- Title
-
Strip1 regulates retinal ganglion cell survival by suppressing Jun-mediated apoptosis to promote retinal neural circuit formation
- Authors
- Ahmed, M., Kojima, Y., Masai, I.
- Source
- Full text @ Elife
|
(A) Zebrafish retinal neural circuit showing retinal neurons and synaptic layers. GCL, ganglion cell layer; IPL, inner plexiform layer; INL, inner nuclear layer; OPL, outer plexiform layer; ONL, outer nuclear layer; RGCs, retinal ganglion cells; ACs, amacrine cells; BPs, bipolar cells; HCs, horizontal cells; PRs, photoreceptors; MGs, M�ller glia. (B) Morphology of wild-type and rw147 embryos at 4 dpf. Dotted lines demarcate the eye. An arrowhead indicates abnormal lower jaw. An asterisk indicates heart edema. (C) Wild-type and rw147 mutant retinas at 4 dpf. Red and yellow arrowheads indicate the IPL and OPL, respectively. (D) A missense mutation occurs in strip1 gene of rw147 mutants leading to replacement of Leu195 with arginine. (E) Wild-type retinas labeled with anti-Strip1 antibody (upper panels) and anti-Strip1 plus Strip1-blocking peptide as a negative control (lower panels). Nuclei are stained with Hoechst. Scale bar, 50 ?m. Right panels show higher magnification of outlined areas. Scale bar, 10 ?m. (F) Whole-mount labeling of 3-dpf wild-type and strip1rw147 mutant retinas with anti-acetylated ?-tubulin antibody. Bottom panels show higher magnification of outlined areas. Scale bar, 50 ?m. (G) Projection images of single RGCs at 2 dpf expressing ath5:Gal4VP16; UAS:MYFP in wild-type and strip1rw147 mutants. Scale bar, 10 ?m. (H) Projection images of single ACs at 3 dpf expressing ptf1a:GFP in wild-type and strip1rw147 mutants. Scale bar, 10 ?m. (I) Projection images of single BPs at 3 dpf expressing nyx:Gal4VP16; UAS:MYFP in wild-type and strip1rw147 mutants. Scale bar, 10 ?m.
|
|
(A) CRISPR-mediated, targeted mutagenesis of zebrafish strip1 gene. A 10-bp deletion was introduced into exon 9, leading to a premature stop codon in strip1 gene. (B) Morphology of wild-type and strip1crispr?10 embryos at 4 dpf. Dotted lines demarcate the eye outline. The arrowhead indicates an abnormal lower jaw. An asterisk indicates heart edema. (C) Plastic sections of wild-type and strip1crispr?10 mutant retinas stained with toluidine blue at 4 dpf. (D) A polyacrylamide gel image showing PCR amplicons using primers that flank the 10-base deletion of strip1crispr?10. Wild-type and strip1crispr?10 mutant bands are expected to appear at 279 and 269 bps, respectively. Different band sizes were consistently obtained from progeny produced by pairwise crosses of strip1crispr?10 heterozygous mutant fish. (E) Morphology of non-injected and MO-strip1-injected wild-type embryos at 4 dpf. Dotted lines outline the eye. An arrowhead indicates the abnormal lower jaw. An asterisk indicates heart edema. (F) Retinal plastic sections of non-injected and MO-strip1-injected wild-type embryos at 4 dpf. (G) Western blotting of 2-dpf head lysates from non-injected and MO-strip1-injected wild-type embryos. Red and black arrows show expected band sizes for zebrafish Strip1 (93 kDa) and the loading control ?-actin (42 kDa), respectively. (H) Plastic sections of 4 dpf wild-type and strip1rw147 mutant retinas without transgene (top panels), and with the transgenes Tg[hsp:WT.Strip1-GFP] (middle panels) or Tg[hsp:Mut.Strip1-GFP] (bottom panels) post heat shock treatment. Overexpression of Green Fluorescent Protein (GFP)-tagged wild-type Strip1 rescues retinal defects in strip1rw147 mutants, while the GFP-tagged mutant form fails to rescue. (I) A staggered bar graph, which represents the percentage of strip1rw147 mutants with a defective retina vs. mutants with a rescued retina in strip1rw147 mutants without transgenes, and with Tg[hsp:WT.Strip1-GFP] or Tg [hsp:Mut.Strip1-GFP].
|
|
( |
|
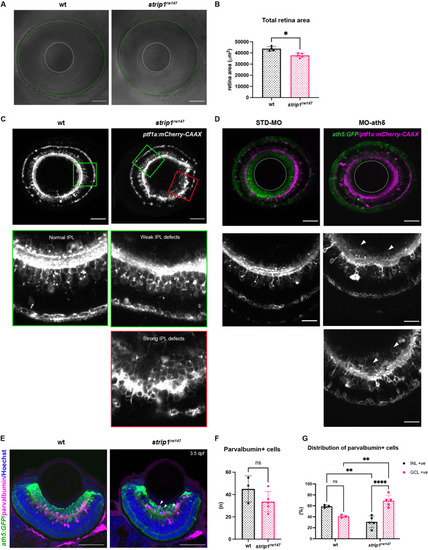
(A) Confocal sections of wild-type and strip1rw147 mutant retinas combined with the transgenic line Tg[ath5:GFP; ptf1a:mCherry-CAAX] to label RGCs and amacrine cells (ACs). Middle panels represent higher magnification. Lower panels show the magenta channel. Arrowheads indicate abnormal positioning of ptf1a+ ACs in the GCL. Asterisks show AC dendritic patterning defects. INL, inner nuclear layer; GCL, retinal ganglion cell layer. Scale bars, 50 ?m (upper panels) and 10 ?m (middle and lower panels). (B) Percentage of ath5+ area relative to total retinal area. Student?s t-test with Welch?s correction, n ? 4. (C) AC numbers per unified retinal area (8500 ?m2). Student?s t-test with Welch?s correction, n ? 3. (D) Distribution of ACs (GCL or INL) per unified retinal area (8500 ?m2). Two-way analysis of variance (ANOVA) with the Tukey multiple comparison test, n ? 3. (E) Wild-type and strip1rw147 mutant retinas at 3 dpf labeled with anti-Pax6 antibody which strongly labels ACs. Arrows indicate strong Pax6+ cells that infiltrate the GCL. Nuclei are stained with TOPRO3. Scale bar, 50 ?m. (F) The number of strong Pax6+ cells per retina. Student?s t-test with Welch?s correction, n = 5. (G) Percentage of strong Pax6+ cells (GCL+ or INL+) to the total number of strong Pax6+ cells. Two-way ANOVA with the Tukey multiple comparison test, n = 5. (H) Wild-type and strip1rw147 mutant retinas at 3 dpf labeled with anti-Prox1 antibody. Arrows indicate Prox1+ cells that infiltrate the GCL. Nuclei are stained with TOPRO3. Scale bar, 50 ?m. (I) The number of Prox1+ cells per retina. Student?s t-test with Welch?s correction, n = 5. (J) Percentage of Prox1+ cells (GCL+ or INL+) to the total number of Prox1+ cells. Two-way ANOVA with the Tukey multiple comparison test, n = 5. For all graphs, data are represented as means?�?standard deviation (SD). ns, not significant, *p < 0.05, **p?< 0.01, and ****p?< 0.0001.
|
|
( |
|
Labeling of wild-type and |
|
(A) Transferase dUTP nick end labeling (TUNEL) of wild-type and strip1rw147 mutant retinas carrying the transgene Tg[ath5:GFP] to label RGCs. Nuclei are stained with Hoechst. Scale bar, 50 ?m. (B) The number of TUNEL+ cells in ganglion cell layer (GCL). Two-way analysis of variance (ANOVA) with the Tukey multiple comparison test, n ? 3. (C) Percentage of ath5+ area relative to total retinal area. Two-way ANOVA with the Tukey multiple comparison test, n ? 3. (D) Cell transplantation design to evaluate the cell autonomy of Strip1 in RGC survival. Donor embryos from a strip1rw147 mutant background are labeled with dextran rhodamine and transplanted into host wild-type embryos. Hosts that show transplanted retinal columns at 60 hpf were subjected to TUNEL. (E) 60-hpf host retinas stained with TUNEL FL to visualize apoptotic cells in wild type to wild type (upper panel) or strip1rw147 mutant to wild type (lower panel). Arrows indicate the presence of apoptotic donor cells. Scale bar, 10 ?m. (F) Percentage of TUNEL+ donor RGCs relative to total donor RGCs. Mann?Whitney U-test, n = 4. For all graphs, data are represented as means?� SD. *p?< 0.05, **p?<?0.01, and ****p?< 0.0001.
|
|
( |
|
( |
|
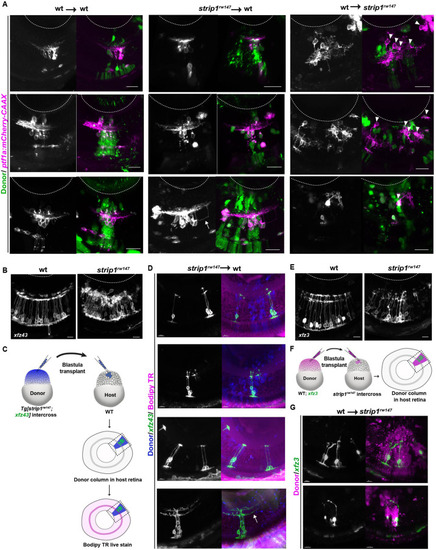
(A) Time-lapse imaging of wild-type and strip1rw147 mutant retinas combined with the transgenic line Tg[ath5:GFP; ptf1a:mCherry-CAAX] to track ACs and RGCs during IPL formation. Asterisks denote empty areas in the ganglion cell layer (GCL). Arrowheads represent infiltration of ACs into empty spaces in the GCL. Panels on the right show higher magnification of outlined areas. Scale bar, 50 ?m. (B) Cell transplantation design to evaluate the cell autonomy of Strip1 in AC-mediated IPL formation. Donor embryos are from intercross of strip1rw147 heterozygous fish combined with Tg[ptf1a:mCherry-CAAX] to label ACs. Host embryos are generated by nontransgenic intercross of strip1rw147 heterozygous fish. Donor cells are labeled with dextran Alexa-488 and transplanted into host embryos to make chimeric host retinas with donor-derived retinal columns. (C) Confocal images of four combinations of transplantation outcomes: wild type to wild type, wild type to mutant, mutant to wild type, and mutant to mutant. Arrowheads indicate abnormal positioning of ACs in basal side of IPL. Scale bar, 20 ?m. (D) Percentage of ACs (either at the apical or the basal side of the IPL) relative to the total number of ACs within a transplanted column. Two-way analysis of variance (ANOVA) with the Tukey multiple comparison test, n ? 4. Data are represented as means �?standard deviation (SD). **p?<?0.01 and ****p?< 0.0001.
|
|
( |
|
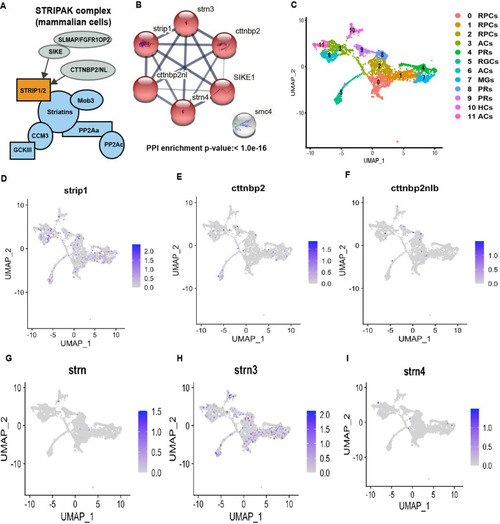
(A) Design of co-immunoprecipitation coupled with mass spectrometry (Co-IP/MS) to identify zebrafish Strip1-interacting partners. Embryos carrying the transgenes Tg[hsp:WT.Strip1-GFP], Tg[hsp:Mut.Strip1-GFP], or Tg[hsp:Gal4;UAS:GFP] were used to pull-down wild-type GFP-tagged Strip1, mutant GFP-tagged Strip1 or only GFP, respectively. Head lysates from 2-dpf zebrafish embryos were subjected to immunoprecipitation using GFP-Trap beads. Immunoprecipitates were digested and analyzed by mass spectrometry (MS). (B) Western blotting of whole head lysates (input) and immunoprecipitates (IP) using anti-GFP antibody. Red and black arrowheads indicate the expected band sizes for Strip1-GFP (120 kDa) and GFP (26 kDa), respectively. (C) Venn diagram comparing proteins significantly enriched in WT.Strip1-GFP relative to Control GFP (blue) and WT.Strip1-GFP relative to Mut.Strip1-GFP (magenta). Six proteins are commonly enriched in both groups, FC >2, p ?<? 0.05. n = 3 for WT.Strip1-GFP and Mut. Strip1-GFP and n = 2 for GFP-control. (D) Five components of the STRIPAK complex found from six proteins commonly enriched in (C). (E) Transferase dUTP nick end labeling (TUNEL) of 60-hpf retinas of Tg[ath5:GFP] transgenic embryos injected with standard MO and MO-strn3. RGCs and apoptotic cells are labeled with ath5:GFP and TUNEL, respectively. Nuclei are stained with Hoechst (blue). (F) The number of TUNEL+ cells in ganglion cell layer (GCL). Mann?Whitney U-test, n ? 6. (G) Percentage of ath5+ area relative to total retinal area. Student?s t-test with Welch?s correction, n ? 3.(H) Confocal images of retinas of 76-hpf Tg[ath5:GFP; ptf1a:mCherry-CAAX] transgenic embryos injected with standard MO and MO-strn3. ath5:GFP and ptf1a:mCherry-CAAX label RGCs and amacrine cells (ACs), respectively. (I) Percentage of ath5+ area relative to total retinal area. Student?s t-test with Welch?s correction, n = 8. Scale bar, 50 ?m (E, H). For all graphs, data are represented as means?� standard deviation (SD). **p < 0.01, ***p?<?0.001, and ****p <?0.0001.
|
|
( |
|
(A) Western blotting of 2-dpf head lysates from STD-MO-injected and MO-strn3-injected wild-type embryos. A ~90 kDa band corresponding to zebrafish Strn3 is reduced in the morphants. (B) Quantification of Strn3 protein level relative to loading control protein, ?-actin. Mann?Whitney U-test, n = 4. Data are represented as means � standard deviation (SD). *p?<?0.05.
|
|
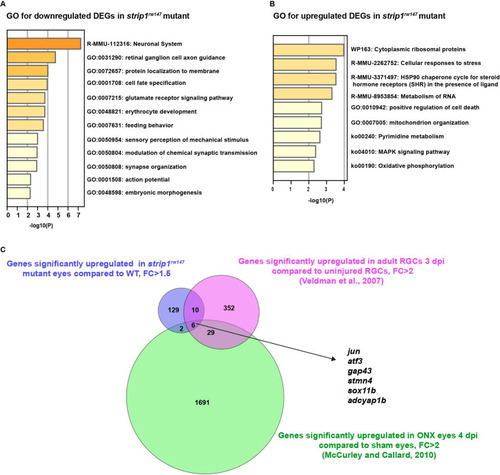
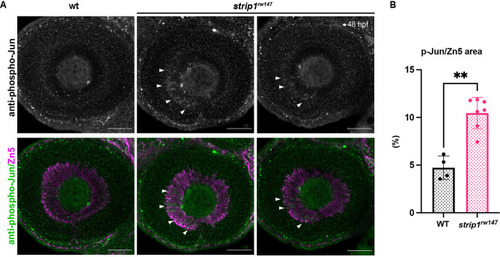
(A) Volcano plot showing differentially expressed genes (DEGs) in strip1rw147 mutants compared to wild-type siblings. Colored points represent genes that are significantly upregulated (131 genes, red) or downregulated (75 genes, blue). Data are obtained from four independent collections of 62-hpf embryo eye cups. FDR < 0.05, log2 FC > |1|. (B) Heatmap of expression values (z-score) representing selected DEGs in strip1rw147 mutants compared to wild-type siblings. (C) Whole-mount labeling of 54-hpf wild-type and strip1rw147 mutant retinas with anti-phospho-Jun antibody and zn5 antibody, which label active Jun and RGCs, respectively. (D) Percentage of phospho-Jun area relative to zn5 area at 54?58 hpf. Student?s t-test with Welch?s correction, n = 6. (E) Whole-mount labeling of 49-hpf wild-type embryos injected with standard MO or MO-strn3 with anti-phospho-Jun antibody and zn5 antibody, respectively. (F) Percentage of phospho-Jun area relative to zn5 area at 49 hpf. Student?s t-test with Welch?s correction, n = 5. (G) Transferase dUTP nick end labeling (TUNEL) and zn5 antibody labeling of 60-hpf wild-type and strip1rw147 mutant retinas injected with standard MO and MO-Jun. Nuclei are stained with Hoechst. (H) The number of TUNEL+ cells in GCL per retina. Two-way analysis of variance (ANOVA) with the Tukey multiple comparison test, n = 6. (I) Confocal images of 76-hpf wild-type and strip1rw147 mutant retinas injected with standard-MO and MO-Jun. Embryos carry the transgene Tg[ath5:GFP] to label RGCs and are stained with bodipy TR methyl ester to visualize retinal layers. (J) Percentage of ath5+ area relative to total retinal area. Two-way ANOVA with the Tukey multiple comparison test, n ? 3. Scale bar, 50 ?m (C, E, G, I). For all graphs, data are represented as means �?SD. ns, not significant, *p?< 0.05, **p?< 0.01, and ****p?< 0.0001.
|
|
List of enriched GO terms in DEGs downregulated ( |
|
( |
|
( |
|
( |