- Title
-
The iguana/DZIP1 protein is a novel component of the ciliogenic pathway essential for axonemal biogenesis
- Authors
- Tay, S.Y., Yu, X., Wong, K.N., Panse, P., Ng, C.P., and Roy, S.
- Source
- Full text @ Dev. Dyn.
|
Igu is essential for primary cilia formation. A: Wild-type myotome showing abundant primary cilia (arrows) on the surface of muscle cells. B: Myotome of an igu mutant embryo showing complete absence of primary cilia. C: Primary cilia (arrows) in differentiating retinal cells of a wild-type embryo. D: Lack of primary cilia from the retina of an igu mutant. E: Primary cilia (arrows) in paraxial mesodermal cells of a wild-type embryo. F: Absence of primary cilia from paraxial mesodermal cells of an igu mutant embryo. G: Severe reduction in numbers of primary cilia (arrow) from paraxial mesodermal cells of an igu morphant embryo. H: An igu mutant embryo injected with igu mRNA showing rescue of the primary cilia (arrows) in muscle cells. Axonemes of primary cilia were visualized with anti-acetylated tubulin antibodies (red), anti-βcatenin antibodies were used to highlight cell membranes (green; A-D and H), and nuclei were visualized with DAPI (blue; 4′,6-diamidine-2-phenylidole-dihydrochloride). In A-D and H, embryos are depicted with anterior to the left and dorsal to the top; E-G show dorsal views of the paraxial mesodermal cells. Scale bars = 10 μm. |
|
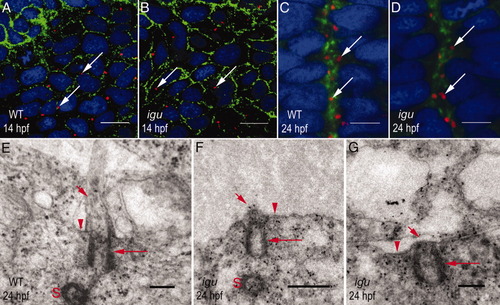
Igu is required for axonemal biogenesis, but not for the docking of basal bodies to apical membranes. A: Distribution of basal bodies (arrows) in paraxial mesodermal cells of a wild-type embryo. B: Paraxial mesodermal cells of an igu mutant embryo with wild-type pattern of basal body distribution (arrows). C: Polarized orientation of basal bodies (arrows) toward the apical membranes of cells abutting the midline of the neural tube of a wild-type embryo. D: Apical polarization of basal bodies (arrows) in the neural tube is unaffected in an igu mutant embryo. E: A representative TEM section through the neural tube of a wild-type embryo showing the basal body (long arrow) and ciliary axoneme (short arrow). The apical membrane of the ciliary pit is indicated (arrowhead). The sister centriole (S) is also visible. F,G: Similar sections through the neural tube of igu mutant embryos showing basal bodies (long arrows) apparently docked to apical membranes, but a complete lack of axonemal outgrowth (short arrows). No invagination of the apical membranes (arrowheads) to form ciliary pits is visible. In F, the sister centriole (S) is visible. In A-D, basal bodies were visualized with anti-γ-tubulin antibodies (red), cell membranes with anti-βcatenin antibodies (green) and nuclei with DAPI (blue). A and B show dorsal views of the paraxial mesodermal cells; C-G represent transverse sections through the neural tube. Scale bars = 10 μm in A,B, 5 μm in C,D, 0.2 μm in E,G, 0.5 μm in F. PHENOTYPE:
|
|
Motile ciliogenesis is less strongly perturbed in igu mutants. A: Motile cilia (arrows) in Kupffer′s vesicle (KV) of a wild-type embryo. B: Reduction in the numbers of motile cilia (arrows) in KV of an igu mutant embryo. C: Motile cilia (arrows) in the proximal segment of the pronephric duct of a wild-type embryo. D: Numbers of motile cilia (arrows) are reduced in the proximal pronephric duct of an igu mutant embryo. E: Motile cilia (arrows) in the distal segment of the pronephric duct of a wild-type embryo. F: Substantial reduction in the numbers of motile cilia (arrows) in the distal pronephric duct of an igu mutant embryo. G: A wild-type embryo with Foxj1a mis-expression (green) in muscle cells of the myotome, showing ectopic long cilia (arrows). H: No ectopic cilia formation in an igu mutant embryo with Foxj1a mis-expression (green) in myotomal muscle cells. In all panels, ciliary axonemes were labeled with anti-acetylated tubulin antibodies (red) and nuclei with DAPI (blue; 4′,6-diamidine-2-phenylidole-dihydrochloride). In A and B, circumference of KV is outlined with the dashed circle. Ectopic Foxj1a expression (green) in G and H was detected with antibodies against the HA tag at the C-terminus of the Foxj1a protein. A and B represent ventral views of KV; embryos in C-H are depicted anterior to the left, dorsal to the top. Scale bars = 10 μm. PHENOTYPE:
|
|
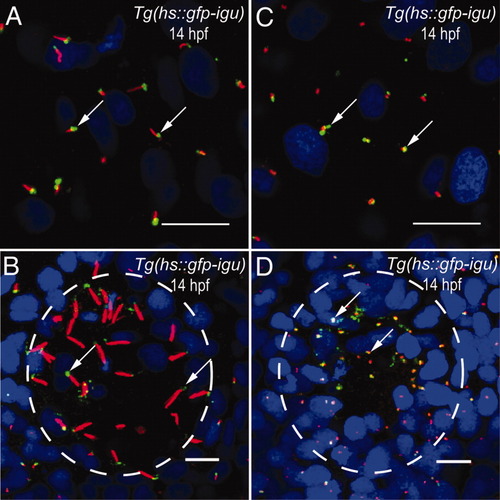
The Igu protein localizes to the bases of primary and motile cilia. A: Localization of green fluorescent protein (GFP) -Igu (green, arrows) to the bases of primary cilia (red) on paraxial mesodermal cells of a Tg(hs::gfp-igu) embryo. B: Localization of GFP-Igu (green, arrows) to the bases of Kupffer′s vesicle (KV) motile cilia (red) of a Tg(hs::gfp-igu) embryo. C: Localization of GFP-Igu (green, arrows) to basal bodies (red) of primary cilia on paraxial mesodermal cells of a Tg(hs::gfp-igu) embryo. D: Localization of GFP-Igu (green, arrows) to basal bodies (red) of KV motile cilia of a Tg(hs::gfp-igu) embryo. Embryos depicted in A and B were stained with anti-GFP (green) and anti-acetylated tubulin (red) antibodies, and those in C and D with anti-GFP (green) and anti-γ-tubulin antibodies (red). Nuclei were labeled with DAPI (blue). In B and D, circumference of KV is outlined with the dashed circle. A and C depict dorsal views of paraxial mesodermal cells; B and D represent ventral views of KV. Scale bars = 10 μm. |

Unillustrated author statements PHENOTYPE:
|