- Title
-
Zebrafish N-cadherin, encoded by the glass onion locus, plays an essential role in retinal patterning
- Authors
- Malicki, J., Jo, H., and Pujic, Z.
- Source
- Full text @ Dev. Biol.
|
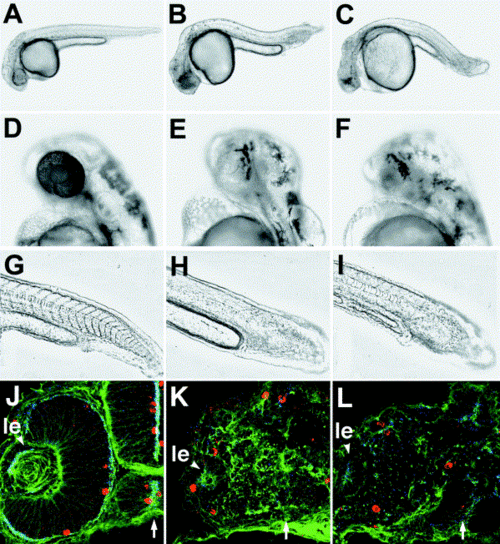
Knockdown phenotype of N-cadherin. Anti-N-cadherin morpholino-modified antisense oligonucleotides reproduce the glass onion phenotype. Gross morphology of the wild type (A), glom117 (B), and anti-N-cadherin morpholino-treated animals (C) at 36 hpf. Eye pigmentation of the wild type (D), glom117 (E), and morpholino-treated animals (F) at 48 hpf. Tail and somites of the wild type (G), glom117 (H), and morpholino-treated animals (I) at 30 hpf. Somite boundaries are poorly defined in both mutant and morpholino-treated embryos. Analysis of cryosections indicates that retinal neuroepithelium is severely disorganized at 28 hpf in morpholino-treated animals (K, L compare with the wild type in J). In (J?L), centrosomes are stained with anti-γ-tubulin antibody (blue), adherens-junction associated actin foci are visualized with Alexa-488-conjugated phalloidin (green), while M- phase nuclei are identified by anti-phosphohistone antibody staining (red). Embryos in (K) and (L) were treated with anti N-cadherin morpholinos 2 and 3, respectively. In (G-I) anterior is right and dorsal is top. ?le? in (J?L) indicates lens. Arrows indicate the midline. |
|
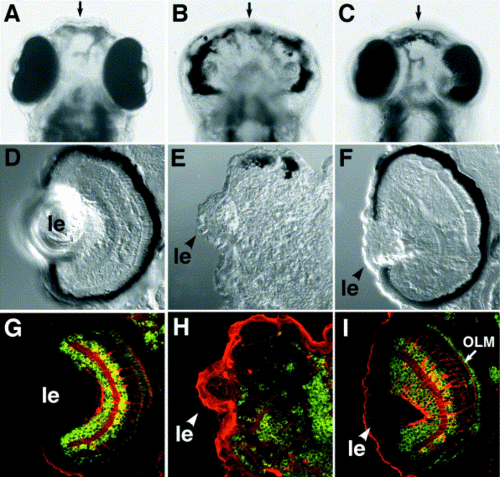
Rescue of the glom117 phenotype by N-cadherin mRNA expression. Eye pigmentation and retinal architecture of the wild type (A, D, G), glom117 (B, E, H), and rescued glom117 embryos (C, F, I) at 4 dpf. (A?C) Ventral views of whole embryos. (D?F) Images of transverse sections through retinae. In (G?I), transverse sections of the retina were costained with anti-Hu (green, stains ganglion and amacrine cells), Zpr-1 (green, stains cone photoreceptor cells), and anti-carbonic anhydrase (red, stains Muller glia) antibodies. Note that the architecture of Muller glia and the outer limiting membrane (OLM, arrow) are restored in rescued animals. For rescued embryos, mutant genotype was confirmed by using a polymorphic marker linked to the N-cadherin locus. In (D?J), ?le? indicates lens, dorsal is up. Arrows in (A?C) indicate the midline. |
|
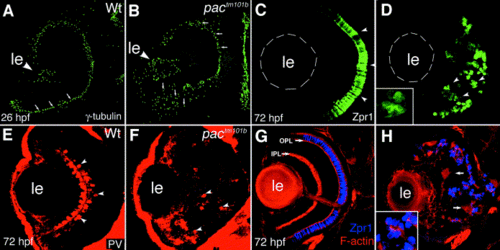
Phenotype of the pactm101b mutant allele. pactm101b produces defects in the retinal neuroepithelium and in the organization of retinal neurons. In wild-type retinal neuroepithelium, all centrosomes are positioned apically at 26 hpf (A). By contrast, the centrosomes of pactm101b mutant retina are scattered along the apicobasal axis of the neuroepithelium in the ventral portion of the eye cup (vertical arrows in B), while the dorsal retina appears unaffected (horizontal arrows in B). At 3 dpf, photoreceptor cells of pactm101b mutant animals are scattered throughout most of the retina (arrowheads in D), while their wild-type counterparts form a uniform layer (arrowheads in C). Ectopic photoreceptors adhere to each other forming clusters, rosettes (inset in D). Parvalbumin-positive amacrine cells localize to ectopic positions in pactm101b retina (arrowheads in F). In wild-type zebrafish larvae, these cells form a layer in the center of the retina (arrowheads in E). Neuronal processes of the zebrafish retina form two uniform layers that strongly stain with phalloidin: outer plexiform layer (OPL) and inner plexiform layer (IPL) (arrows in G). In the retinae of pactm101b/tm101b mutants, neuronal processes form disorganized patches instead of layers (arrows in H). The lens, ?le?, is outlined with dashes in (C) and (D). All panels show transverse cryosections through the retina stained with the following reagents: anti-γ- tubulin antibody (A, B), Zpr-1 antibody (C, D, G, H), anti-parvalbumin antibody (E, F), and phalloidin (G, H). In all panels, dorsal is up. |
|
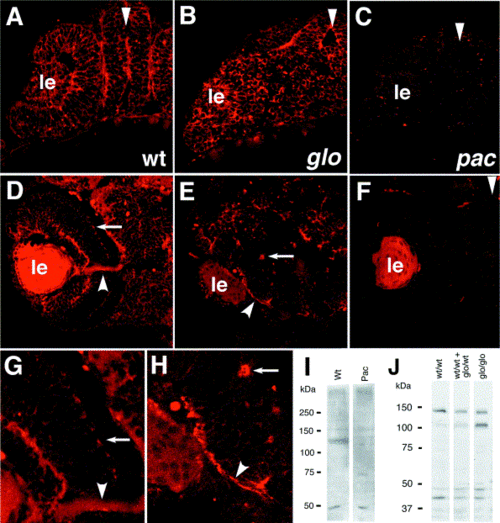
Protein product localization in mutant retinae. At 24 hpf, N-cadherin polypeptide is present in wild-type (A), glom117 (B) but not in pactm101b (C) retinae. This situation persists at later stages of development as shown for the wild-type (D), glom117 mutants (E), and pactm101b mutants (F). Residual staining present in (C) and (F) is most likely due to background fluorescence. As reported previously in wild-type animals, N-cadherin is expressed in plexiform layers (arrows in D and G) and in the optic nerve (arrowheads D in G). The glom117 mutant polypeptide appears to retain this distribution. It is present both in the optic nerve (arrowheads in E and H) and in ectopically localized plexiform patches (arrows in E and H). Western blotting indicates that N-cadherin polypeptide is absent in pactm101b/tm101b mutant embryos (I) but it persists in glom117/m117 animals (J) at 3 dpf. (D, E, G, H) Retinae at 50 hpf. Retina in (F) is at 72 hpf. ?le? indicates lens. Arrowheads in (A?C) indicate the midline. In (A?H) dorsal is up. |
|
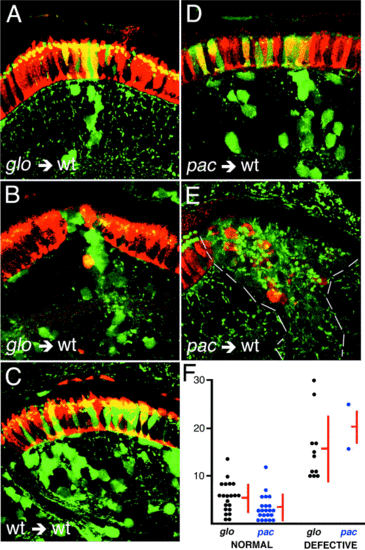
Mosaic analysis of the glass onion phenotype at 3 dpf. The phenotype of glom117/m117 mutant photoreceptor cell clones in the environment of the wild-type retina depends on clone size. In small clones, mutant double cones (yellow) display proper morphology and localize to the photoreceptor cell layer (A). In larger clones, the photoreceptor cell layer is frequently disrupted and ectopic photoreceptors are sometimes observed (B). This is never seen in control experiments when wild-type clones develop in the environment of wild-type retina regardless of clone size (C). When pactm101b mutant clones develop in the environment of the wild-type retina, the photoreceptor cell layer remains normal (D). Rare exceptions to this are seen in the case of particularly large clones (E). The correlation of photoreceptor cell layer abnormalities with clone size is quantitated for mutant to wild-type transplants in (F). Each dot in (F) represents a donor-derived clone that was physically separate from other donor-derived clones. Clone size is plotted on the vertical axis. Larger clones have a clear tendency to cause patterning defects in the retina. Data for glom117 and pactm101b alleles are plotted in separate columns. Donor-derived cell clones are in green, while double cones are stained with the Zpr-1 antibody and are depicted in red. Donor-derived double cones appear yellow. Dashes in (E) outline a large mutant clone in otherwise wild-type retina. |
Reprinted from Developmental Biology, 259(1), Malicki, J., Jo, H., and Pujic, Z., Zebrafish N-cadherin, encoded by the glass onion locus, plays an essential role in retinal patterning, 95-108, Copyright (2003) with permission from Elsevier. Full text @ Dev. Biol.