- Title
-
Temporal Vestibular Deficits in synaptojanin 1 (synj1) Mutants
- Authors
- Gao, Y., Nicolson, T.
- Source
- Full text @ Front. Mol. Neurosci.
|
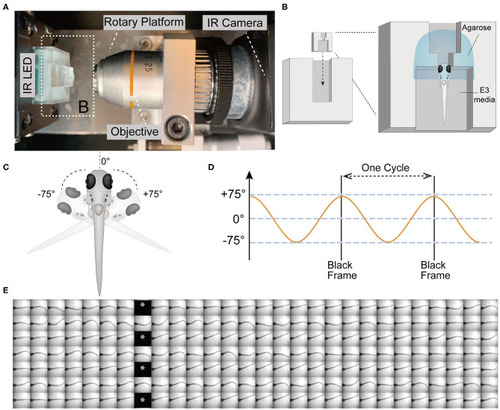
The VSR in zebrafish larvae at 5 days post-fertilization (5 dpf). |
|
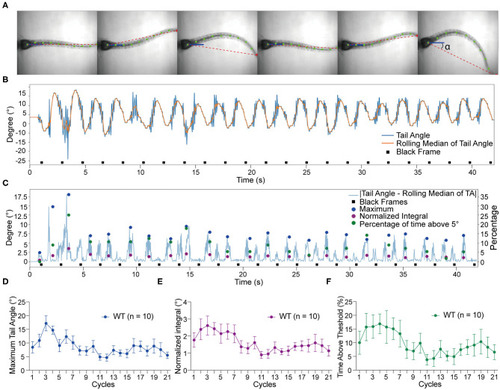
Tracking of tail movements using ZebraZoom software. |
|
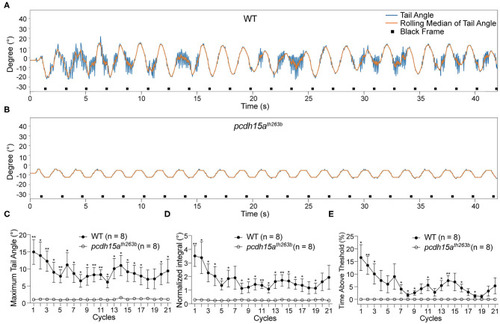
The VSR is absent in PHENOTYPE:
|
|
The larval lateral line organ does not contribute to the VSR. PHENOTYPE:
|
|
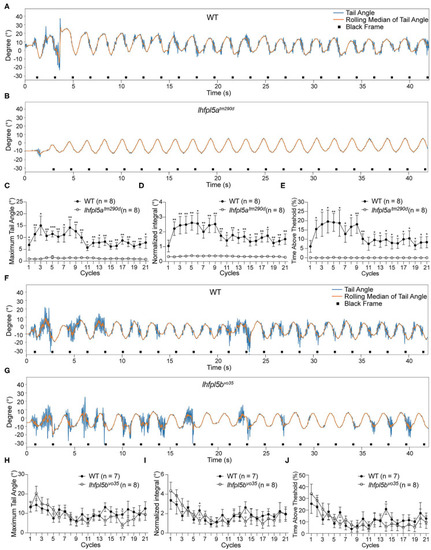
The VSR rapidly decreases over time in PHENOTYPE:
|
|
Longer pauses in activity and increased recovery time required for the resumption of the VSR in PHENOTYPE:
|