- Title
-
Highly Efficient CRISPR-Cas9-Based Methods for Generating Deletion Mutations and F0 Embryos that Lack Gene Function in Zebrafish
- Authors
- Hoshijima, K., Jurynec, M.J., Klatt Shaw, D., Jacobi, A.M., Behlke, M.A., Grunwald, D.J.
- Source
- Full text @ Dev. Cell
|
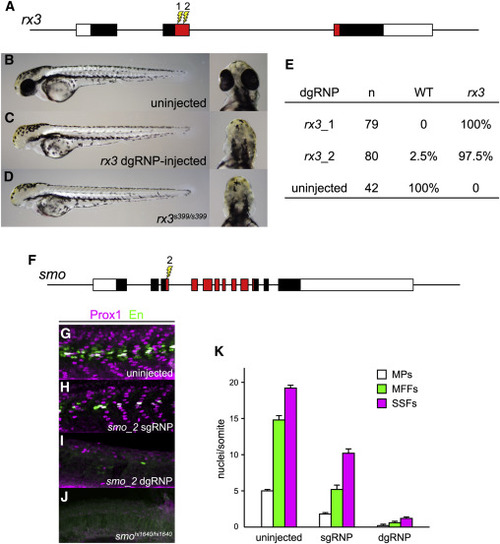
dgRNPs Can Produce Accurate Phenocopies of Null Mutants (A?E) F0 embryos that develop from WT eggs injected with rx3 dgRNPs phenotypically resemble null rx3 mutants. (A) Two dgRNPs were designed to target sites (lightning bolts) within the region of rx3 exon 2 that encodes the DNA binding domain of the Rx3 transcription factor (red-shaded exon sequences). (B) WT, (C) rx3 dgRNP-injected, or (D) homozygous mutant rx3 embryos at 2 dpf. (E) Nearly all embryos injected with dgRNP complexes targeting either sites rx3_1 or rx3_2 lacked eyes and were indistinguishable from homozygous null mutants. (F?K) Injection of dgRNP complexes targeting smoothened (smo) produces F0 embryos that phenotypically resemble null smo mutants. (F) gRNPs were designed to target site smo_2 (lightning bolt) within the 5? portion of the region that encodes the ?Frizzled-domain? of Smoothened (red-shaded exon sequences). (G?K) Development of three distinguishable types of muscle fibers is dependent on Sonic hedgehog signaling and smoothened activity. Nuclei of slow muscle pioneer cells (MPs) express both the Prox1 and Engrailed (En) transcription factors, whereas medial fast fibers (MFFs) only express En, and superficial slow fibers (SSFs) only express Prox1. Shh-dependent muscle fiber nuclei, marked by Prox1 (magenta) and/or En (green) expression, were visualized following immunostaining of (G) uninjected, (H) smo_2 sgRNP-injected, or (I) smo_2 dgRNP-injected WT embryos, or (J) smohi1640 null mutant embryos. (K) The presence of Shh-dependent muscle fibers was determined by analysis of the nuclear staining in a standard set of five trunk somites in each embryo. The average numbers of Shh-dependent muscle fiber nuclei per somite for each embryo is plotted (uninjected [n = 6], smo_2 sgRNP-injected [n = 5], and smo_2 dgRNP-injected [n = 6]). See also Figure S3. |
|
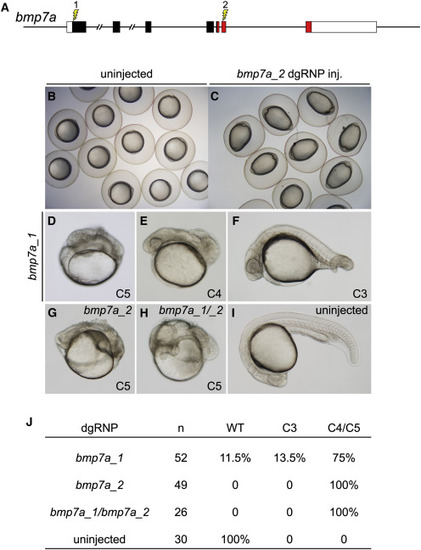
Cell Non-autonomous Phenotypes Can Be Recapitulated Accurately in F0 Embryos (A) dgRNP complexes were designed to target sequences in bmp7a that either encoded the N-terminal signal sequence (bmp7a_1) or the mature ligand domain (red sequences, bmp7a_2) of Bmp7a. (B and C) At the tailbud stage, all embryos injected with bmp7a_2 dgRNP targeting the ligand domain displayed a distinctive ovoid morphology typical of severely dorsalized embryos (C) that is not seen in WT embryos (B). (D?J) Targeting of bmp7a produced dorsalized 24 hpf F0 embryos. Embryos were scored for dorsalized phenotypes as in (Mullins et al., 1996), where C3 indicates intermediate dorsalization and C4 and C5 are considered severe phenotypes. Targeting the sequences encoding the N terminus of Bmp7a generated dorsalized embryos of varying severity (D?F and J). Targeting sequences encoding the ligand domain, or targeting both sequences simultaneously (bmp7a_1/ bmp7a_2) yielded only severely dorsalized embryos (G, H, and J). |
|
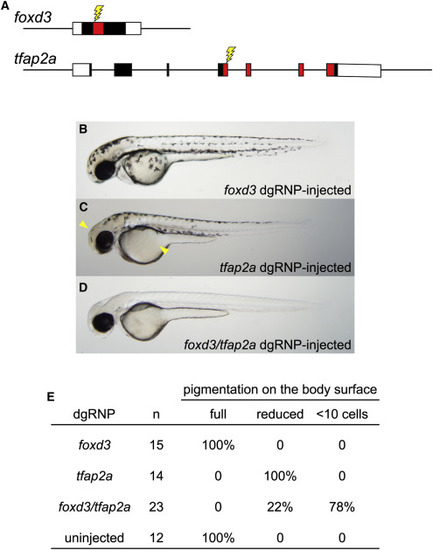
Redundant Gene Functions Can Be Uncovered by Targeting Two Genes Simultaneously with dgRNPs (A) dgRNPs were designed to target sequences (lightning bolts) encoding the DNA binding domains (red-shaded exon sequences) of the transcription factor products of the foxd3 and tfap2a genes. (B?E) Embryos targeted with either foxd3 dgRNPs or tfap2a dgRNPs resemble single mutants: (B) embryos targeted with foxd3 dgRNPs appear morphologically near-normal at 2 dpf, and (C) embryos targeted with tfap2a dgRNPs have moderately reduced pigmentation most notable on the head and yolk (arrowheads). (D) Most doubly injected embryos were devoid of melanophores at 2 dpf, resembling true foxd3;tfap2a double mutants (Arduini et al., 2009). (E) Three-fourths of the doubly injected embryos were devoid of pigment; the remaining embryos had markedly reduced pigmentation, consistent with incomplete loss of function of the targeted genes. |
|
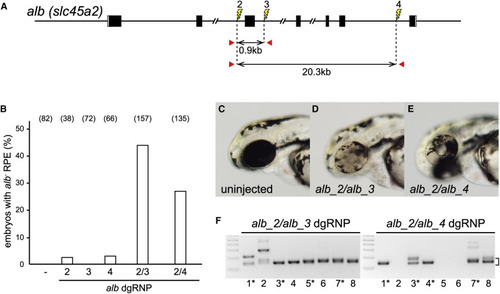
Deletion Mutations Are Readily Generated by Simultaneously Targeting Two Sites on a Chromosome (A) dgRNPs were designed to target sites (lightning bolts) in intron 2 (alb_2), intron 3 (alb_3), or intron 6 (alb_4). The approximate distances between target sites and the locations of primer pairs (red arrows) used to detect deletion mutations that result in joining of target sites are indicated. (B) Uninjected embryos (?) or embryos injected with individual or pairs of dgRNPs targeting alb locus introns were inspected for pigmentless patches of tissue in the RPE at 2?3 dpf. The incidence of embryos with alb mutant tissue is plotted (numbers of scored embryos are indicated). (C?E) Embryos injected with dgRNP pair alb_2 and alb_3 (D) or alb_2 and alb_4 (E) often exhibited large clones of pigmentless mutant tissue not seen in the eyes of uninjected embryos (C). (F) Deletion mutations were detected in the genomes of F0 embryos injected with pairs of dgRNPs by PCR amplification with primer pairs flanking the targeted sites. Junction fragment amplicons of the expected size (bracket) were detected in eight of eight 4 dpf embryos that had been injected simultaneously with alb_2 and alb_3 dgRNPs and in five of eight 4 dpf embryos injected with alb_2 and alb_4 dgRNPs. Junction fragments recovered from starred embryos were sequenced and reported in Figure S4. See also Figures S4?S7. |
|
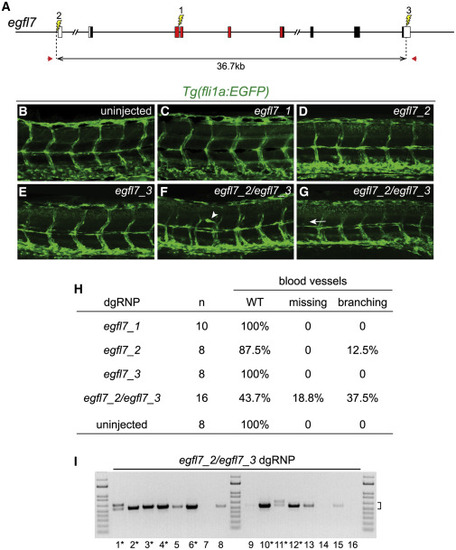
Deletion Mutations Uncover Gene Functions in F0 Embryos (A) The gene structure of egfl7 is indicated. Red regions encode conserved domains of EGFL7, including an EMI (EMILIN) domain, an EGF domain, and a Ca-binding EGF-like domain. dgRNPs designed to target sites (lightning bolts) in exon 3 (egfl7_1), just upstream of the transcription start site (egfl7_2), or in the 3?UTR (egfl7_3) of egfl7. (B?H) Embryo carrying the fli1a:EGFP reporter transgene were scored for morphology of intersegmental vessels after injection with individual egfl7 dgRNPs or a combination of egfl7_2 and egfl7_3 dgRNPs. Embryos injected with individual dgRNPs (C?E) exhibited WT morphology (B), with the exception of a single embryo in which the promoter region had been targeted (H). Embryos targeted simultaneously at the 5? and 3? ends of egfl7 often had vasculature defects (F?H). (I) Genomic DNA from embryos targeted simultaneously at the 5? and 3? ends of egfl7 was amplified with primers (A, red arrows) flanking the egfl7_2 and egfl7_3 sites, revealing junction fragments whose size (bracket) was consistent with the presence of induced deletion mutations. Junction fragments recovered from starred embryos were sequenced and reported in Figure S5. EXPRESSION / LABELING:
|
Reprinted from Developmental Cell, 51(5), Hoshijima, K., Jurynec, M.J., Klatt Shaw, D., Jacobi, A.M., Behlke, M.A., Grunwald, D.J., Highly Efficient CRISPR-Cas9-Based Methods for Generating Deletion Mutations and F0 Embryos that Lack Gene Function in Zebrafish, 645-657.e4, Copyright (2019) with permission from Elsevier. Full text @ Dev. Cell