- Title
-
Collagen COL22A1 maintains vascular stability and mutations in COL22A1 are potentially associated with intracranial aneurysms
- Authors
- Ton, Q.V., Leino, D., Mowery, S.A., Bredemeier, N.O., Lafontant, P.J., Lubert, A., Gurung, S., Farlow, J.L., Foroud, T.M., Broderick, J., Sumanas, S.
- Source
- Full text @ Dis. Model. Mech.
|
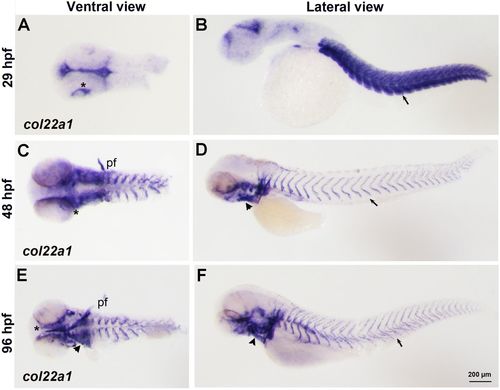
col22a1 expression pattern analyzed by WISH. (A-F) col22a1 expression in flat-mounted embryos is viewed in ventral and lateral direction at 29?hpf (A,B), 48?hpf (C,D) and 96?hpf (E,F). The expression was detected in the somitic muscle (arrow, B) and at the myotendinous junction (arrows, D,F). col22a1 expression was also detected around the eyes (asterisks), in the ears, pectoral fins (pf) and pharyngeal arches (arrowheads). |
|
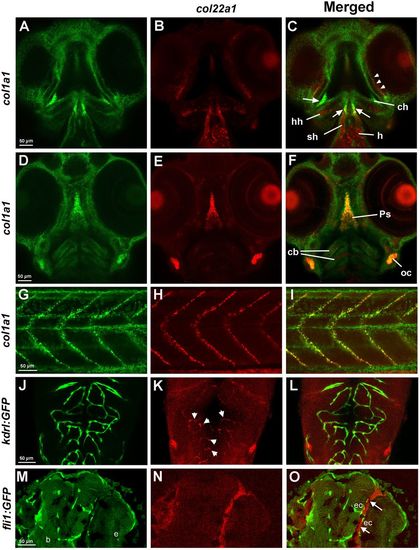
col22a1 expression in the head and trunk region analyzed by FISH and FISH/immunofluorescence. (A-I) Expression of col22a1 (red) and col1a1 (green) at 3?dpf, which commonly marks connective tissue. Expression was analyzed by HCR. In the transverse confocal sections of the cranial tissue (ventral view, A-F), col22a1 expression is apparent in the periocular tissue (arrowheads, C), craniofacial muscle, including sternohyoideus (sh) and hyohyoideus (hh) muscles, parasphenoid bone (Ps), the heart (h), presumptive chondrocytes within ceratobranchial (cb) and ceratohyal arches (ch), and connective tissue within the developing ear (otic capsule, oc). Note that the expression of both markers overlaps within the parasphenoid bone, otic tissue, connective tissue around the eyes (arrowheads, C), and the tendon/ligament attachment sites of the sternohyoideus and hyohyoideus muscles (arrows, C). Within the myotendinous junction in the trunk region (G-I), all col22a1-expressing cells were also positive for col1a1 expression. Note that col1a1 also labels keratinocytes within the epidermis. (J-L) col22a1 expression analyzed by HCR partially overlaps with vascular endothelial expression of kdrl:GFP. Cranial vasculature of whole-mounted embryos at 3?dpf was imaged dorsally by confocal microscopy. Anterior is to the top. Maximal-intensity projection of selected slices is shown. (M-O) Longitudinal cranial sections of embryos stained by WISH/immunofluorescence for col22a1 (red) and vascular endothelial fli1:GFP (green) at 72?hpf. col22a1 expression is apparent within the perivascular stromal cells (arrows, K). b, brain; e, eye; ec, endothelial cells. Note that these sections are located more ventrally compared with the vascular col22a1 expression shown in J-L. EXPRESSION / LABELING:
|
|
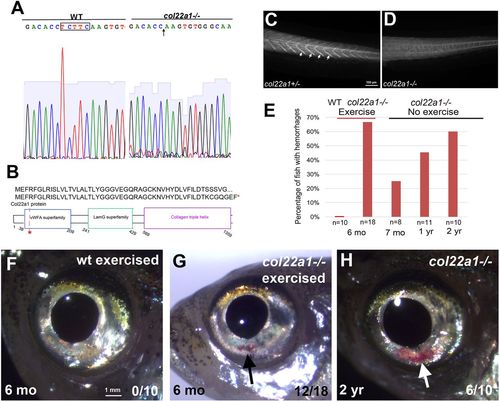
col22a1?/? mutant adults exhibit hemorrhages. (A,B) Generation of col22a1 zebrafish mutant line using TALEN. Sanger sequencing data of homozygous col22a1 mutant show that there is a 5?bp deletion TCTTC (boxed; arrow in the right panel) that results in a frame shift (A) and a premature stop codon in the protein sequence (B). (C,D) Immunostaining against zebrafish Col22a1 at 3?dpf in col22a1+/? and col22a1?/? embryos. Staining is absent at the myoseptae in the col22a1?/? mutant (arrows) compared with the col22a1+/? embryo, suggesting that the mutation is likely null. Selected sections from Z-stack were merged using the Extended Focus feature within the Axiovision software (Zeiss). (E) The frequency of hemorrhages increases with age in col22a1?/? mutants, while exercise caused col22a1?/? mutants to exhibit the phenotypes at an earlier age. (F,G) A 6-month-old col22a1?/? adult experiences visible blood accumulation in the eyes after a 3-week course of intermittent forced exercise (black arrow, G), whereas the WT adult does not (F). (H) Without exercise, a 2-year-old col22a1?/? adult also exhibits visible blood accumulation in the eyes (white arrow). EXPRESSION / LABELING:
PHENOTYPE:
|
|
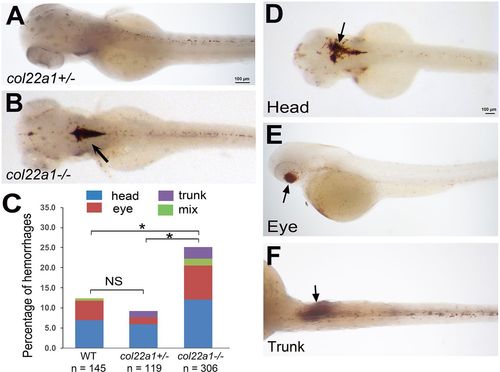
Homozygous col22a1?/? embryos show increased percentage of hemorrhages when raised at increased temperature. Embryos were stained with o-Dianisidine at 3?dpf. (A) The heterozygous col22a1+/? embryo shows small scattered blood cells in the head, while a large hemorrhage is apparent within the brain in the col22a1?/? embryo (arrow, B). (C) Compared with the WT and col22a1+/? embryos, a significantly higher percentage of col22a1?/? mutant embryos exhibit hemorrhages after heat stress. *P<0.005, determined by chi-square test. Embryo numbers were combined from three independent experiments (two for col22a1+/? embryos). The scoring numbers from each experiment are shown in Table S1. (D-F) Examples of hemorrhages (arrows) at different locations in heat-stressed col22a1?/? embryos. PHENOTYPE:
|
|
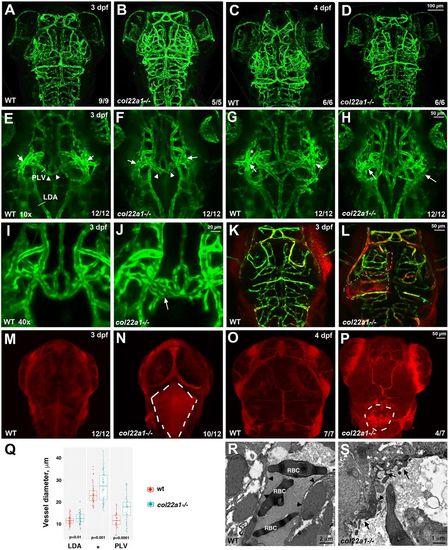
Homozygous col22a1?/? mutant embryos show dilated and dysmorphic cranial vessels and have increased vascular permeability. (A-D) Vascular patterning is not affected in col22a1 mutants at 3?dpf and 4?dpf. Confocal maximal-intensity projections of live kdrl:GFP embryos, dorsal view, anterior is to the top. (E-J) Dilated and dysmorphic vessels are observed in col22a1 mutants. Note the dilated and fused vessels at the vascular branch point in the periocular region (arrows) and the dilated and dysmorphic palatocerebral vein (PLV, arrowheads). Higher magnification (40�) images of the PLV are shown in I and J. Note that a portion of the PLV forms a plexus (arrow, J), which is not observed in WT embryos. Confocal maximal-intensity projections of selected slices of live kdrl:GFP embryos are shown, dorsal view, anterior is to the top. Cross-sections in the PLV, lateral dorsal aorta (LDA, E) and at the periocular branch point (asterisk, G) label the vessels selected for diameter measurement in Q. Different Z-stack projections of the same embryos are shown in E and G, and in F and H; I and J show different embryos. (K-P) Microangiography analysis of vascular permeability. FluoSpheres polystyrene microspheres (red, K,L) or low Mw (10?kDa) TRITC-dextran (red, M-P) were injected into the circulatory system of kdrl:GFP-positive WT and col22a1?/? embryos. Dorsal views of merged images (red, dextran/microspheres; green, kdrl:GFP) show rhodamine dye leakage at the brain ventricle and choroid plexus (dashed lines, N,P) and microsphere accumulation outside the vasculature (dashed lines, K,L) in col22a1?/? embryos at 3?dpf and 4?dpf. Maximal-intensity projections of confocal Z-stacks are shown. (Q) Vessel diameters of the LDA (E), PLV and the periocular branching point (asterisk, G) of heat-stressed col22a1 mutants and wild-type (wt) embryos at 3?dpf. The measurements were made at three randomly selected points for each vessel in 12 mutant and wt embryos obtained in two independent experiments. LDA and periocular vessels were measured at both left and right sides where possible. The total number of measured points n=57 for LDA (s.d.=1.9 and 2.5 for wt and mutant embryos, respectively), n=54 and 57 for the periocular vessel (s.d.=4.3 and 6.5 for wt and mutant embryos, respectively), and n=36 for the PLV (s.d.=3.2 and 5.9 for wt and mutant embryos, respectively). P-values were calculated using Student's t-test between wt and col22a1?/? embryos for all measured points in all embryos. (R,S) TEM analysis of cranial vascular endothelium. Note the highly dysmorphic vascular endothelium (arrows, S) in col22a1 mutants compared with normal vascular endothelium in WT embryos (arrowheads, R). RBC, red blood cells. EXPRESSION / LABELING:
PHENOTYPE:
|
|
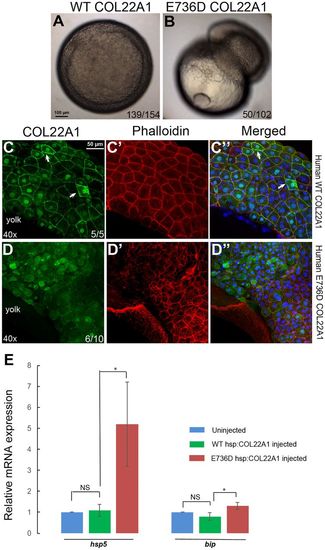
Overexpression of human E736D COL22A1 mRNA causes cell detachment during gastrulation, while the protein is retained in the cytoplasm leading to an increase in ER stress. (A,B) Cells fail to undergo epiboly and detach from the yolk in the E736D COL22A1 mRNA-injected embryo (B) during gastrulation compared with the human WT COL22A1 mRNA-injected embryo (A). (C-D?) Human E736D COL22A1 protein is retained in the cytoplasm during gastrulation, whereas WT COL22A1 is secreted into the extracellular space (arrows). Confocal microscopy analysis of human COL22A1 protein immunofluorescence (green, C,D) and phalloidin staining (red, C?,D?). Blue, nuclear DAPI staining. (E) Aggregation of human mutant COL22A1 in the cytoplasm induces ER stress. qPCR confirms the elevation of ER stress markers hsp5 and bip in human E736D COL22A1 mRNA-injected embryos compared with the human WT mRNA-injected embryos. Expression in the uninjected samples was normalized to 1. *P<0.05, determined using Student's t-test. Error bars represent s.d. |