- Title
-
A Cellular-Resolution Atlas of the Larval Zebrafish Brain
- Authors
- Kunst, M., Laurell, E., Mokayes, N., Kramer, A., Kubo, F., Fernandes, A.M., Förster, D., Dal Maschio, M., Baier, H.
- Source
- Full text @ Neuron
|
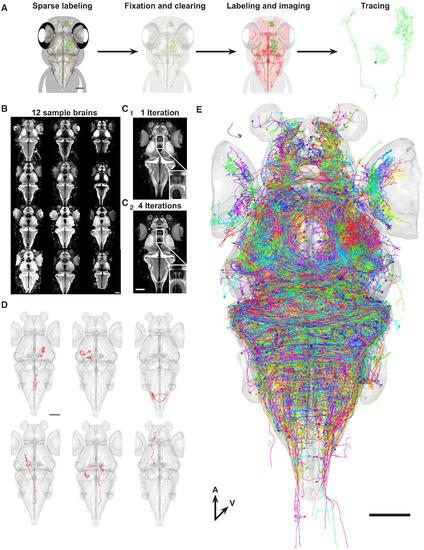
Construction of a Single-Neuron Atlas (A) Workflow for generating single-neurons tracings. (B) Images of 12 representative brains used to generate a shape-based average template. Brains were labeled with an antibody against the presynaptic protein Synapsin. (C) Shape-based average template brain after 1 iteration (C1) and 4 iterations (C2). Inset shows higher magnification of the torus longitudinalis neuropil. (D) Examples of individually traced neurons from different brains aligned to our template brain (gray surface). (E) Overlay of all neurons (n = 1,955) generated by our workflow. Colors of neurons are assigned randomly. All scale bars represent 100 μm. |
|
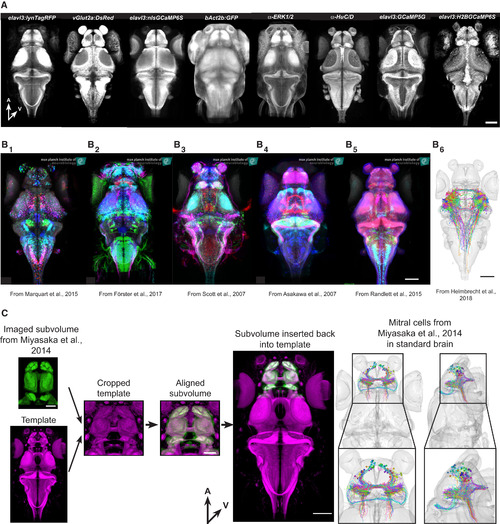
Integration of External Data (A) 8 examples of additional reference stains that can be used to align data to our resource. A, anterior; V, ventral. Scale bar represents 100 μm. (B) Examples of Gal4-line expression patterns and single-neuron tracings from different projects aligned to our common coordinate system. (B1) Enhancer-trap (ET) lines expressing UAS-Kaede from Marquart et al. (2015). (B2) BAC-trangenic lines expressing UAS-Dendrafrom Förster et al. (2017). (B3) ET lines expressing UAS-Dendra from Scott et al. (2007). (B4) Enhancer-trap (ET) lines expressing UAS-GFPfrom Asakawa et al. (2008). (B5) Various expression pattern from Randlett et al. (2015). (B6) Tectal projection neurons from Helmbrecht et al. (2018). All scale bars represent 100 μm. (C) Integration of mitral cell tracing from Miyasaka et al. (2014). Our reference brain was cropped to match the template of the original data. After successful alignment, the cropped volume was reinserted into the full-site reference brain. The various transformation steps were also applied to the mitral cell tracings. A, anterior; V, ventral. Scale bars in subvolume represent 50 μm; all other scale bars represent 100 μm. |
|
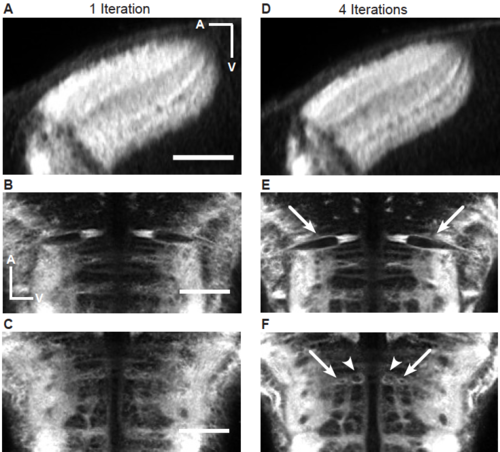
Improvement of granularity of the shape-based average brain after multiple iterations. (A-C) Single optical slices through the tectum (A) and the hindbrain (B and C) after one iteration. All scale bars represent 50 μm. (D-E) Single optical slices through the tectum (A) and the hindbrain (B and C) after four iterations. In the case of the tectum, individual tectal layers become more obvious and clearer after 4 iterations compared to one. Arrows in (B) highlight the Mauthner cells. Arrows and arrowheads in (C) highlight additional identifiable reticulospinal neurons. In both cases the neurons become either more sharply defined (B), or in the case of (C), only are recognizable after multiple iterations. |
|
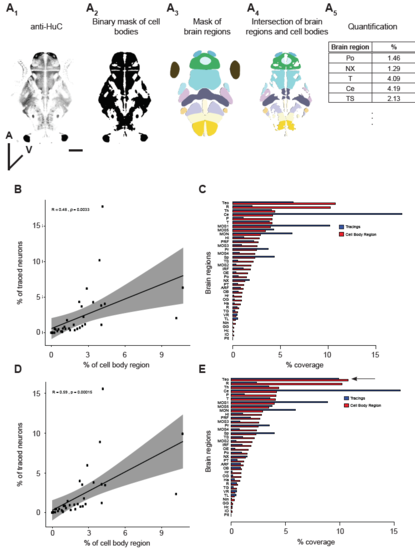
Analysis of labeling bias. (A) Workflow to calculate the expected relative contribution of each brain region to the single-neuron database. Neuronal cell bodies were labeled using an antibody directed against the post-mitotic marker HuC/D (A1). The staining was converted into a binary mask (A2) and this mask was intersected with a mask of 36 brain regions (A3). The result of this intersection is a mask that contains only the cellular portions of each brain region (A4). Based on this, we calculated the volume of each brain region in relation to the total volume (A5). Scale bar represents 100 μm. (B and C) Scatterplot (B) and barplot (C) showing correlation between the cell body region and the soma of traced neurons for individual brain regions for the pan-neuronal dataset. (D and E) Same as in B and C but for the complemented dataset. |
|
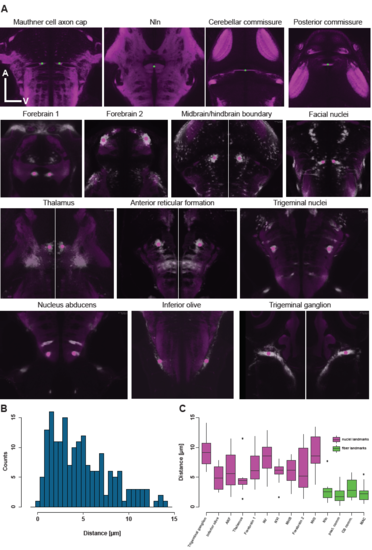
Location of fiducial points used for measurement of registration accuracy. (A) Single optical planes throughout the SYP-standard brain as well as through multiple ,different average Gal4-pattern indicating the location of the fiducial points (fiber landmarks are in green and nuclei landmarks are in magenta). (B) Histogram of all distances measured between fiducial points aligned to the standard brain and their respective counterpart in the standard brain. (C) Boxplot indicating the alignment accuracy for the individual fiducial points. Magenta refers to nuclei (Gal4-pattern) based landmarks and green to fiber (SYP-pattern) based landmarks. |
|
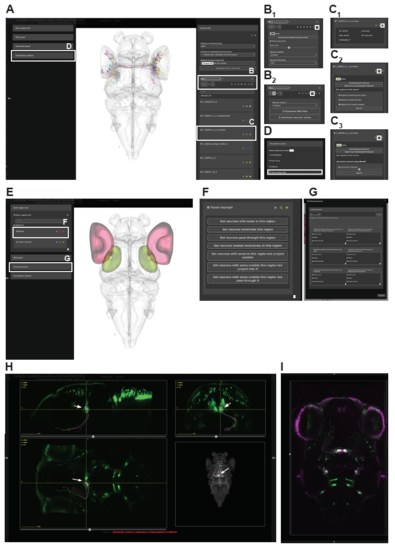
An interactive web-portal to explore single neuron data. (A) Interactive 3D viewer showing individual RGCs. White boxes indicate specific submenus to manipulate neurons and letters refer to figure panels with further explanation. (B-D) Specific submenus highlighted in (A). White box indicates icon that needs to be clicked to expand the menu and show option. (B) Manipulation of entire neuron groups. (B1) Visualization options for all selected neurons in a group. Users can change the color, show only the soma and also change the size of the displayed soma, flip the entire group to one hemisphere and adjust the thickness level of neurites. (B2) All selected neurons can be either downloaded as .swc files or their names can be downloaded into a text file that can be later uploaded to our website to only display those neurons. When downloading the tracings as a .swc file, the user can choose to download them in their original state or flipped to one of the two hemispheres. (C) Manipulation of single neuron tracings. (C1) Each neuron has metadata attached to it including a unique ID, the lab of origin, the Gal4-line that drove expression in that neuron, the cell-type (if already known), as well as the publication (in the form of a hyperlink), in which the tracing was originally described. (C2) For each individual neuron it is possible to display the brain regions innervated by it. Users can limit their search to specific parts of the neuron. (C2) Once a neuron of interest was found users, can look for neurons with a similar morphology using NBLAST. (D) Users can export the current 3D view by taking a screenshot and saving the image to their hard drive. (E-G) Atlas based search functions. (E) Interactive 3D viewer showing selected brain regions. In this case we chose the retina (red) and tectal neuropil (green). White boxes indicate specific submenus to manipulate search function and letters refer to figure panels with further explanation. (F) Options to search for neurons in a single brain region. (G) Advanced search options for multiple brain regions. Users can perform simple AND/OR search functions and can specify to only for search for specific parts of the neuron in each brain region. (H) User-defined ROI search function guided by a specific expression pattern. In this case we used Chat:Gal4 to visualize cranial nuclei in green. Arrows indicate the position of the ROI in the oculomotor nuclei with the accompanying oculomotor neurons resulting from a search for neurons with their soma within that ROI. (I) Overlay of Chat:Gal4 and isl1:GFP |