- Title
-
Fate mapping melanoma persister cells through regression and into recurrent disease in adult zebrafish
- Authors
- Travnickova, J., Muise, S., Wojciechowska, S., Brombin, A., Zeng, Z., Young, A.I.J., Wyatt, C., Patton, E.E.
- Source
- Full text @ Dis. Model. Mech.
|
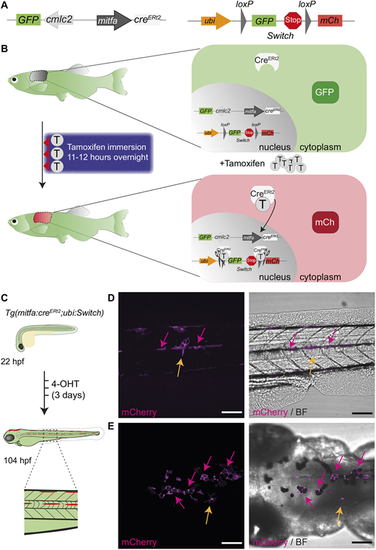
A tamoxifen-inducible mitfa:creERt2/loxP system in zebrafish labels melanocytes. (A) A schematic of mitfa:creERt2 (left) and ubi:Switch (right) (Mosimann et al., 2011) constructs to enable a green-to-red switch in mitfa promoter-expressing cells after tamoxifen treatment. The cardiac myosin light chain 2 (cmlc2; also referred to as myl7) promoter drives GFP expression and facilitates screening for the creERt2 line based on GFP+ heart myocardium. mCh, mCherry. (B) A schematic of the concept behind the colour-switching creERt2/loxP system that allows green-to-red switch after tamoxifen (T) treatment in cells expressing the mitfa promoter driving creERt2. (Left) A three-night course of treatment with tamoxifen for 11-12 h by immersion in the dark with a drug-free period during the day. (Right) The principle of colour switching upon tamoxifen treatment as a result of CreERt2 trans-localisation to the nucleus, causing excision of GFP and a stop codon, enabling mCherry expression in recombined cells. (C) An illustration of the expected green-to-red fluorophore switch in the zebrafish embryo upon three consecutive daily treatments with 20 �M 4-hydroxytamoxifen (4-OHT). Enlarged view of dashed line rectangle shows ?switched? melanocytes in the embryonic stripes. hpf, h post fertilisation. (D,E) Lateral (D) and dorsal (E) views of a zebrafish larva after 4-OHT treatment at 104 hpf (4.5 dpf) validates mitfa:creERt2 specificity to melanocyte lineage. Standard deviation intensity (STD) projection of a confocal z-stack (left, mCherry) and merged with a single z-plane acquisition in brightfield (BF) channel (right, merge) at 6 h after the end of 4-OHT treatment. mCherry-expressing melanocytes are visible and express black melanin pigment (pink arrows). Two unpigmented, star shaped cells expressing mCherry are also visible and may represent xanthophore progenitors (yellow arrows). N=6 fish for each view, scale bars: 100 �m. EXPRESSION / LABELING:
|
|
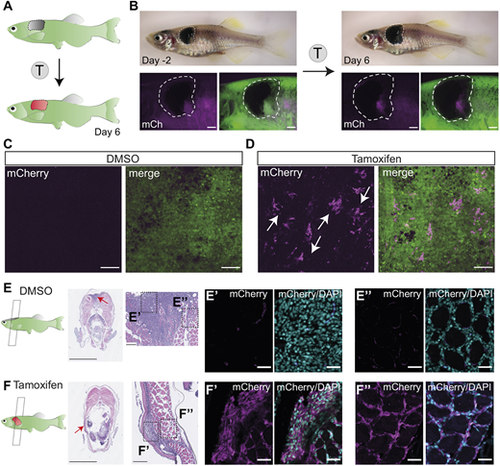
Successful fluorophore switch in adult zebrafish primary melanoma after tamoxifen treatment. (A) An illustration of the expected fluorophore switch in the primary melanoma tumour upon tamoxifen (T) treatment and imaged 6 days after the start of the treatment. (B) An adult zebrafish with a fully developed primary melanoma tumour (delineated with white dashed line) 2 days before the tamoxifen treatment course (left) and 6 days after the start of the treatment (right) showing de novo expression of mCherry protein in pigmented trunk tumour after tamoxifen treatment (4 �M). N=2 fish, scale bars: 1 mm. (C,D) STD projections of confocal z-stacks of DMSO (0.04%)-treated fish (C, left mCherry, right merged with GFP) and tamoxifen-treated fish (D, left mCherry, right merge) show small clusters of mCherry-expressing cells in the tamoxifen-treated group only (white arrows). N=2 fish, scale bars: 100 �m. (E,F) Paraformaldehyde (PFA)-fixed paraffin-embedded (FFPE) transverse sections of primary melanoma fish (left) stained with Haematoxylin and Eosin, with zoomed images of the tumour (red arrows pointing to the location of the zoomed images). N=2 fish, scale bars: 2.5 mm and 100 �m, respectively. Dashed line boxes indicate area of section shown in E?,F? and E?,F?. (E?,F?) Immunofluorescence staining of bulk tumour with mCherry antibody (magenta), counterstained with the nuclear marker DAPI (cyan), shows mCherry signal in tamoxifen-treated samples only (F?) compared to DMSO control (E?). N=2 fish, scale bars: 25 �m. (E?,F?) Immunofluorescence staining of invasive tumour shows successful fluorophore switching and mCherry expression in the tamoxifen-treated group only (F?) compared to DMSO control (E?). N=2 fish, scale bars: 25 �m. See also Fig. S1A. |
|
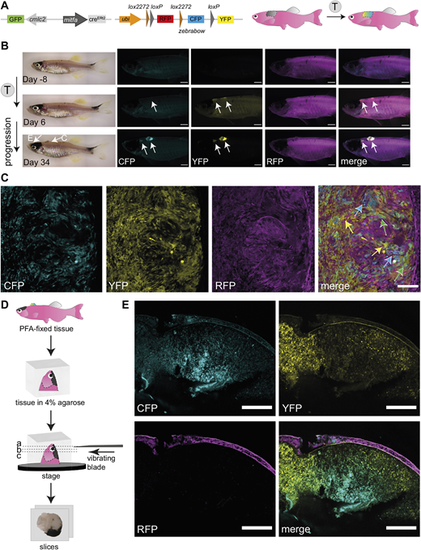
Successful fluorophore switch using the zebrabow system in adult zebrafish primary melanoma following tamoxifen treatment. (A) A schematic of mitfa:creERt2 and ubi:zebrabow constructs (Pan et al., 2013), which enables recombination from red signal (RFP) to a stochastic combination of cyan (CFP), yellow (YFP) and red (RFP) signal in mitfa:creERt2-expressing cells after tamoxifen (T) treatment. The cmlc2 promoter drives GFP expression and facilitates screening for the creERt2 line based on GFP+ heart myocardium. (B) Multicolour labelling in adult zebrafish melanomas. An adult zebrafish with two primary melanomas 8 days before the tamoxifen treatment course (top), and 6 days (middle) and 34 days (bottom) after the start of the treatment. Increasing de novo expression of CFP and YFP proteins in both pigmented and unpigmented tumours can be detected after tamoxifen treatments (4 �M). N=3 fish, scale bars: 1 mm, white arrows point to two tumour locations. The arrows on the brightfield image (Day 34) point to the melanomas shown in panels C and E, respectively. (C) Confocal multicolour imaging of zebrafish melanoma. Single z-plane of confocal acquisition of tamoxifen-treated fish shows individual cells acquiring varied combinations of CFP, YFP and RFP (blue, green and yellow arrows on merged image, respectively). N=3 fish, scale bar: 100 �m. (D) Overview of the vibratome sectioning protocol. The PFA-fixed tissue is mounted in 4% agarose and cut using a vibrating blade into 400 �m-thick sections to capture the pigmented tumour (E). (E) Vibratome section imaging of a tamoxifen-treated fish (tissue location as shown in B). Clusters of CFP, YFP or double-expressing cells are clearly visible in areas of the pigmented tumour. STD projections of confocal z-stacks of a PFA-fixed sectioned tissue. N=3 fish, scale bars: 200 �m. See also Fig. S1B-D. |
|
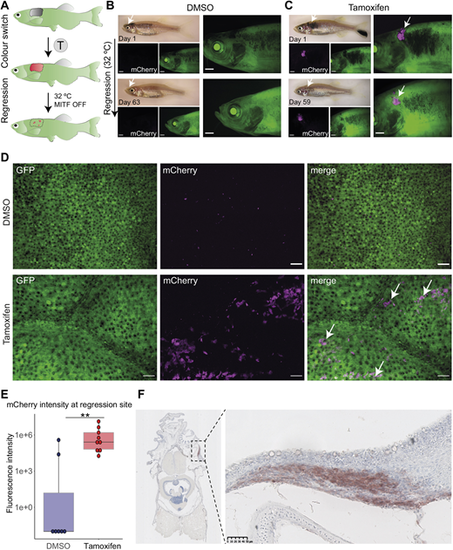
Persister cells originate from the primary tumour. (A) The experimental workflow of tamoxifen (T) treatment and melanoma regression at the higher water temperature (32�C, MITF OFF). At the higher temperature, mitfa RNA is still expressed but is not spliced correctly and therefore MITF activity is abolished. Loss of MITF activity leads to tumour regression with remaining persister cells at the minimal residual disease (MRD) site. (B) A fish treated with vehicle (0.05% DMSO) showing no green-to-red recombination in the primary tumour (top) or after regression (bottom). Scale bars: 1 mm, white arrows point at the tumour site, N=4 fish. (C) A fish treated with tamoxifen (5 �M) showing expression of mCherry+ cells in a primary tumour (magenta, white arrows) that are still detected 59 days after initiation of melanoma regression (bottom, white arrows). Scale bars: 1 mm, N=7 fish. (D) STD projections of confocal z-stack acquisitions of regression sites of fish treated with DMSO or tamoxifen. White arrows point to mCherry+ cells (magenta) only present in the tamoxifen-treated condition. Scale bars: 50 ?m; DMSO, N=4 fish; tamoxifen, N=7 fish. (E) Box plot of fluorescence intensity quantification of mCherry signal using average intensity projections of confocal images of residual disease. Lines in boxes indicate the medians, hinges correspond to 25th and 75th percentiles, and whiskers indicate data within 1.5 interquartile range of the upper and lower quartiles. DMSO, N=4 fish with seven regression sites; tamoxifen, N=7 fish with nine regression sites; **P<0.01, Wilcoxon test. (F) Immunohistochemistry of the MRD site. FFPE transverse section stained with anti-mCherry antibody reveals ?switched? cells at the MRD site. Right panel shows an enlarged view of MRD, outlined by the black dashed line rectangle (left panel). Signal in red (AEC substrate) counterstained with nuclear marker Haematoxylin (blue). Scale bar: 50 �m, N=3 fish. |
|
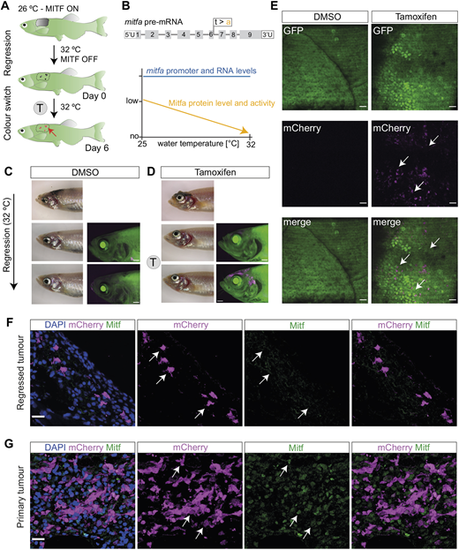
Successful GFP-to-mCherry fluorophore switch in persister cells. (A) Schematic representation of the experimental workflow of tamoxifen (T) treatment following melanoma regression. The red arrow points to the location of recombined melanoma persister cells at MRD site. (B) Overview of mitfavc7 mutant controlling Mitfa activity. (Top) Schematic of mitfa pre-RNA with the location of vc7 mutation at the end of exon 6. (Bottom) Graphical overview of the effect of mitfavc7 mutation on Mitfa protein level and activity (orange) at increasing water temperatures. Mitfa protein and activity levels decrease with increasing water temperature, unlike the mitfa RNA levels, which remain expressed (blue). Schematic adapted from Travnickova et al. (2019). This image is not published under the terms of the CC-BY license of this article. For permission to reuse, please see Travnickova et al. (2019). (C) A representative image of a control fish with no mCherry+ cells detectable at the MRD site without tamoxifen treatment (top). Following melanoma regression and subsequent treatment with DMSO (0.04% at 31-32�C) there is no green-to-red recombination in the melanoma MRD (bottom row) compared to pre-treatment (middle row). Scale bars: 1 mm, N=3 fish. (D) A representative image of a tamoxifen-treated fish with newly ?switched? mCherry+ cells (magenta) detectable at the MRD site (top). Following melanoma regression and subsequent treatment with tamoxifen (4 �M, at 31-32�C) there is specific green-to-red recombination in the melanoma MRD (bottom row) compared to pre-treatment (middle row). Scale bars: 1 mm, N=3 fish. (E) STD projections of confocal z-stack acquisitions showing MRD sites in fish treated with DMSO or tamoxifen. White arrows indicate clusters of mCherry+ cells present only in tamoxifen-treated condition. Scale bars: 50 �m, N=3 fish. (F) Melanoma cells at the MRD site express mCherry, but lack Mitfa protein (white arrows). STD projections of confocal z-stack acquisitions of immunofluorescence staining of the tamoxifen-treated MRD with antibodies for mCherry and Mitfa proteins and with DAPI nuclear staining. Scale bar: 15 �m. (G) Melanoma cells in the primary tumour express both mCherry and Mitfa protein (white arrows). STD projections of confocal z-stack acquisitions of immunofluorescence staining of the tamoxifen-treated primary tumour, showing staining of mCherry and Mitfa proteins with DAPI nuclear staining Scale bar: 15 �m. See also Fig. S2. |
|
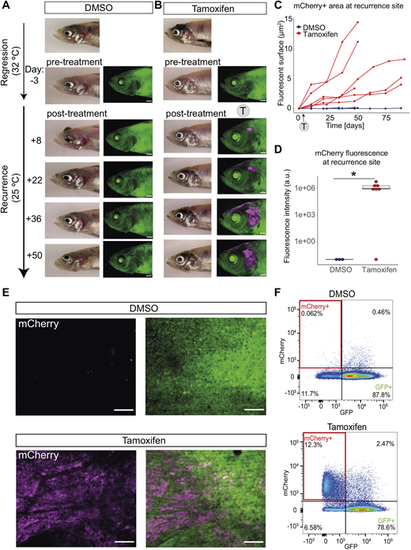
Persister cells directly contribute to melanoma recurrent disease. (A) Representative images of control fish with no mCherry fluorescence. A fish with a regressed melanoma (top image, primary tumour; below, regressed tumour) treated with vehicle (0.04-0.05% DMSO at 32�C) showing no green-to-red fluorophore switch in the melanoma MRD compared to the pre-treatment image. Follow-up brightfield and fluorescent acquisitions (every 14 days) show progression of tumour recurrence over time with no appearance or increase in mCherry fluorescence. Scale bars: 1 mm, N=5 fish (sum of two independent experiments). (B) mCherry+ persister cells from MRD are present through to recurrence. A fish with a regressed melanoma (top image, primary tumour; below, regressed tumour) treated with 4-5 �M tamoxifen (at 32�C) showing green-to-red fluorophore switch (in magenta) in the melanoma MRD. Follow-up brightfield and fluorescent acquisitions (every 14 days) show progression of tumour recurrence over time with increasing mCherry signal. Scale bars: 1 mm, N=7 fish (sum of two independent experiments). (C) mCherry+ persister cells increase over time. Line plot of area quantification of mCherry signal using fluorescent acquisitions over time during melanoma recurrence. DMSO (blue), N=5 fish; tamoxifen-treated group (red), N=6 fish (sum of two independent experiments for both groups). Trajectories of melanoma growth represent individual fish. (D) Box plot of fluorescence intensity quantification of mCherry signal of recurrent disease that has grown from MRD treated with tamoxifen. Intensity was measured using average intensity projections of confocal images of recurrence sites comparing DMSO to tamoxifen condition. Lines in boxes indicate the medians, hinges correspond to 25th and 75th percentiles, and whiskers indicate data within 1.5 interquartile range of the upper and lower quartiles. Dots beyond end of whiskers represent outliers. DMSO, N=3 fish; tamoxifen-treated group, N=4 fish with seven recurred melanomas; *P<0.05, Wilcoxon test. Outlier sample in tamoxifen treatment group is likely to be false negative as mCherry fluorescence was later confirmed using flow cytometry in this highly pigmented sample. a.u., arbitrary units. (E) mCherry is present only in recurrent disease that has grown from MRD treated with tamoxifen. STD projections of z-stack confocal acquisition show recurred melanoma sites of fish treated with vehicle (DMSO, top row) or tamoxifen (bottom row) at the MRD stage. Scale bars: 100 �m. (F) mCherry+ cells in recurrent disease are detectable by fluorescence-activated cell sorting. A representative pseudocolour flow cytometry plot of a recurred melanoma lesion from DMSO-treated fish and tamoxifen-treated fish. mCherry+ cells highlighted in red rectangles (12.3% versus 0.062%). DMSO, N= 4 fish; tamoxifen, N=5 fish (sum of two independent experiments). |
|
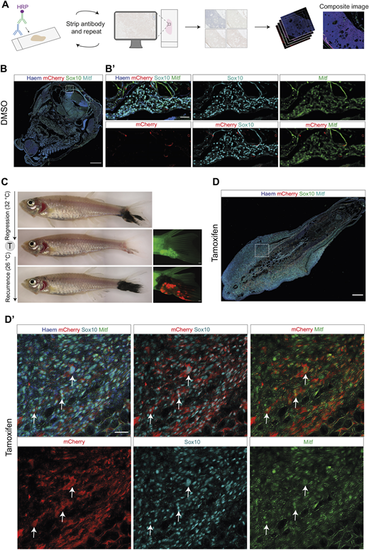
MITF-independent persister cells regain Mitfa protein expression in recurrent disease. (A) Overview of the multiplex immunohistochemistry protocol. The antibody staining is repeatedly imaged and stripped, and images are pseudocoloured and stacked on ImageJ to generate a composite image. HRP, horseradish peroxidase. (B) DMSO-treated melanoma recurrent disease on top of the head, labelled with Sox10, mCherry and Mitfa antibodies overlaid with Haematoxylin counterstain; white dashed line box indicates area of section shown in B?. Scale bar: 250 ?m. (B?) Enlarged views of DMSO-treated tumour section shown in B, showing lack of mCherry staining in melanoma cells positive for Sox10 and Mitfa. Scale bar: 50 ?m. Observable mCherry signal represents trapped antibody in loose tissue fragments. (C) (Left, top to bottom) Brightfield images of melanoma tail fin tumour through growth, regression and recurrence. (Right, middle and bottom) Fluorescent images of regressed and recurred melanoma tail fin tumour, showing mCherry fluorescence in the recurred tumour following tamoxifen-mediated recombination in the MRD. Scale bars: 500 ?m. (D) Recurrent disease from MITF-independent melanoma persister cells regain Mitfa protein expression. Overview of tamoxifen-treated melanoma recurrent disease in the tail fin, showing Sox10, mCherry and Mitfa protein staining overlaid with Haematoxylin counterstain; white dashed line box indicates area of section shown in D?. Scale bar: 250 ?m. (D?) Enlarged views of tumour section shown in D, showing individual staining of Sox10, Mitfa and mCherry proteins. Scale bar: 25 ?m. White arrows highlight cells positive for Sox10, Mitfa and mCherry staining. See also Fig. S3. |