- Title
-
Direct and indirect roles of Fgf3 and Fgf10 in innervation and vascularisation of the vertebrate hypothalamic neurohypophysis
- Authors
- Liu, F., Pogoda, H.M., Pearson, C.A., Ohyama, K., Löhr, H., Hammerschmidt, M., and Placzek, M.
- Source
- Full text @ Development
|
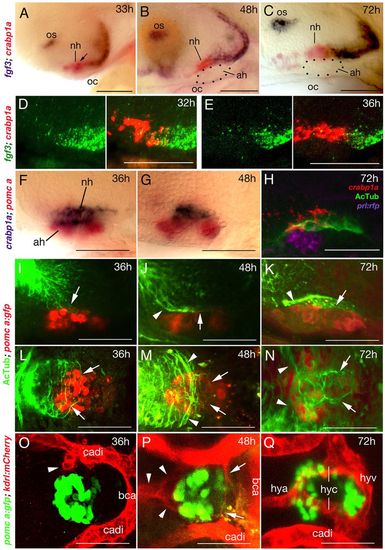
fgf3 expression and NH innervation and vascularisation in zebrafish. Lateral (A-K) or ventral (L-Q) views of wild-type zebrafish; anterior to the left. (A-C) fgf3 expression in posterior cells of the crabp1a+ NH (overlap in A indicated by arrow) and ventral-most cells of the posterior hypothalamus from 33 to 72 hpf. Dotted outline in B,C shows AH. (D,E) Double fluorescent in situ hybridisation reveals a partial overlap of fgf3 (green) and crapb1a (red) expression in posterior regions of the crabp1a domain at 32 hpf (D), whereas at 36 hpf, fgf3+ cells lie exclusively posterior of the crabp1a domain (E). (F,G) The crabp1a+ NH lies directly dorsal to the pomca+ AH. (H) Triple staining reveals innervation (AcTub) of NH (crabp1a) directly dorsal to AH (prl:RFP transgene product in lactotropes). Note that expression of AcTub and crabp1a signals are suboptimal owing to different fixation requirements. (I-N) Pioneering axons enter the anterior/dorsal NH anlage at 36 hpf (I,L). Axons are detected in the anterior half of the NH at 48 hpf (J,M), and throughout the NH at 72 hpf (K,N). Axons within the NH are indicated by arrows, axons outside the NH with arrowheads. For spatial reference, specimens are counterstained for the AH marker pomca. (O-Q) Endothelial cells (arrowhead) are detected at 36 hpf close to anterior regions of the pituitary (marked by pomca expression in AH) (O). At 48 hpf, endothelial cells assembling to vessels are detected anteriorly (arrowheads) and posteriorly (arrows) (P). At 72 hpf, the hypophyseal blood vessel system is completed (Q). ah, adenohypophysis; bca, basal communicating artery; cadi, caudal division of internal carotid artery; h, hours post fertilisation (hpf); hya, hypophyseal artery; hyc; hypophyseal capillary; hyv, hypophyseal vein; nh, neurohypophysis; oc, oral cavity; os, optic stalk. Scale bars: 50 μm. EXPRESSION / LABELING:
|
|
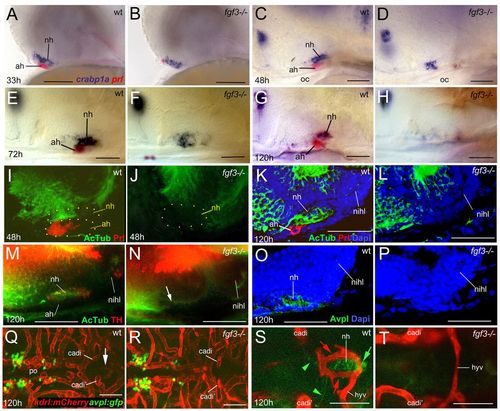
NH innervation and vascularisation are compromised in zebrafish fgf3 mutants. Whole-mount in situ hybridisation (A-H), or immunohistochemical analyses (whole-mounts: I,J,M,N; sections: K,L,O,P), and confocal images (Q-T) of wild-type (wt) and fgf3–/– zebrafish; lateral (A-P) or ventral (Q-T) views; anterior to the left. (A-H) fgf3–/– display robust crabp1a expression in the NH anlage at 33 hpf (B) and 48 hpf (D); prl+ AH cells are largely (B) or completely (D) lost. At 72 hpf, crapb1a expression is moderately reduced (E,F); only a remnant of diffuse expression detected at 120 hpf (G,H). (I-L) Axons enter the NH and then extend throughout its antero-posterior length in wt larvae, but remain outside the fgf3 mutant NH. AH and NH outlined by yellow dots. (M-P) At 120 hpf, AcTub+ axons in the wt NH include both TH+ (M) and Avpl+ (O) axons. Neither is detected in fgf3 mutant NH (N,P). (Q-T) fgf3 mutants display normal numbers of Avpl+ neurons in the hypothalamus (Q,R), but lose Avpl+ axonal NH innervation (T), the hypophyseal artery and the two bilateral hypophyseal capillaries (T). The hypophyseal vein remains present (T). Q and R display maximal confocal projections through midbrain area. Arrow (Q) points to pituitary region, magnified and intensified in S. Panels S and T show single confocal sections at higher magnification in same specimens as in Q and R. Red arrows (S) point to hypophyseal capillaries; green arrowheads to GFP+ axons of avpl:egfp+ neurons projecting to NH; green arrow to GFP+ axonal termini within NH. ah, adenohypophysis; cadi, caudal division of internal carotid artery; hyv, hypophyseal vein; nihl, nucleus of the inferior hypothalamic lobe; nh, neurohypophysis; oc, oral cavity; po, preoptic area. Scale bars: 50 μm. |
|
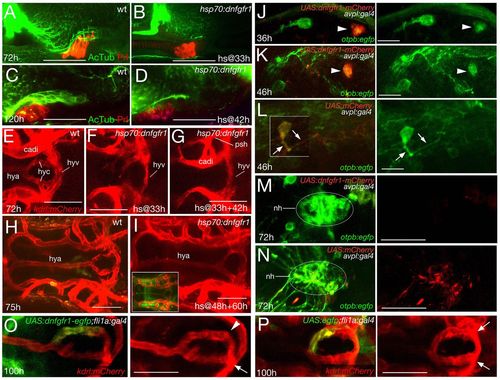
FGF signalling affects NH innervation and vascularisation independently of its effect on AH development and via direct effects on hypothalamic neurons and endothelial cells. Whole-mount immunolabelling (A-D) and confocal in vivo images (E-P) of wild-type (wt) or transgenic fish, as indicated. A-D: lateral views; E-I,M-P: ventral views, anterior to left; J-L: ventral views, anterior to top. (A-D) Double-labelled specimens for AcTub and Prolactin. (A,B) Maximal confocal projections at 72 hpf (A,B) or 120 hpf (C,D) in wt or after heatshock-activated dnFgfr1 expression at 33 hpf (B) or 42 hpf (D). Prolactin expression in the AH is normal, whereas NH axons appear to stall (compare B with Fig. 5I, and D with Fig. 5J). (E-I) Maximal projections of kdrl-driven mCherry fluorescence in endothelial cells at 72 hpf (E-G) or 75 hpf (H,I). Heatshock-activated dnFgfr1 expression at 33 hpf and 48 hpf leads to loss of hypophyseal artery and capillaries, but the hypophyseal vein remains present (compare panel G with E and with Fig. 6T). Single heatshock application at 33 hpf leads to an intermediate vessel phenotype (F), whereas heatshock applications at 48 hpf and 60 hpf leave hypophyseal vascularisation unaffected (compare panel I with H; inset in I shows strong and widespread expression of dnFgfr1-GFP). (J-N) Injection of UAS:dnfgfr1-mcherry plasmid (J,K,M) or UAS:mCherry plasmid (L,N) into tg(otpb:egfp);tg(avlp:gal4) double transgenic animals leads to mosaic expression of dominant-negative Fgfr1-mCherry fusion protein (J,K) or mCherry protein (L) in a subset of pre-optic hypothalamic cells (marked by otpb-driven EGFP expression, green). Avpl+ neurons expressing dnFgfr1-mCherry (arrowheads in J,K) fail to project axons and to innervate the NH (M), whereas neurons lacking dnFgfr1-mCherry display normal axogenesis (J,K). Axogenesis (L) and NH innervation (N) are also normal in Avpl+ neurons of control transgenics expressing mCherry (arrows in L). (O,P) Injection of UAS:dnfgfr1-egfp plasmid into tg(fli1:gal4);tg(kdrl:mcherry) double transgenic animals leads to mosaic expression of dominant-negative Fgfr1-EGFP fusion protein (green) in a subset of endothelial cells (marked by kdrl-driven mCherry expression, red). Contribution of cells expressing dnFgfr1 to one of the hypophyseal capillaries abrogates its fusion with the hypophyseal vein (O; arrowhead), whereas the fusion is not affected by cells carrying a UAS.egfp control plasmid (instead of UAS:dnfgfr1-egfp) (P; arrows). Arrow in O indicates normally wired non-transgenic endothelial cells. cadi, caudal division of internal carotid artery; hya, hypophyseal artery; hyc; hypophyseal capillary; hyv, hypophyseal vein; nh, neurohypophysis; phs, primary head sinus. Scale bars: 50 μm in A-I,M-P; 15 μm in J-L. EXPRESSION / LABELING:
PHENOTYPE:
|
|
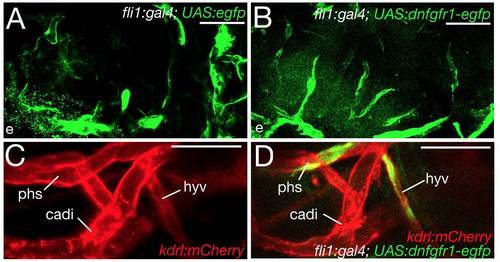
Endothelial cells expressing the dominant-negative Fgfr1 receptor contribute normally to other cephalic blood vessels and do not affect morphogenesis of the hypophyseal vein. (A-D) Confocal in vivo images of different transgenic fish, as indicated; 100 hpf; ventral views, anterior to the left. Injection of the UAS:egfp control (A) or the UAS:dnfgfr1-egfp plasmid (B,D) into tg(fli1a:gal4); tg(kdrl:mcherry) double transgenic animals leads to mosaic expression of EGFP (A) or the truncated Fgfr1 receptor fused to EGFP in endothelial cells. For simplicity, the red channel is omitted in A and B, and the green channel omitted in C. (A,B) Endothelial cells with activation of the UAS-egfp transgene (A) or the UAS:dnfgfr1-egfp transgene (B) display comparable contributions to cephalic blood vessels. Nevertheless, their contributions to the hypophyseal artery and the two branches encompassing the pituitary differ (80% versus 40%; data not shown). (C,D) Morphogenesis of the hypophyseal vein is unaffected by endothelial cells expressing the UAS:dnfgfr1-egfp transgene. cadi, caudal division of internal carotid artery; e, eye; hyv, hypophyseal vein; phs, primary head sinus. Scale bars: 50 μm. |