images show magnified views of boxed regions in A-C. |