|
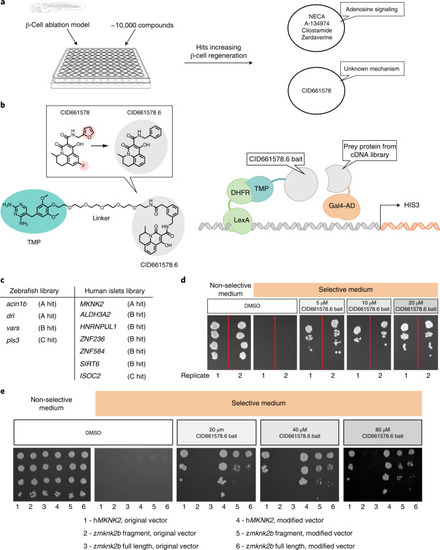
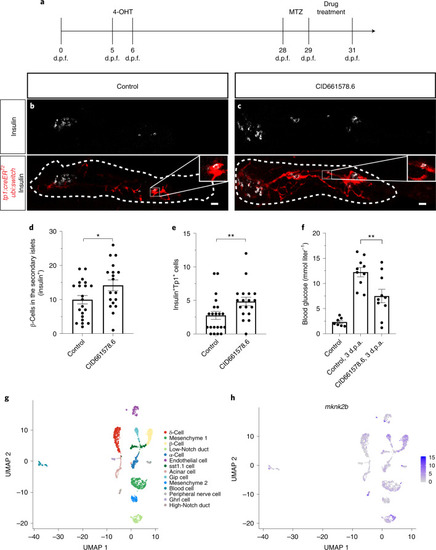
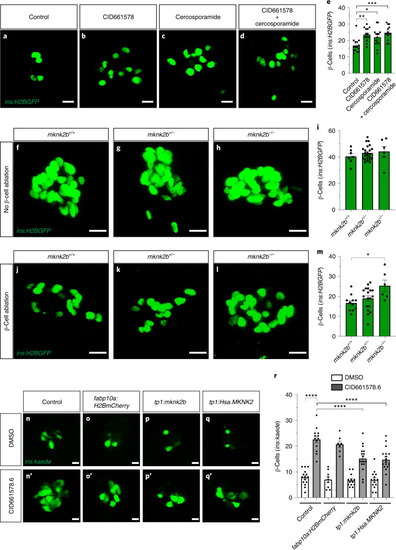
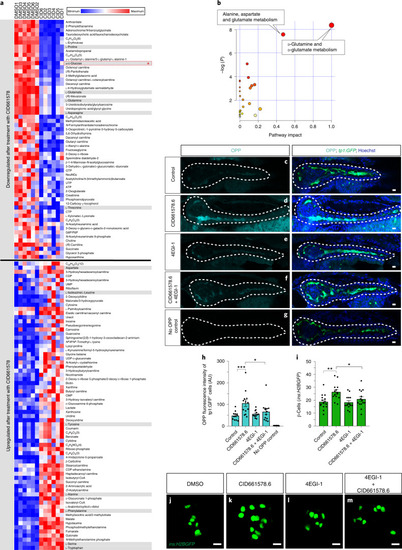
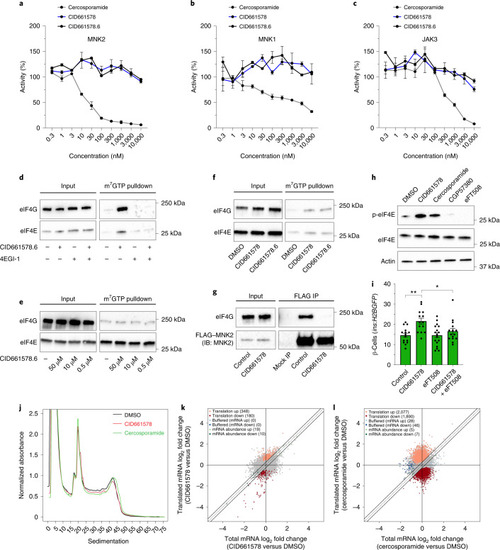
CID661578 increases the interaction between eIF4G and eIF4E and leads to translational changes, without affecting the kinase activity of MNK2.a–c, Dose–response of CID661578, CID661578.6 or cercosporamide on MNK2 (a), MNK1 (b) and JAK3 (c) kinase activity in vitro; n = 2 for each concentration tested. Data are presented as mean values ± s.e.m. d, Immunoblotting against eIF4G and eIF4E after an m7GTP pulldown assay in lysates of COLO 320HSR cells after 6-h treatment with DMSO, CID661578.6, 4EGI-1 or CID661578.6 together with 4EGI-1. For a loading control, 5% of the input was used. e, Immunoblotting against eIF4G and eIF4E after an m7GTP pulldown assay in rabbit reticulocytes treated with the indicated concentrations of CID661578.6. f, Immunoblotting against eIF4G and eIF4E after an m7GTP pulldown assay in lysates of PANC-1 cells treated with DMSO, CID661578 or CID661578.6 for 6 h. For a loading control, 1% of the input was used. g, Immunoblotting against eIF4G and FLAG–MNK2 after an immunoprecipitation (IP) assay with anti-FLAG in lysates of PANC-1 cells that were treated for 6 h with DMSO or CID661578. For a loading control, 1% of the input was used; IB, immunoblot. h, Immunoblotting against phospho-eIF4E (Ser 209; p-eIF4E), total eIF4E and actin in lysates of PANC-1 cells after 6-h treatment with DMSO, CID661578, cercosporamide, CGP57380 or eFT508. i, Quantification of the number of β-cells in 6 d.p.f. zebrafish larvae following β-cell ablation and treatment for 48 h with DMSO, CID661578, eFT508 or a combination of CID661578 and eFT508; n = 15 (control), n = 14 (CID661578), n = 17 (eFT508) and n = 15 (CID661578.6 + eFT508). A one-way ANOVA followed by Dunnett’s multiple comparisons test was used to assess significance for i (**P = 0.0014 (control versus CID661578) and *P = 0.0283 (CID661578 versus CID661579 + eFT508)). Data are presented as mean values ± s.e.m. Experiments in d–h were repeated at least two times. j, Representative polysome tracings from optimized sucrose gradients of PANC-1 cells treated with DMSO, CID661578 or cercosporamide. k,l, Scatter plots showing log2 fold changes for total mRNA (x axis) and polysome-associated mRNA (y axis) for the comparisons of CID661578 (k) and cercosporamide (l) to DMSO. Color codes indicate significantly affected mRNAs identified by anota2seq analysis. Source data
|