- Title
-
A high throughput zebrafish chemical screen reveals ALK5 and non-canonical androgen signalling as modulators of the pkd2-/- phenotype
- Authors
- Metzner, A., Griffiths, J.D., Streets, A.J., Markham, E., Philippou, T., Van Eeden, F.J.M., Ong, A.C.M.
- Source
- Full text @ Sci. Rep.
|
A high through-put zebrafish screen of PHENOTYPE:
|
|
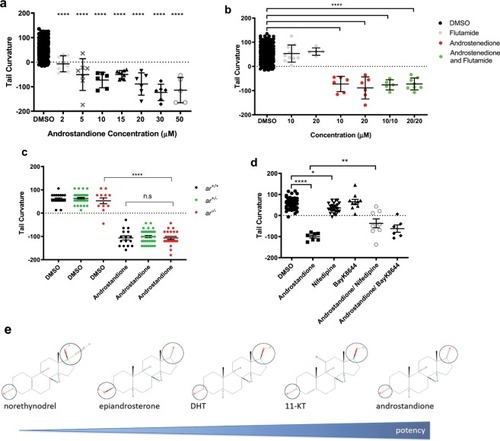
Androgens modulate tail curvature in PHENOTYPE:
|
|
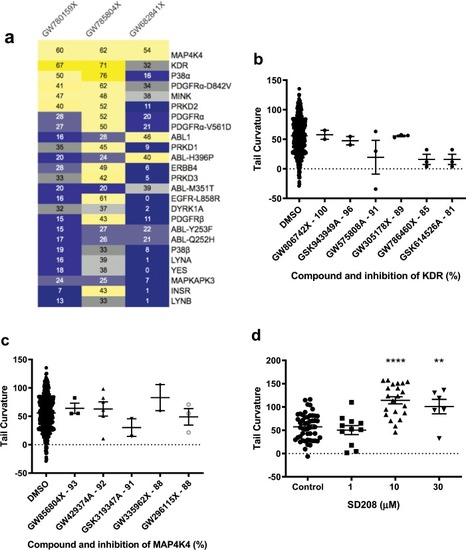
PKIS zebrafish screen revealed four PKIS repressors of the PHENOTYPE:
|
|
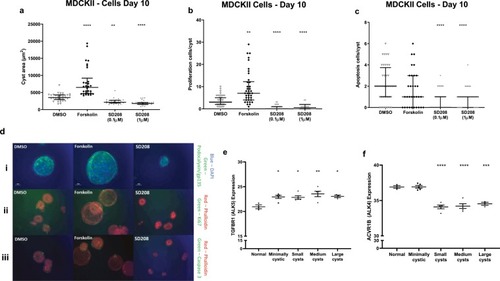
Chemical compounds and PKIS inhibitors identified from zebrafish screen in two cystic cell culture models, MDCKII and Ox161c1. Cyst area of MDCKII ( |
|
Identification of ALK5 as a PHENOTYPE:
|
|
ALK5 as a modulator of cystic growth in cell culture and expression of ALK4/5 in human ADPKD kidneys. Effects of SD208 (1 and 0.1 µM) after 10 days exposure on cyst area ( |
|
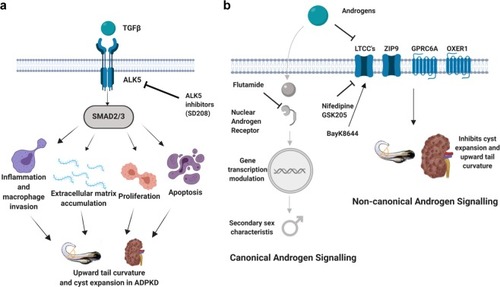
Model for role of ALK5 and non-canonical androgen signaling in ADPKD. ( |

ZFIN is incorporating published figure images and captions as part of an ongoing project. Figures from some publications have not yet been curated, or are not available for display because of copyright restrictions. PHENOTYPE:
|