- Title
-
The broad role of Nkx3.2 in the development of the zebrafish axial skeleton
- Authors
- Waldmann, L., Leyhr, J., Zhang, H., �hman-M�gi, C., Allalou, A., Haitina, T.
- Source
- Full text @ PLoS One
|
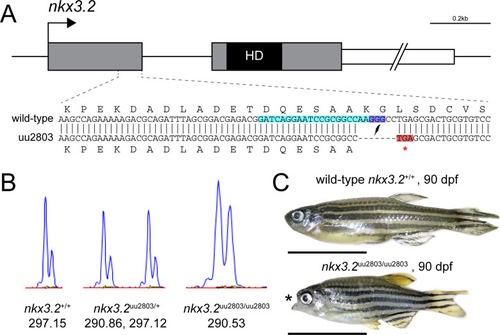
(A) To-scale schematic of the two-exon zebrafish nkx3.2 gene locus on chromosome 14. Grey boxes represent the coding sequence, the black box labelled ?HD? marks the location of the homeodomain-coding sequence in the second exon, and the white box marks the 1,226bp 3? UTR, truncated for illustrative purposes. gRNA target site is marked by turquoise background. The sequence of the zoomed in section of exon 1 shows the alignment between the wild-type and mutant (uu2803) alleles. The mutant allele has a 7 base-pair deletion that results in a frameshift and a premature stop codon (highlighted in red) immediately after the deletion site. (B) Wild-type, heterozygous nkx3.2uu2803/+ and homozygous nkx3.2uu2803/uu2803 fish were genotyped by fragment length analysis. The wild-type displayed one peak (297,15), nkx3.2uu2803/+ fish one wild-type (297,12) and one mutant (290,86) peak and nkx3.2uu2803/uu2803 fish one mutant peak (290,53). (C) nkx3.2uu2803/uu2803 fish at 90 dpf display a prominent fixed open mouth phenotype compared to wild-type at 90 dpf. Scale bars: 1 cm. PHENOTYPE:
|
|
(A-H?) Maximum projections of confocal live imaging Z-stacks acquired from ventral side of wild-type zebrafish head at 3 dpf (A, A?), 5 dpf (B, B?), 7dpf (C, C?), 14 dpf (D, D?) and nkx3.2uu2803/uu2803 zebrafish head at 3 dpf (E, E?), 5 dpf (F, F?), 7 dpf (G, G?), 14 dpf (H, H?) in Tg(sox10:egfp) background. Corresponding single Z-planes are shown in A??H?. (A-D��) The jaw joint in wild-type zebrafish (dashed box) is magnified in A?-D?. The retroarticular process is visible from 3 dpf onwards, marked by white arrowhead. Chondrocytes of the anterior Meckel?s cartilage and posterior palatoquadrate align in stacks. Posterior Meckel?s cartilage and anterior palatoquadrate articulate the jaw joint. (E-H?) Fusion of jaw joint articulating elements in nkx3.2 mutants. The fusion site is magnified in (E?-H?) and indicated by asterisks. (E?-F? and E?-F?) At 3 dpf and 5 dpf nkx3.2 mutants display unorganised and rounded cells in the anterior palatoquadrate, posterior to the jaw joint fusion site (G?, G?) At 7 dpf nkx3.2 mutants display elongated chondrocytes that start to align in stacks. (H?, H?) At 14 dpf nkx3.2 mutants display elongated chondrocytes that are aligned throughout the fused Meckel?s-palatoquadrate element. m?Meckel?s cartilage, pq?palatoquadrate. Scale bars: 100 ?m (A-H), 25 ?m in (A?-H? and A?-H?). EXPRESSION / LABELING:
PHENOTYPE:
|
|
Sagittal sections of wild-type 14 dpf (A, A?), 30 dpf (C, C?) and 90 dpf (E, E?) and nkx3.2 mutant 14 dpf (B, B?) 30 dpf (D, D?) and 90 dpf (F, F?) zebrafish stained with Nuclear Red. (A, A?, C, C?, E, E?) Stained histology sections displaying a normal jaw joint development between Meckel?s cartilage (m) and palatoquadrate (pq), respectively articular (art) and quadrate (q) in wild-type fish. (B, B?, D, D?, F, F?) nkx3.2 mutants do not display a jaw joint. Chondrocyte maturation and ossification seem not to be affected in nkx3.2uu2803/uu2803 besides the absence of joint-typical cells lining the articulating elements. Dashed box in (A-F) marks the magnified region in (A?-F?). Asterisk marks the fused element. m?Meckel?s cartilage, pq?palatoquadrate, art?articular, q?quadrate. Scale bars: 200?m (A-F), 100?m (A?-F?). PHENOTYPE:
|
|
(A, D, G) Maximum projection of 5 dpf wild-type group (n = 10). (B, E, H) Maximum projection of 5 dpf nkx3.2 mutant (n = 11) group. (C, F, I) Maximum projection of both groups with coloured voxels representing voxels with statistically significant (p<2.5x10-4) differences in intensity. Magenta shows voxels with higher intensity and cyan shows voxels with lower intensity in nkx3.2 mutant group. Asterisk marks the fusion site. ch?ceratohyal, m?Meckel?s cartilage, ac?auditory capsule, pq?palatoquadrate, rap?retroarticular process. Scale bars: 150?m. |
|
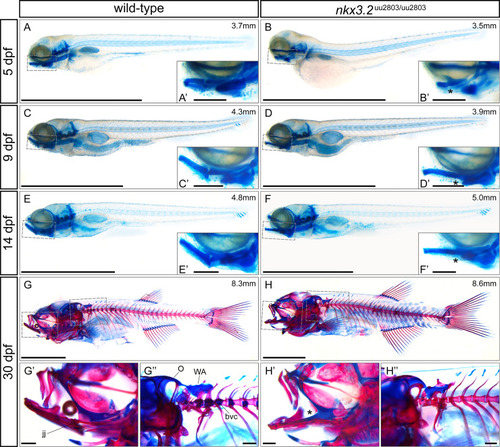
(A-F) lateral views of cartilage-stained wild-types and nkx3.2 mutants at 5, 9 and 14 dpf. (G-H) lateral views of cartilage- and bone-stained wild-types and nkx3.2 mutants at 30 dpf. Boxes in (A-H) indicate the zoomed-in regions in the insets or zoomed-in panels (G?, G?, H?, H?). The measurements in mm refer to standard length (SL). Asterisks indicate the fusion between Meckel?s cartilage and palatoquadrate in the jaw joint. jj?jaw joint, O?occipital region, bvc?basiventral cartilage, WA?Weberian apparatus. Scale bars: 2mm (A-H), 200?m (A?-H? and G?-H?). PHENOTYPE:
|
|
(A, B) lateral view of wild-type and nkx3.2 mutant zebrafish at 90 dpf. (C, D, E) lateral view of the head of wild-type with closed mouth (CM), wild-type with artificially open mouth (AOM), and nkx3.2 mutant. (F, G, H) Anterior view of wild-type with CM, wild-type with AOM, and nkx3.2 mutant. (I, J, K) ventral view of isolated wild-type dentary (CM), wild-type dentary (AOM), and nkx3.2 mutant dentary. (L, M, N) Virtual thin sections (60?m thick, averaged) through the symphysis of the dentaries displayed in I, J, K, respectively. The measurements in mm refer to standard length (SL). Asterisks in (B, E) indicate the fixed open mouth phenotype caused by fused jaw joint. The arrowhead in (E) indicates the downturned ceratohyal phenotype relative to (D). Angles in (I, J) show the flexibility of the symphysis in wild-types, the arrow in K and N indicates the thickened and deformed symphysis phenotype in nkx3.2 mutants. Colour scheme: Red?lower jaw; dark blue?infraorbitals; dark green?ceratohyal and anal fin; blue?basihyal; lilac?branchial arches; dark purple?supraoccipital; lime green?exoccipital and basioccipital; yellow?all remaining craniofacial bones; cyan?cleithrum and pectoral fins, arctic blue?Weberian apparatus; blue violet?vertebrae; green?pelvic fins; bronze?dorsal fin; orange?caudal fin vertebrae and hypurals; magenta?caudal fin rays. AA?anguloarticular, AF?anal fin, BH?basihyal, CF?caudal fin, CH?ceratohyal, DF?dorsal fin, DT?dentary, io3 ?infraorbital 3, jj?jaw joint, le?lateral ethmoid, mx?maxilla, O?occipital region, PCF?pectoral fin, pmx?premaxilla, PVF?pelvic fin, QT?quadrate, WA?Weberian apparatus. Scale bars: 5mm (A, B), 1mm (C-N). PHENOTYPE:
|
|
(A-F) Dorsolateral views of rib-bearing vertebrae in 30?35 dpf, and 90 dpf wild-type and nkx3.2 mutant zebrafish. (A-D) Cartilage- and bone- stained juvenile zebrafish, (E, F) ?CT models. (G, H) ?CT virtual transverse cross-sections of wild-type and nkx3.2 mutant rib-bearing vertebrae at 90 dpf. The measurements in mm refer to standard length (SL). Arrowheads in (B, D, F) indicate the absence of parapophyses on the rib-bearing vertebrae. Parapophyses (pop) are highlighted in green in (E). c?centrum, r?rib. Scale bars: 200?m(A-D), 500?m (E-H). PHENOTYPE:
|
|
Lateral, ventral, anterior, and posterior views of wild-type (A, C, E, G) and homozygous mutant (B, D, F, H) occiput and Weberian apparatus. Cervical vertebrae (CV) 2?4 have been removed in (G, H) for clarity. Asterisk in B, D indicates the absence or severe reduction of lateral process 2 from CV2. Arrowhead in B, D indicates the absence of anterior ramus of tripus on CV3. Arrow in D indicates the V-shaped anteroventral edge of basioccipital. Asterisk in F indicates the posterior fusion of the medial gap between the paired exoccipital bones. Dotted line in G highlights the cavum sinus impar (csi), while the asterisk in H indicates its absence. The measurements in mm refer to standard length (SL). AO?asteriscus otolith, BO?basioccipital, csi?cavum sinus impar, cl?claustrum, EO?exoccipital, lof?lateral occipital fenestrae, lp?lateral process, na?neural arch, os?os suspensorium, r4 ?rib 4, sc?scaphium, sn2 ?supraneural 2, SO?supraoccipital, tr?tripus. Scale bars: 500?m. PHENOTYPE:
|

ZFIN is incorporating published figure images and captions as part of an ongoing project. Figures from some publications have not yet been curated, or are not available for display because of copyright restrictions. PHENOTYPE:
|