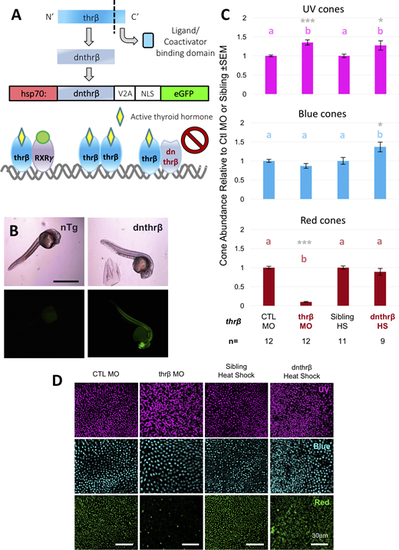
A transgenic zebrafish model of conditional Thr? disruption reveals additional roles in cone development. (A) Generation of a dominant negative thyroid hormone receptor ? was accomplished via an 11-amino acid C-terminal deletion in thr?, which was then cloned into a transgene for dominant negative thr? (dnthr?) and eGFP under the hsp70 promoter. Endogenous Thr? dimerizes with other factors, such as Thr? or RXR?, and binds thyroid hormone to activate transcription (left), whereas dnThr? would bind endogenous receptors but, lacking ligand and coactivator binding domains, would render dimers inactive (right). (B) Transgenic embryos express the transgene, including GFP, throughout the body. Scale bar: 1 mm. (C) Thr? MO causes dramatic reduction in red cone abundance and increased UV abundance at 10-ng dose. Our dominant negative model shows the same increase in UV cones but also a significant increase in blue cones (HS, heat shocked). Values with matching letters are not significantly different (Kruskal-Wallis test with Mann-Whitney pairwise comparisons, gray asterisks indicate *P < 0.05, **P < 0.01, ***P < 0.001 relative to CTL MO or sibling heat-shocked [HS] controls). Thr? MO data are normalized to CTL MO data; dnthr? HS data are normalized to sibling HS data.
|

