- Title
-
Nodal signaling activates differentiation genes during zebrafish gastrulation
- Authors
- Bennett, J.T., Joubin, K., Cheng, S., Aanstad, P., Herwig, R., Clark, M., Lehrach, H., and Schier, A.F.
- Source
- Full text @ Dev. Biol.
|
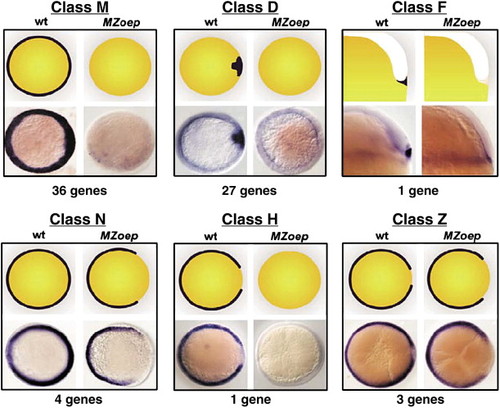
Diverse expression patterns of Nodal-regulated genes. All 72 Nodal-regulated genes are grouped into six classes based on dorsal?ventral expression pattern at the beginning of gastrulation. The top two images of each cluster are cartoon representations of the expression pattern, the bottom two are whole-mount ISH. Genotypes are indicated. All images are animal pole views except class F, which is a lateral view. |
|
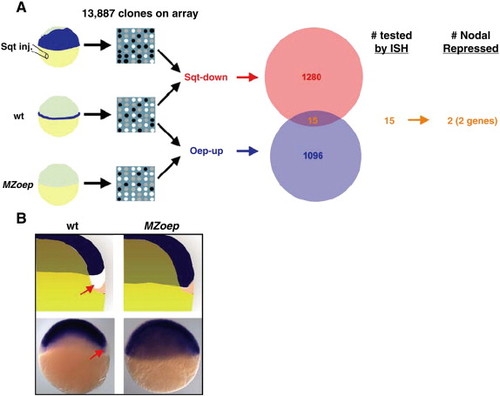
Identification of 2 Nodal-repressed genes. (A) The Venn diagram indicates the number of identified Sqt-down (red) and Oep-up (blue) clones. The overlap (orange) is indicated. Overlapping clones were screened by ISH. Two Nodal-repressed genes, sox2 and sox3, were identified. (B) Expression patterns of sox2 and sox3. The top two images are cartoon representations; the bottom two images are whole-mount ISH of sox3. Genotypes are indicated. Arrows indicate the region that lacks expression in wild type. |
|
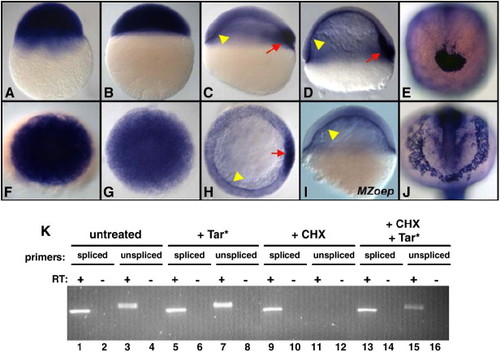
Nodal signaling directly induces xbp1 transcription. (A?J) Expression of xbp1 mRNA was detected by whole-mount ISH. Maternally provided xbp1 is ubiquitous at 2.75 hpf (A, F) and 4 hpf (B, G) and becomes restricted to the YSL (arrowheads) and dorsal mesoderm (arrow) at 30% epiboly (C, H) and 60% epiboly stages (D). Nodal signaling is required for dorsal mesoderm expression of xbp1 but not YSL expression (I). xbp1 is expressed in dorsal mesoderm derivatives, including the polster at tailbud stages (E) and the hatching gland at 18 somites (J). All embryos are wild type except (I), MZoep. (A?D, I) lateral, (F?H) animal, (E, J) anterior view. (K) Activation of xbp1 expression by Nodal signaling does not require protein translation. Lanes 1?15, RT-PCR for spliced/unspliced xbp1 mRNA from 5.5 hpf embryos. Untreated embryos contain both spliced (lane 1) and unspliced (lane 3) transcripts. Unspliced xbp1 transcription is moderately induced by tar* mRNA injection (lane 7). Tar* mRNA injection induces high-level ubiquitous xbp1 expression by whole-mount ISH (not shown); the relatively moderate induction of unspliced xbp1 transcripts may reflect elevated rates of xbp1 splicing. Cycloheximide (CHX) blocks the expression of unspliced xbp1 mRNA (lane 11), although spliced xbp1 mRNA persists (lane 9). Tar* mRNA injection induces unspliced xbp1 even in the presence of CHX (lane 15), although at reduced levels (compare lanes 15 and 7). The reduced levels of tar*-induced unspliced xbp1 expression in the presence of CHX may reflect an additional indirect component to Nodal activation of xbp1 expression, or it could be due to reduced production of Tar* protein in the presence of CHX. We initially assayed xbp1 expression via whole-mount ISH, a technique previously used to report the direct/indirect status of other Nodal-regulated genes (Poulain and Lepage, 2002). Although Tar* injection did lead to ectopic expression of xbp1, so did treatment with CHX alone (not shown), making the results of this experiment uninterpretable (Taverner et al., 2005). Given the strong maternal expression of xbp1 and the rapid kinetics with which it is extinguished at the beginning of zygotic transcription, we hypothesize that CHX treatment might stabilize the ubiquitous maternal xbp1 message. EXPRESSION / LABELING:
|
|
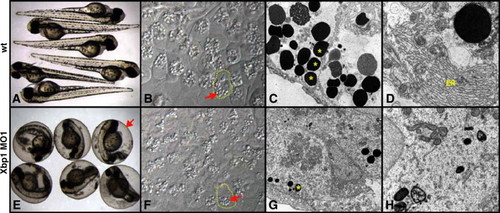
Xbp1 is required for terminal differentiation of the hatching gland. (A?D) Wild-type embryos, (E-H) Xbp1 morpholino (MO1) injected. (A, E) Live, 4 dpf embryos. Embryos injected with 5 ng MO1 (E) do not hatch from their chorions (arrow). Embryos injected with 2 ng MO2 are identical (not shown). (B, F) 40� light microscopy of fixed hatching gland tissue at 35 hpf. Embryos injected with 7.5 ng MO1 (F) have a drastic reduction in size of secretory vesicles (arrow). Single hatching gland cells are outlined. Distinct cell boundaries are present in Xbp1 MO1-injected hatching gland cells, but they are less visible in this focal plane. (C, D, G,H) Electron micrographs of hatching gland tissue (35 hpf), at 4400X (C, G) and 11 500� (D, H) magnification. Embryos injected with 7.5 ng MO1 (G) have fewer and smaller secretory vesicles (asterisk) than wild-type (C). The elaborate cisternae of the rough ER (D) are not seen in MO injected embryos (H). |
Reprinted from Developmental Biology, 304(2), Bennett, J.T., Joubin, K., Cheng, S., Aanstad, P., Herwig, R., Clark, M., Lehrach, H., and Schier, A.F., Nodal signaling activates differentiation genes during zebrafish gastrulation, 525-540, Copyright (2007) with permission from Elsevier. Full text @ Dev. Biol.