- Title
-
Establishment of a transgenic zebrafish line for superficial skin ablation and functional validation of apoptosis modulators in vivo
- Authors
- Chen, C.F., Chu, C.Y., Chen, T.H., Lee, S.J., Shen, C.N., and Hsiao, C.D.
- Source
- Full text @ PLoS One
|
The krt4 promoter can target transgene expression in the superficial skin layer in zebrafish. (A) Plastic section at 2 μm thickness showing that zebrafish skin, at 5 days post-fertilization, consists of an outer enveloping layer (EVL) and inner basal epidermal layer (BEL). The basement membrane is highlighted by the dotted line. (B) Plastic section at 2 μm thickness showing that adult zebrafish (aged at 3 months) skin consists of three major layers, including the superficial cells (arrowhead), middle and basal cells (arrows), and mucous cells (asterisk). (C) BrdU incorporation experiment showing that most skin layers in adult zebrafish are mitotically active. The BrdU+ cells (brown signals) can be detected in most skin layers at adult stage. (D) CK5/6 and (E) p63 antibodies differentially label the superficial skin layer and the putative epidermal stem cells in adult zebrafish skin, respectively. For immunohistochemistry, the 5 μm thick paraffin sections were immunostained with antigen-specific antibodies and visualized with DAB coloring substrate (brown). To visualize the cell morphology, the slides were counterstained with hematoxylin (blue). (F) Whole-mount immunostaining of p63 (red) on Tg(krt4:nlsEGFP)cy34 (green) embryonic yolk aged at 24 hpf, showing that the krt4 promoter targets the outermost EVL. The relative position of the captured image is highlighted at the upper right corner. (G?I) Immunohistochemistry of GFP (brown) on paraffin sections derived from Tg(krt4:nlsEGFP)cy34 aged at 3 months. The positive signals (brown) show that the krt4 promoter targets the superficial layer in the skin (G), esophagus (H) and gill (I) at adult stage. |
|
Establishment of Tg(krt4:NTR-hKikGR)cy17 killer line. (A) The work flow to conditionally ablate zebrafish skin using NTR/Met-mediated system. The superficial skin-specific krt4 promoter controls NTR-hKikGR fusion protein. Tol2 transposon elements flank the whole transgene cassette and enhance the germ-line transmission rate. Dimerization of NTR-hKikGR transfers electrons from NADH/NADPH to Met prodrug. Activated Met crosslinks DNA and specifically triggers apoptotic death in skin. (B) The killer line carrying the krt4:NTR-hKikGR transgene was crossed with several testing lines which overexpress human constitutively active myrAkt1 (myrAkt1), mouse constitutively active Stat3 (Stat3), or HPV16 E6 (E6) genes. The double transgenics were then subjected to Met incubation to assay the potential function of apoptosis modulators. (C?F) The ontogenic expression pattern of NTR-hKikGR fusion protein in killer line aged from 24 to 96 hpf. (G) The living fluorescent signals detected in double transgenics from the crossing of the killer line and Tg(krt4:nlsEGFP)cy34 aged at 72 hpf show that the NTR-hKikGR+ signals (yellow) aggregated adjacently to the nlsEGFP+ (green) skin nucleus. (H) Model to illustrate the spatial distribution of NTR-hKikGR fusion protein in zebrafish embryo skin. (I) The native hKikGR protein displays cytoplasmic distribution pattern in the skeletal muscle of Tg(mylz2:hKikGR). The relative position of the captured image is highlighted at the upper right corner. (J) The NTR-hKikGR fusion protein aggregated in skeletal muscle of Tg(mylz2:NTR-hKikGR). EVL, enveloping layer; BEL, basal epidermal layer; Met, metrodinazole. |
|
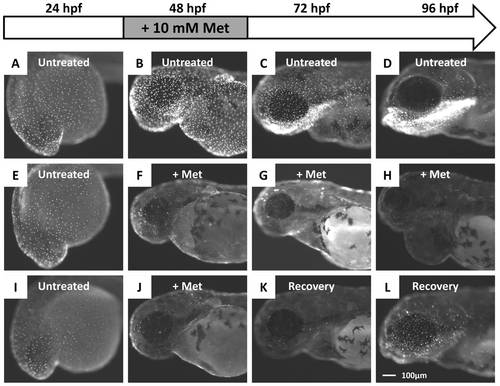
Administration of Met caused the killer line to lose the NTR-hKikGR+ fluorescent signals. (A?D) The ontogenic expression of NTR-hKikGR fusion protein in killer line embryos aged from 24 hpf to 96 hpf. (E?H) Consecutive incubation of killer line embryos with 10 mM Met, from 24 hpf to 96 hpf, caused the NTR-hKikGR+ fluorescent signals to gradually diminish by 48 hpf, totally disappear by 72 hpf, and show pericardial edema in Met-treated embryos by 96 hpf. (I?L) If Met was withdrawn and replaced with fresh fish water from 48 hpf onwards, the NTR-hKikGR+ fluorescent signals partially restored by 96 hpf. Scale bar = 100 μm in L (applies to A?L). The experimental design and work flow are illustrated at the top panel. Met, metrodinazole. |
|
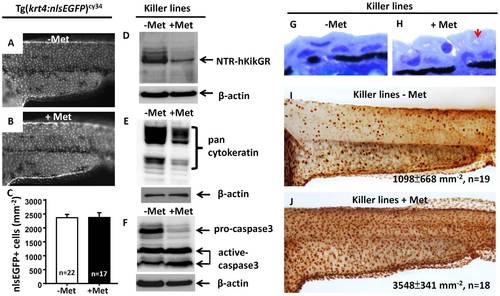
Apoptotic cell death mediates the loss of NTR-hKikGR+ fluorescent signals in Met-treated killer line. krt4 promoter suppression does not mediate loss of NTR-hKikGR+ fluorescence in Met-treated killer line because downregulation of nlsEGFP+ fluorescence signal intensity (A and B) or nlsEGFP+ cells (C) in Tg(krt4:nlsEGFP)cy34 did not occur with or without Met treatment. (D?F) Western blot analysis showed that apoptotic cell death mediates the loss of NTR-hKikGR+ fluorescent signals in Met-treated killer line embryos because the relative expression levels of NTR-hKikGR fusion protein (D), pan-cytokeratin (E) and pro-caspase 3 (F) greatly reduced in Met-treated embryos. Note that the relative intensities of the cleavaged caspase 3-immunoreactive signals greatly increased in the Met-treated killer line (F). (G?H) Plastic sections at 2 �m thickness show the greatly compromised superficial skin integrity in Met-treated killer line embryos (arrow). Whole-mount TUNEL assay demonstrated significantly enhanced cell death signals in Met treated killer line embryos (J) compared to the untreated group (I). The cell number is presented as the mean�S.D. Student′s t-test was used to make statistical comparisons between untreated (-Met) and Met-treated (+Met) killer lines. Met, metrodinazole. |
|
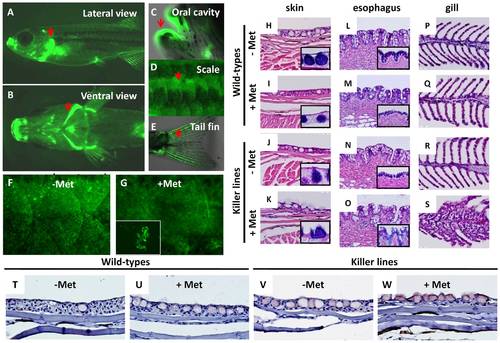
Evaluation of skin ablation by NTR/Met ablation system in adult killer line. The lateral (A) and ventral (B) views of fluorescent appearance of NTR-hKikGR fusion protein in the killer line at adult stage. Some regions like gill operculum (A, B), oral cavity (C), scale (D) and tail fin (E), which are rich in epithelial structures, showed robust fluorescent signals (heighted by arrows). (F?G) Test of the possibility of performing skin ablation in the adult killer line. Treatment of the killer line with 2.5 mM Met for three consecutive days resulted in greatly compromised skin integrity and some detached skin debris (NTR-hKikGR+) in the fish tank. (H?S) Histological assessment of skin integrity in wild-types or killer lines treated with or without 2.5 mM Met. Paraffin sections stained with hematoxylin and eosin showing the serial morphological changes in the regions of superficial skin (H?K), esophagus (L?O) and gill epithelium (P?S). Included for comparison, the normal epidermal histology in superficial skin (H), esophagus (L) and gill (P) before performing skin ablation. To highlight the position of mucous cells, PAS staining (blue color) in paraffin sections derived from skin (H?K) and esophagus (L?O) shown in lower right corners. (T?W) Detection of cell apoptosis in the damaged skin by activated caspase 3 antibody staining on paraffin sections (brown signals). The killer line adults were first incubated with 2.5 mM Met solution for three consecutive days to execute cell ablation and then paraffin sections, at 5 μm intervals, were cut for histological assay or immunohistochemistry. Met, metrodinazole; PAS, Periodic Acid Schiff. |
|
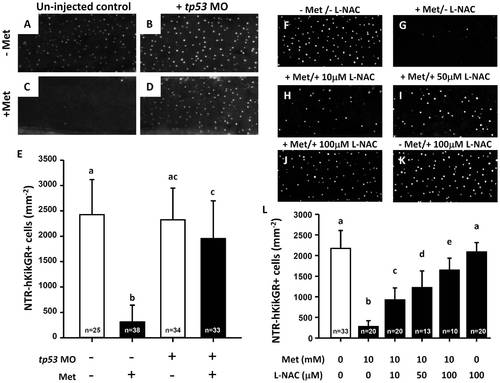
The loss of NTR-hKikGR+ fluorescent signals in Met-treated killer line correlates with tp53 activity and oxidative stress in skin. (A?D) Test of tp53-dependency on the loss of NTR-hKikGR+ fluorescent signals by tp53 morpholino injection. (E) Statistical comparison of the relative number of NTR-hKikGR+ fluorescent signals in tp53 morphants treated with or without Met. (F?K) Test of oxidative status-dependency on the loss of NTR-hKikGR+ fluorescent signals by incubation with various concentrations of L-NAC as an anti-oxidant. (L) Statistical comparison of the relative number of NTR-hKikGR+ fluorescent signals in killer line embryos treated with Met and/or L-NAC. The cell number is presented as the mean�S.D. Different letters above the error bars indicate significant differences, as tested by one-way ANOVA with Tukey′s pair-wise comparison method. L-NAC, N-acetyl-L-cysteine. |
|
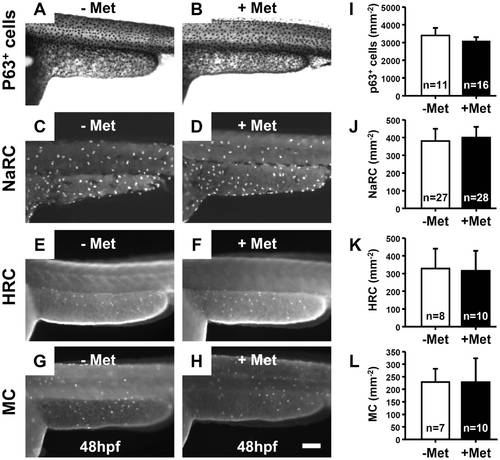
Skin ablation in Met-treated killer line specifically targeted the EVL and showed no bystander effect on the neighboring cells. Whole-mount immunostaining on killer line embryos aged at 48 hpf showing that the number of epidermal stem basal cells (A and B, stained with p63 antibody), NaRC (C and D, stained with Na,K-ATPase antibody), HRC (E and F, stained with H-ATPase antibody) and MC (G and H, stained with anterior gradient 2 antibody) did not significantly change between untreated (-Met) and treated (+Met) groups. (I?L) Statistical comparisons, using Student′s t-test, of the relative cell numbers of p63+ cells, NaRC, HRC and MC between untreated (-Met) and Met-treated (+Met) groups. The cell number is presented as the mean�S.D. NaRC, Na-K-ATPase rich cell; HRC, H-ATPase rich cells; MC, mucous cells; Met, metrodinazole. |
|
Functional validation of potential apoptosis modulators in the living killer line embryos by crossing transgenic lines or transient plasmid DNA injection. (A?H) The NTR-hKikGR+ fluorescent signals in embryos derived from crossing of killer line with testing line carrying human constitutively active Akt1 (myrAkt1) (C, D), mouse constitutively active Stat3 (Stat3) (E, F), or HPV16 E6 (E6) transgenes (G, H). (I?N) NTR-hKikGR+ fluorescent signals in killer line embryos after injection with plasmids carrying myrAkt1 (I, J), Stat3 (K, L), or E6 transgenes (M, N). (O) Statistical comparison of the relative number of NTR-hKikGR+ fluorescent signals in embryos derived from stable line assay (black bars) or transient assay (red bars). The cell number is presented as the mean�S.D. Different letters above the error bars indicate significant differences as tested using one-way ANOVA with Tukey′s pair-wise comparison method. Met, metrodinazole. |
|
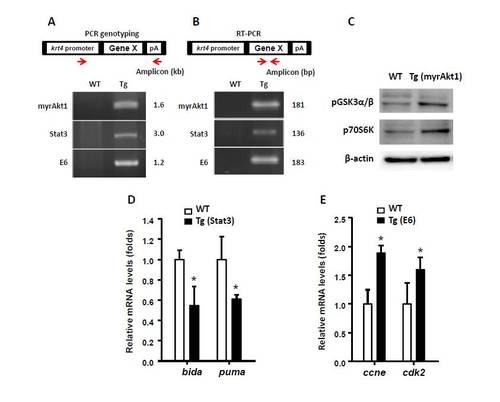
Validation of the transmission, expression and function of testing lines in stable transgenics using genotyping, RT-PCR and Western blot analysis. Assay of the stable transmission and expression of transgenes in different testing lines at the DNA and mRNA levels by genotyping (A) and RT-PCR (B), respectively. The relative position of the primers used to perform genotyping or RT-PCR is illustrated by red arrows. The predicted amplicon size is also illustrated in the right panel. The plasmid architectures were highlighted in the upper panel. (C) Western blot assay on protein lysates extracted from adult tail fins showing highly phosphorylated Akt downstream targets of GSK3α/β and 70S6K in myrAkt1 testing line. β-actin served as a protein loading control. (D) Quantitative real-time RT-PCR showed that overexpression of pro-survival genes of Stat3 down-regulates the pro-apoptotic genes bida and puma in transgenic embryos aged at 48 hpf. (E) Quantitative real-time RT-PCR showed that overexpression of virus oncogene of HPV16 E6 up-regulates the cell cycle-related genes ccne and cdk2 in transgenic embryos aged at 48 hpf. |