Fig. 3
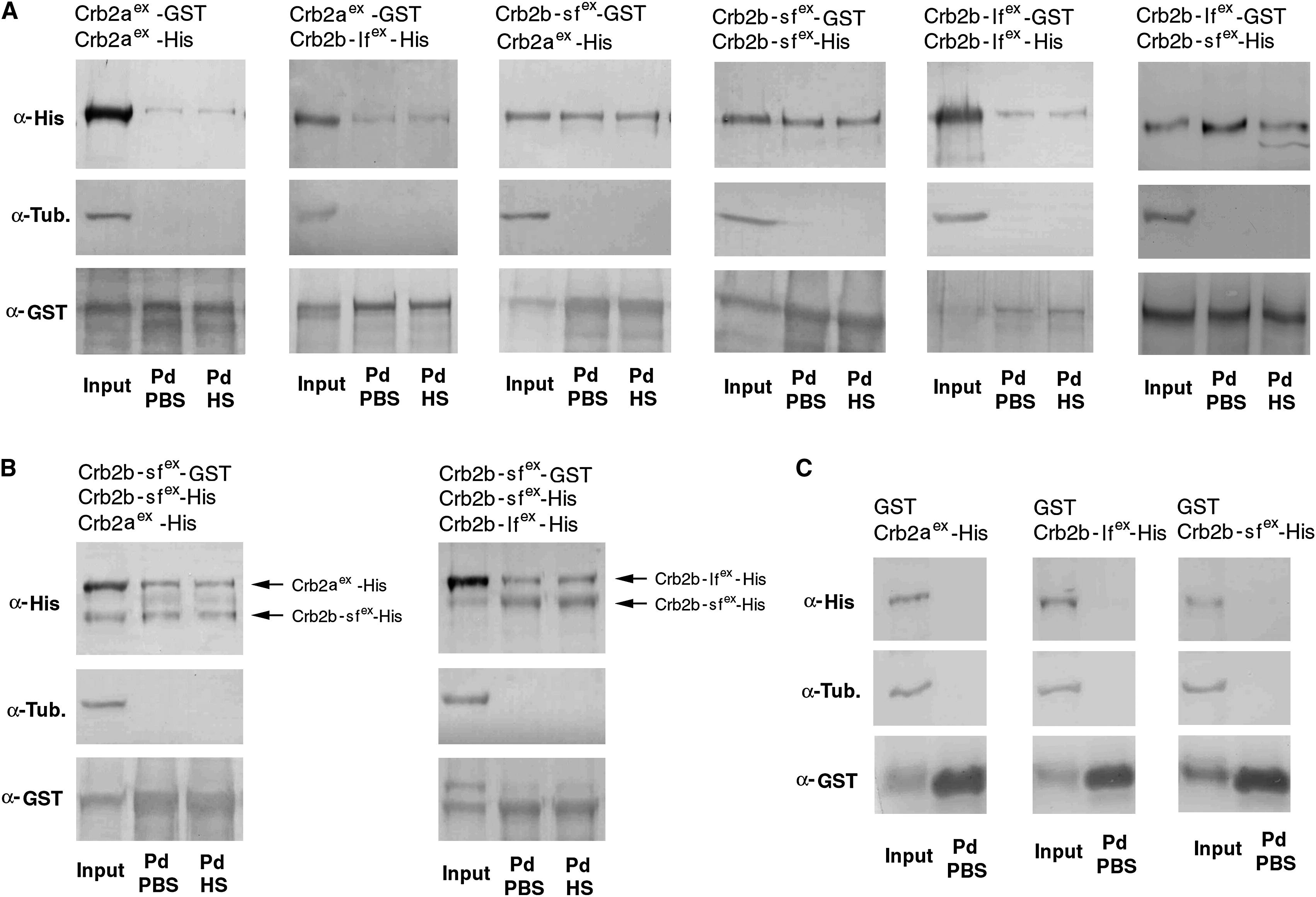
The Extracellular Domains of Crb2a, Crb2b-lf, and Crb2b-sf Mediate Homophilic and Heterophilic Physical Adhesion Various combinations (indicated above the blots) of GST and His fusions of the extracellular domains of Crb2a, Crb2b-lf, and Crb2b-sf were coexpressed in sf9 cells. The cell lysates were analyzed by GST pull-down and western blotting assays. Anti-GST blots confirmed the efficient pull down of GST fusion proteins. Anti-α-tubulin blotting indicated the absence of cytoplasmic protein contamination in the pull-down fractions (Pd). The pull-down fractions were washed either with PBS (PBS) only or with PBS as well as 1 M NaCl high-salt solution (HS). (A) Pull-down analyses of pairwise and coexpressed GST- and His-fusions indicated that the extracellular domain of Crb2a, Crb2b-lf, and Crb2b-sf physically adhered to each other both homo- and heterophilically. In addition, the adhesion was strong enough to withstand high ionic washes. (B) Competition pull-down analyses revealed that Crb2b-sfex-GST bound to Crb2b-sfex-His more efficiently than it bound to Crb2b-lfex-His or Crb2aex-His, as suggested by the higher ratios between Crb2b-sfex-His and its Crb2b-lfex-His or Crb2aex-His competitors in the pull-down fractions than in the input fractions. (C) As specificity controls, when coexpressed with Crb2aex-His, Crb2b-lfex-His, or Crb2b-sfex-His, GST itself did not pull down any Crb-His fusions, indicating that these His-fusions did not bind to GST or glutathione resin nonspecifically.
Reprinted from Developmental Cell, 22(6), Zou, J., Wang, X., and Wei, X., Crb Apical Polarity Proteins Maintain Zebrafish Retinal Cone Mosaics via Intercellular Binding of Their Extracellular Domains, 1261-1274, Copyright (2012) with permission from Elsevier. Full text @ Dev. Cell