- Title
-
Identification and genetic analysis of rare variants in myosin family genes in 412 Han Chinese congenital heart disease patients
- Authors
- Zhang, Y., Peng, R., Wang, H.
- Source
- Full text @ Mol Genet Genomic Med
|
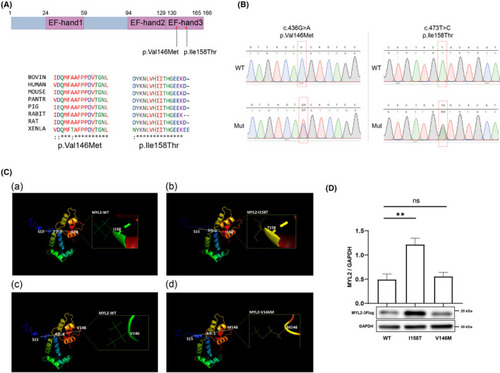
Schematic representation of missense MYL2 variants. (A) MYL2 protein with 166 amino acids showing three EF?hand domains. The red stars indicate positions of MYL2 variants, and their evolutionary conservation across the species is shown below. (B) DNA sequence chromatogram of wild?type and mutant variants. The sequences were confirmed by Sanger sequencing. The red squares mark the positions of variants. WT, wild?type; MUT, mutant. (C) The secondary structure prediction of WT, p. Ile158Thr and p. Val146Met MYL2 was performed using I?TASSER. (a). MYL2?WT structure, which emphasized the Ile158 site. The distance between the C?? of Ser15 and the C?? of Ile158 is 27.5 �. (b) MYL2?I158T structure, which emphasized the Thr158 site. The distance between the C?? of Ser15 and the C?? of Thr158 is 30.0 �, which is longer than the relevant distance in MYL2?WT. (c). MYL2?WT structure, which emphasized the Var146 site. The distance between the C?? of Ser15 and the C?? of Var146 is 40.4 �. (d). MYL2?V146 M structure, which emphasized the Met146 site. The distance between the C?? of Ser15 and the C?? of Met146 is 43.1 �, which is shorter than the relevant distance in MYL2?WT. (D) Western blot analysis of wild?type and mutant MYL2 proteins in HEK293T cells. GAPDH was used as a loading control for the cell lysate. ns p > .05, *p < .05, ** < .01. |
|
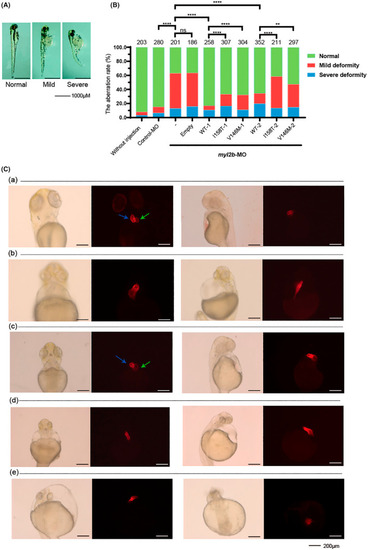
MYL2 variant effects in zebrafish embryos. (A) Typical three?day postfertilization zebrafish morphology in three categories according to severity. We clustered all injected zebrafish embryos into normal/mild deformity/severe deformity groups. The zebrafish were mainly divided by heart abnormalities. ?Normal? indicated no apparent malformation in either the heart or body. ?Mild? and ?severe? indicated the degree of heart failure and malformation. (B) Myl2b?knockdown zebrafish embryos rescued by human WT, p. Ile158Thr, p. Val146Met MYL2 plasmids, respectively. Two different dosage sets were injected into zebrafish embryos. The WT?1, I158T?1 and V146 M?1 groups were injected with 40 ng/?l plasmids. The WT?2, I158T?2 and V146 M?2 groups were injected with 50 ng/?l plasmids. The Y?axis represents the percentages of the three categories according to degrees in Figure 2A. The number above each bar is the total number of embryos examined under each experimental condition. ns p > .05, *p < .05, ** < .01, ***p < .001, ****p < .0001. (C) Transgenic zebrafish line Tg (Cmlc2: mCherry) embryos in which cardiomyocytes are specifically labeled with red dye were injected with human MYL2, p. Ile158Thr, p. Val146Met plasmids to depict the effects of p. Ile158Thr and p. Val146Met on zebrafish heart. The figure shows the five groups. (a). Wild?type zebrafish. (b). The zebrafish injected with myl2b?MO. (c). The zebrafish injected with myl2b?MO and human MYL2?WT plasmids. (d). The zebrafish injected with myl2b?MO and human MYL2 p. Ile158Thr plasmids. (e). The zebrafish injected with myl2b?MO and human MYL2 p. Val146Met plasmids. Two representative orientations of each experimental condition were presented, and each orientation consisted of a light field and a fluorescence microscope. The blue arrows indicate the heart ventricle, and the green arrows indicate the heart atrium. PHENOTYPE:
|
|
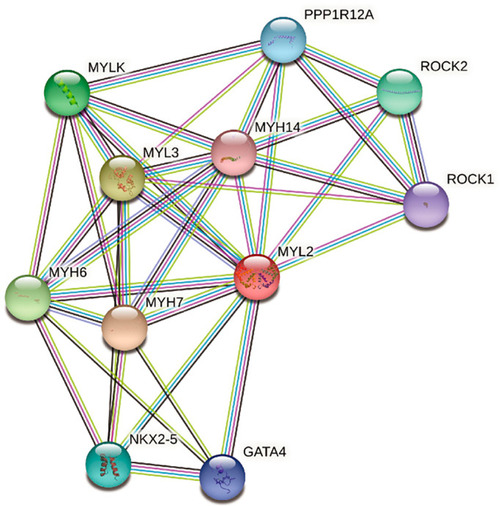
Ten potential direct interactors of MYL2 detected by the STRING database (https://string?db.org/) |