- Title
-
Podocalyxin regulates pronephric glomerular development in zebrafish
- Authors
- Ichimura, K., Powell, R., Nakamura, T., Kurihara, H., Sakai, T., and Obara, T.
- Source
- Full text @ Physiol. Rep.
|
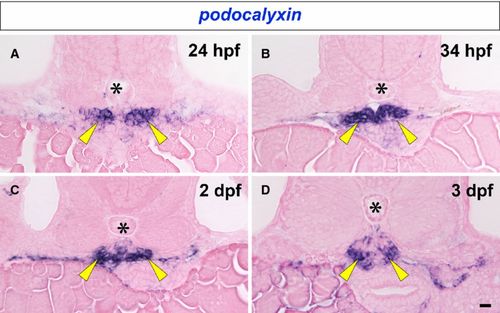
Expression of podocalyxin mRNA in developing pronephric glomerulus. podocalyxin mRNA is detected by in situ hybridization in the paired glomerular primordia at 24 and 34 hpf (arrowheads in A and B). Glomerular primordia fuse at the midline to form a glomerulus by 2 dpf, and podocalyxin expression continues in the glomerulus to 2 and 3 dpf (arrowheads in C and D). Asterisks indicate the notochord. Bar scale, 10 μm. EXPRESSION / LABELING:
|
|
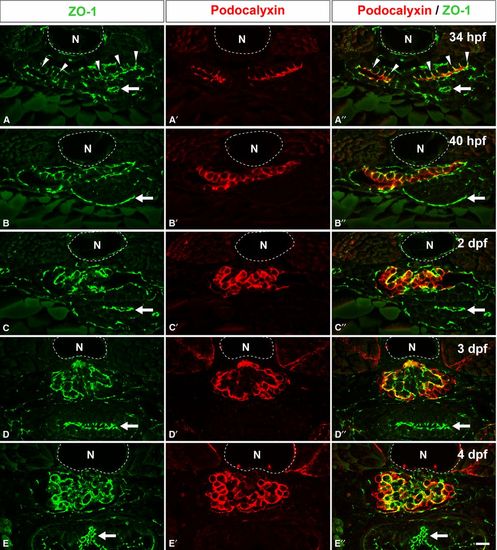
Localization of Podocalyxin in the developing pronephric glomerulus. Double immunofluorescence labeling for ZO1 (green) and Podocalyxin (red) was performed to examine the localization of Podocalyxin in zebrafish at 34 hpf (A-A′′), 40 hpf (B-B′′), 2 dpf (C-C′′), 3 dpf (D-D′′), and 4 dpf (E-E′′). At 34 hpf, immunoreactivity for ZO1 is found at the tight junctions between the primitive columnar podocytes (arrowheads in A, A′′). In addition, Podocalyxin is localized at the apical membrane, which is recognized as the luminal surface membrane above the tight junctions (A′, A′′). At 40 hpf, the Podocalyxin-localized apical membrane is expanded (B-B′′). After 2 dpf, immunoreactivity for ZO1 is found along the glomerular wall (C, D, and E), which indicates that interdigitation between the podocytes has become extensively. The Podocalyxin signal is found along the entire surface of the podocyte cell bodies (C′-E′, C′′-E′′). (A′′-E′′) show the merged images. N, notochord; arrows, tight junctions in the intestinal epithelium. Bar scale, 10 μm. |
|
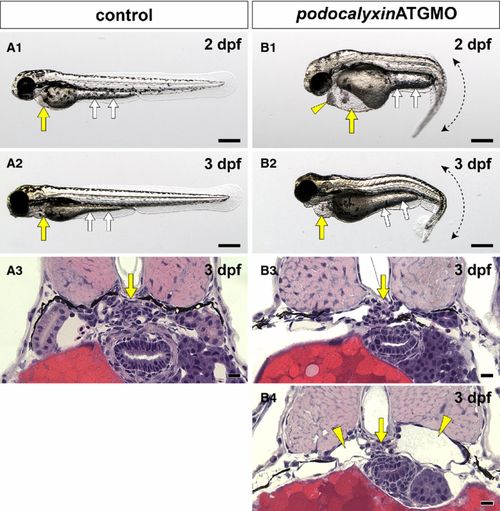
Translational blocking of podocalyxin caused pronephric glomerular hypoplasia. (A1, A2 and B1, B2) Wholebody images. Pericardial sac (yellow arrows) and yolk sac extension (white arrows) are larger in the podxlATGMO morphants on 2 and 3 dpf (B1, B2) than in the uninjected control larvae (A1, A2). This enlargement resulted from edema. The podxlATGMO morphants also have tail curvatures (B1, B2). In the 2-dpf morphants, cellular aggregation is consistently found at the anterior pole of pericardial sac (arrowhead in B1). (A3 and B3, B4) Hematoxylin?eosin staining sections showing pronephric glomerulus. In 3-dpf control larvae, a pair of pronephric nephron primordia merges to form a single glomerulus (arrow in A3). In the podxlATGMO morphants, their pronephric glomeruli exhibit hypoplasia (arrows in B3, B4). Some of the morphants display bilateral cystic dilatation of Bowman′s capsule and pronephric tubule (arrowheads in B4). Bar scales, 250 μm in A1, A2, B1, B2; 10 μm in A3, B3, B4. PHENOTYPE:
|

Translational blocking of podocalyxin caused hypoplasia of the podocyte foot processes. (A1?2) 3-dpf uninjected control embryos. The pronephric glomerulus contains several glomerular capillaries (pink) together with mesangium (brown). Bowman′s capsule (podocytes and parietal epithelial cells) including urinary space is shown in green (A1). Podocytes form interdigitating fine foot processes with slit diaphragm, which exhibited a ?beads-on-a-string? appearance. The cell body of podocyte is not in direct contact with the GBM to form a subpodocyte space under the cell body (A2). (B1?4) 3-dpf podxlATGMO morphants. Glomerular capillaries vary in diameter, and poorly developed mesangium is found within the glomerulus (B1). Unlike in the control larvae, podocyte cell bodies directly adhere to the GBM (B2) and irregularshaped processes covering the GBM (B3) are frequently found in the morphants, although regular foot processes are also formed in some regions (B4). Duplication of the GBM and the interposition of mesangial cell processes between the duplicated GBM are found in some regions (arrowheads in B3). (C) The ratio of the three structures covering the GBM. Cap, glomerular capillary lumen; N, notochord; P, podocyte cell body. Bar scales, 5 μm in A1, B1; 2 μm in B2, 500 nm in A2, B3, B4. PHENOTYPE:
|
|
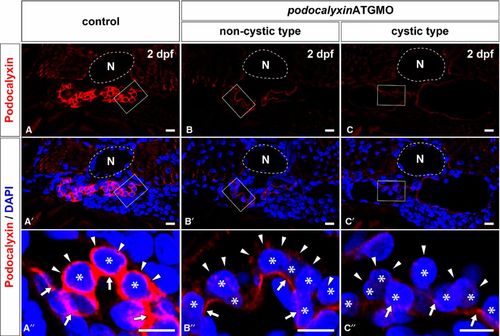
Efficacy for translational blocking of podocalyxin. The expression of Podocalyxin is determined by using immunohistochemistry with a specific antibody. Immunoreactivity for Podocalyxin is remarkably reduced in both the non-cystic type (B, B′) and the cystic type (C, C′) of glomerulus in 2-dpf podxlATGMO morphants in comparison with control (A, A′). In control larva, immunoreactivity for Podocalyxin is recognized at the entire surface of podocyte cell body (arrowheads in A′′). Podocalyxin immunoreactivity surrounding the podocyte cell body is diminished in both the non-cystic and cystic types of glomeruli (arrowheads in B′′, C′′), although a faint signal is found in a linear pattern along the GBM (arrows in B′′, C′′). (A′′?C′′) images are magnification of the regions which are framed by rectangles in the above images. Asterisks indicate the nuclei of podocytes, which are visualized by staining with DAPI. N, notochord. Bar scales, 10 μm in A-C, A′-C′ ; 5 μm in A′′-C′′. EXPRESSION / LABELING:
|
|
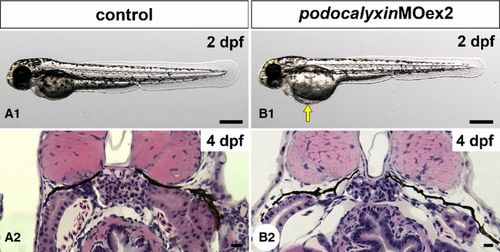
Truncation of podocalyxin exon 2 induces mild pericardial edema. (A1 and B1) Wholebody images of 2-dpf larvae. Approximately 40% of podxlMOex2 morphants have mild pericardial edema (arrow in B1). (A2 and B2) Hematoxylin?eosinstained sections reveal that the pronephric glomerulus in the podxlMOex2 exhibits developed glomerular capillary and mesangium at 4 dpf (B2), as that seen in control (A2). Bar scales: 250 μm in A1, B1; 10 μm in A2, B2. |
|
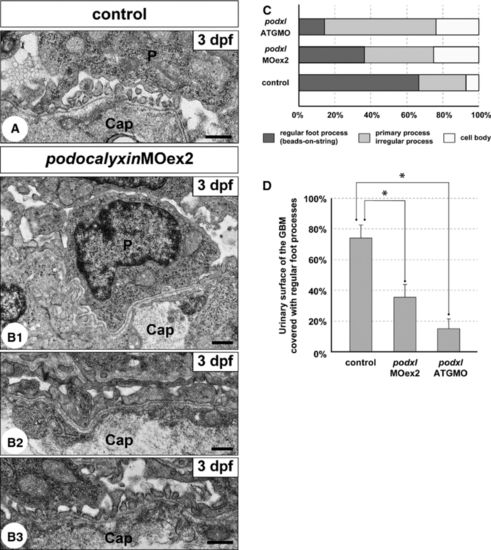
Truncation of podocalyxin exon 2 induces poor development of podocyte foot processes. (A and B1-3) Transmission electron micrographs showing glomerular capillary wall. Unlike control larvae (A), podocyte cell bodies that directly adhere to the GBM (B1) and irregular-shaped processes that cover the GBM (B2) are frequently found in the podxlMOex2 morphants, although regular foot processes with a ?beads-on-a-string? pattern are also formed in some regions (B3). Cap, glomerular capillary lumen; P, podocyte cell body. Bar scales: 500 nm. (C) The ratio of the three structures covering the urinary surface of GBM. (D) Significant reduction in regular foot processes with slit diaphragm in the podxlATGMO and podxlMOex2 morphants. In control larvae, regular foot processes with slit diaphragm cover 66.7 � 7.8% of the urinary surface of the GBM (N = 3). The foot process-covering area is greatly reduced in the podxlMOex2 morphants (36.3 � 5.0%, N = 3) and non-cystic-type podxlATGMO morphants (14.4 � 7.5%, N = 3) with statistical significance. All of the larvae were examined on 3 dpf. *P < 0.01. PHENOTYPE:
|