- Title
-
A genetic model of CEDNIK syndrome in zebrafish highlights the role of the SNARE protein Snap29 in neuromotor and epidermal development
- Authors
- Mastrodonato, V., Beznoussenko, G., Mironov, A., Ferrari, L., Deflorian, G., Vaccari, T.
- Source
- Full text @ Sci. Rep.
|
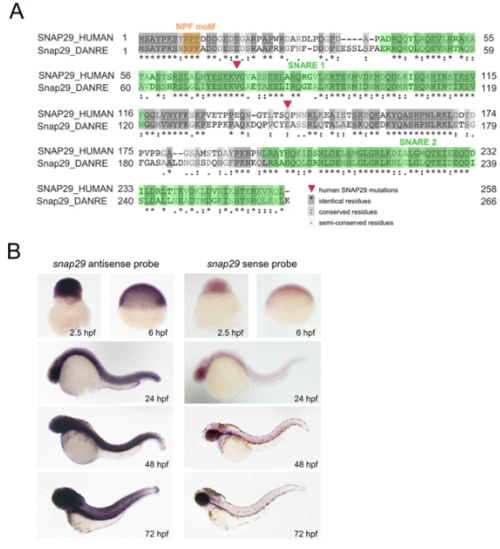
Snap29 conservation and expression in zebrafish. (A) Protein sequence alignment of human and zebrafish Snap29 from UniProt. Red triangles refer to two SNAP29 mutations described in CEDNIK patients. Amino acid residues are shaded according to their degree of conservation, as described in the legend. (B) Whole-mount in situhybridization with snap29 antisense and sense probes on zebrafish embryos at the indicated developmental stages.
EXPRESSION / LABELING:
|
|
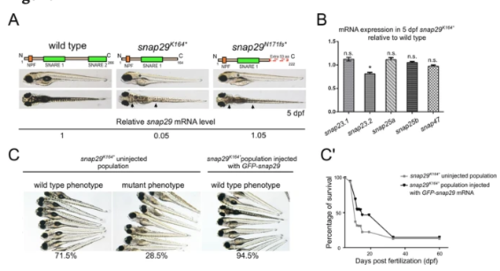
Generation of snap29 mutants and rescue. (A) Lateral and dorsal views of 5 dpf larvae of wild type, ENU snap29K164* mutant and CRISPR/Cas9 snap29N170fs mutants with schematic representation of predicted proteins. Asterisks in lateral views indicate lack of an inflated swim bladder, while arrows in dorsal views point to weaker pigmentation in mutants, compared to wild type. snap29 mRNA relative expression measured in extracts of 5 dpf wild type, snap29K164* and snap29N170fs larvae, and normalized on gapdh is reported below the images. snap29K164* mutants show a reduction of snap29 mRNA of about 95% compared to wild type, while no significant reduction is detectable in snap29N170fs. (B) mRNA expression of snap23.1, snap23.2, snap25a, snap25b and snap47 measured in extracts of 5 dpf snap29K164* relative to wild type. gapdh is used as normalizer. The bars in the graph show means and standard errors of the mean (SEM) of three technical replicates. P-values were computed by multiple t-test. *P ≤ 0.05. (C) Groups of 5 dpf larvae from the progeny of snap29K164* heterozygous mating. Uninjected larvae were classified according to the pigmentation phenotype. The snap29K164* uninjected population is composed by 71.5% of wild type phenotype larvae and by 28.5% of mutant phenotype larvae. The snap29K164*injected population consists of the 94.5% of phenotypically wild type larvae, demonstrating that GFP-snap29 mRNA injection rescues the mutant phenotype. (C’) Survival curve of snap29K164* populations uninjected and injected with GFP-snap29 mRNA. The percentage of survival refers to alive zebrafish larvae or adults recovered over time from the initial number of fertilized embryos. Injected snap29K164* mutants show an increase of survival compared to uninjected animals. |
|
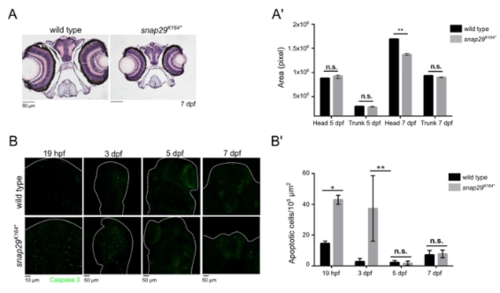
snap29 mutant as a CEDNIK disease model. (A) Hematoxylin-eosin stained paraffin sections of heads of 7 dpf larvae. Note the reduced size of the head in snap29K164* mutant animals compared to wild type. (A’) Quantification of the area of head and of the trunk sections measured in wild type and snap29K164* mutant respectively at 5 dpf and 7 dpf. At 7 dpf, snap29K164* mutants show a significant reduction of head area compared to wild type. Bars in the graph show means and standard deviations. P-values were obtained by unpaired t-test. **P ≤ 0.01, n = 3. (B) Comparable maximum projections of wild type and snap29K164*mutant heads at 19 hpf, 3 dpf, 5 dpf and 7 dpf stained with anti-cleaved Caspase 3 to reveal presence of apoptotic cells. White dashed-lines were drawn to highlight head morphology. (B’) Quantification of the number of apoptotic cells per 105 μm2, measured in wild type and snap29K164* mutants respectively at 19 hpf, 3 dpf 5 dpf and 7 dpf. 19 hpf and 3 dpf snap29K164* mutants show a significant increase of apoptotic cells compared to wild type. The bars in the graph show means and standard deviations. P-values were obtained unpaired t-test with Welch’s correction. *P ≤ 0.05, **P ≤ 0.01, n = 2–9. PHENOTYPE:
|
|
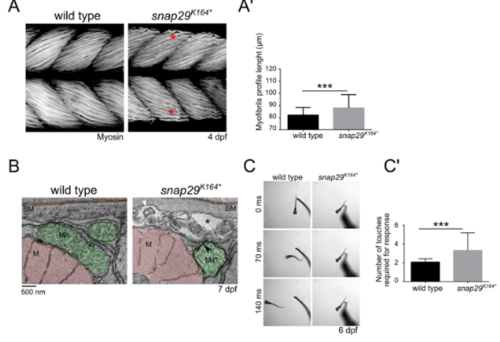
Snap29 supports correct muscle development and function. (A) Lateral views of muscles in the trunk of 4 dpf wild type and snap29K164* mutant larvae, stained with anti-Myosin heavy chain antibody. Compared to wild type, snap29K164* mutants present less compacted and ordered filaments (red asterisk). (A’) Quantification of the length of the profile of superficial myofibrils measured in wild type and snap29K164* mutant 4 dpf larvae. snap29K164* mutants show a significant increase in myofibril length compared to wild type. The bars in the graph show means and standard deviations. P-values were derived from Mann-Whitney test. ***P ≤ 0.001, n = 41–45. (B) Electron microscopy cross-sections of muscle fibers. snap29K164*contain MLO (white arrowhead) within extracellular spaces (asterisks) just beneath the basement membrane and a mitochondria surrounded by a double membrane (black arrowhead), which are not present in wild type animals. M: myofibrils, Mit: mitochondria, BM: basement membrane. (C) Selected frames from movies of a wild type and a snap29K164*mutant larva at 6 dpf recorded for 1 minute after a touch stimulus on the tail. (C’) Quantification of the number of touches required to evoke an escape response. Most of the analyzed snap29K164* mutants require two or more touches to respond. The graph reports means and standard deviations. P-values were obtained by Mann-Whitney test. ***P ≤ 0.001, n = 6. v |
|
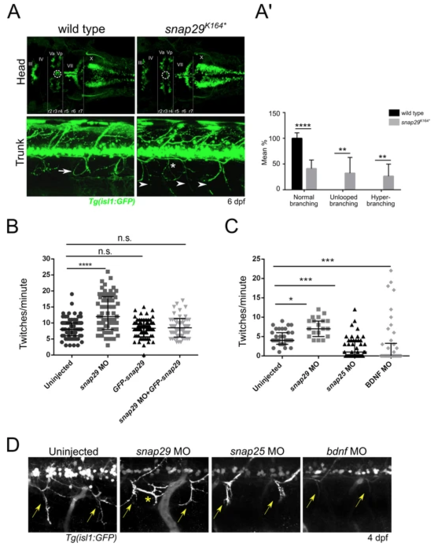
Snap29 impairment causes neuro-developmental defects. (A, upper row) Head dorsal views of the motor neuron reporter Tg(isl1:GFP) in a wild type and a snap29K164* mutant larva at 6 dpf. (r) rhombomere, (III), (IV), (Va) anterior, (Vp) posterior, (VII), (X) cranial nerves. snap29K164* mutant larvae lack a group of nuclei (white dashed circles) located between the third and fourth rhombomere (r3, r4). (A, lower row) Lateral views of trunks of Tg(isl1:GFP)-expressing wild type and snap29K164* mutants at 6 dpf. Compared to the “loop” structures observed in wild type (white arrow), snap29K164* mutants present an altered motor neuron projection pattern (arrowheads), and extra branching (white asterisk). (A’) Quantification of motor neuron branching in wild type and snap29K164* mutant larvae at 4, 5 and 6 dpf. Compared to wild type, snap29K164* mutants larvae show a significant increase of unlooped branches and hyperbranching. The bars show means and standard deviations of the percentage of the phenotypic categories. P-values were obtained by unpaired t test with Welch’s correction. **P ≤ 0.01, ****P ≤ 0.0001, n = 6–10. (B) Quantification of the number of twitches per minute performed respectively by 26 hpf embryos treated as described in figure. snap29 morphants show an increase in the number of twitches per minute compared to wild type embryos. Co-injection of snap29 Morpholino with GFP-snap29 mRNA rescues the increased number of twitches per minute observed in snap29 morphants, while GFP-snap29 mRNA injection per se has no effect on twitching frequency. The graph shows medians, 25th and 75th percentiles. P-values were obtained by Kruskal-Wallis, ****P ≤ 0.0001, n = 69–73. (C) Quantification of the number of twitches as in B. snap29 morphants show an increase twitches frequency per minute compared to wild type embryos, while both snap25and bdnf morphants show a significant decrease. The graph shows medians, 25th and 75thpercentiles. P-values were derived from Kruskal-Wallis, *P ≤ 0.05 ***P ≤ 0.001, n = 20–44. (D) Motor neuron projections in 4 dpf Tg(isl1:GFP)-expressing uninjected larvae and in snap29, snap25 and bdnf morphant larvae. Uninjected embryos show normal motor neuron projection towards the ventral part of the trunk (yellow arrows). snap29 morphants show less elongated projections (yellow arrows) and extrabranching (yellow asterisk), while both snap25 and bdnf morphants show truncated projections. |
|
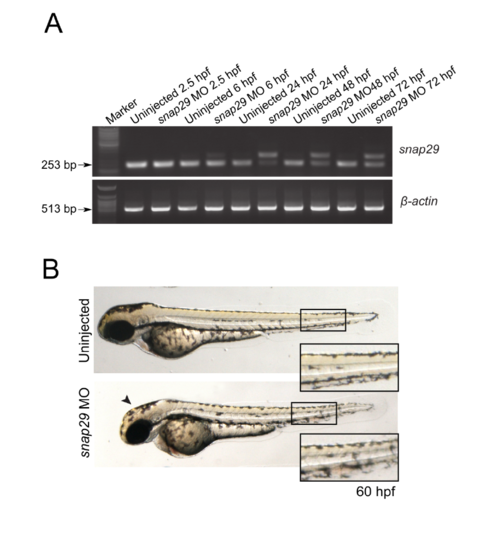
(A) RT-PCR performed to assess snap29 transcript level using cDNA from embryos uninjected or injected with a splicing block Morpholino against snap29 (SB MO snap29) at the indicated hours post fertilization (hpf). β-actin was used as normalizer. (B) 60 hpf uninjected embryos or embryos injected with SB snap29 MO. Morphants display lighter pigmentation at the level of the head (arrowheads) and misaligned melanocytes in the tail contour (high magnification in insets), compared to uninjected embryos. PHENOTYPE:
|

ZFIN is incorporating published figure images and captions as part of an ongoing project. Figures from some publications have not yet been curated, or are not available for display because of copyright restrictions. PHENOTYPE:
|
|
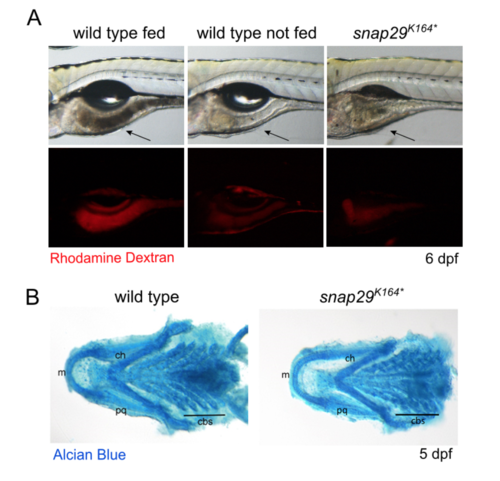
Impaired feeding in snap29 mutants (A) Lateral views of 6 dpf larvae in which guts are indicated by an arrow. Larvae were fed Rhodamine Dextran-containing food that allow detecting the presence of food in the gut. The gut of a snap29K164* mutant animal appears as empty as that of unfed wild type larvae, when compared to fed ones. (B) Ventral views of the splanchnocranium of 5 dpf larvae. Cartilage staining with Alcian Blue did not highlight any difference in organization and/or differentiation between wild type and snap29K164* mutants. m: Meckel’s cartilages, ch: ceratohyal, pq: palatoquadrate, cbs: ceratobranchials. |
|
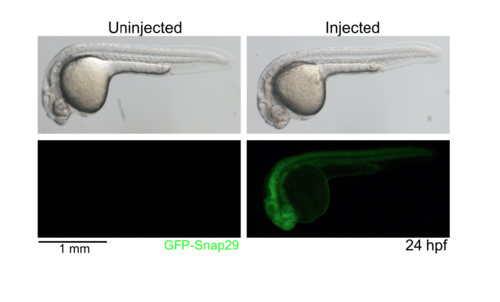
GFP-snap29 expression in zebrafish 24 hpf embryos uninjected and injected at single-cell stage with GFP-snap29 mRNA. Bright-field and fluorescent images are shown, indicating that the mRNA is translated into ectopic protein ubiquitously. |

ZFIN is incorporating published figure images and captions as part of an ongoing project. Figures from some publications have not yet been curated, or are not available for display because of copyright restrictions. |

ZFIN is incorporating published figure images and captions as part of an ongoing project. Figures from some publications have not yet been curated, or are not available for display because of copyright restrictions. |

ZFIN is incorporating published figure images and captions as part of an ongoing project. Figures from some publications have not yet been curated, or are not available for display because of copyright restrictions. |