- Title
-
The ATPase-dependent chaperoning activity of Hsp90a regulates thick filament formation and integration during skeletal muscle myofibrillogenesis
- Authors
- Hawkins, T.A., Haramis, A.P., Etard, C., Prodromou, C., Vaughan, C.K., Ashworth, R., Ray, S., Behra, M., Holder, N., Talbot, W.S., Pearl, L.H., Strähle, U., and Wilson, S.W.
- Source
- Full text @ Development
|
slou45 mutants have disorganised or missing expression of sarcomeric proteins. (A-J) Lateral views of muscle fibres in wild-type (Sib) (A,C,E,G,I) and slou45 (B,D,F,H,J) zebrafish embryos of ages shown bottom left and with reagents/antibodies shown bottom right. (A,B) F-Actin labelling with phalloidin showing a regular arrangement of fibrils in wild-type muscle fibres, whereas the slou45 mutant fibrillar organisation is disrupted. (C,D) Immunohistochemistry for α-Actinin (green) marks the Z-disc, here combined with phalloidin (red); the merge appears yellow. Z-discs are present in the slou45 mutant (white arrowheads, D) but are disordered: the distance between Z-discs is irregular (blue arrowheads) and Z-discs from neighbouring fibrils are not in register with each other (inset, D). Z-discs are flanked by Actin filaments in both siblings and mutants. (E,F) Titin labelling (green) using an antibody that marks a region of the molecule around the Z-disc, counterstained with phalloidin (red). (G-J) Immunohistochemistry for MHC (slow muscle Myosin, F59, red in G,H; pan-Myosin A4.1025, red in I,J). In the slou45 muscle cells, anti-MHC staining is severely reduced and lacks organisation. (K) Western blots using anti-MHC antibody F59 and γ-Tubulin (γ-Tub) as a loading control on lysates of slou45 and slotu44c mutant and sibling embryos at 30 hpf and 48 hpf. MHC protein levels are reduced in mutants of both alleles. Scale bars: 20 μm in A,B,G,H; 10 μm in C-F; 8 μm in I,J. PHENOTYPE:
|
|
Sarcomeres in slou45 mutant myofibrils lack thick filaments. (A-H) Electron micrographs of sarcomeres from wild-type (Sib) (A,C,E,G) and slou45 mutant (B,D,F,H) myofibrils at the ages shown bottom left. (A,B) Caudal myotomes of 24 hpf zebrafish embryos. White arrowheads (A) show immature Z-discs with poorly positioned thick and thin filaments. In slou45 mutants, bundles of thin filaments are present with putative early Z-discs (B, arrowheads). Very rarely, structures of dimensions approximating to those of thick filaments were present (box, B). (C,D) Rostral myotomes of 24 hpf embryos. The wild type has recognisable sarcomeres with almost straight Z-discs (white arrowhead, C) and more fully formed sarcoplasmic reticulum with triads (black arrowhead, C). In the slou45 mutant, I-Z-I brushes are formed [Z-discs (white arrowheads, D) flanked on either side by thin filaments], and a few putative thick filaments are evident (box, D). (E-H) At 48 hpf, wild-type muscle fibres are packed with mature myofibrils (E,G), possessing mature triads at each Z-disc (black arrowhead, G). Mutant muscle fibres contain no thick filaments but numerous I-Z-I brushes are evident (F,H). The Z-lines are surrounded by an electron-dense region (bracket, H; F); beyond this region the thin filaments have light striations on them (see inset, H). Triads are present, although less common (black arrowhead, H). (I) Schematic summarising the principal differences between slou45 mutant and sibling sarcomere ultrastructure. At early stages of myofibrillogenesis (top), mutants have less numerous and malformed thick filaments (red), and I-Z-I brushes (green and blue) are present but less well aligned than siblings. By 48 hpf, wild-type sarcomeres are fully formed, whereas in mutants only misaligned and mis-spaced I-Z-I brushes with striated thin filaments are present (blue). T-tubules and sarcoplasmic reticulum are shown in yellow and purple, respectively. Scale bars: 500 nm in A-D,G,H; 2 μm in E,F; 100 nm in H inset. |
|
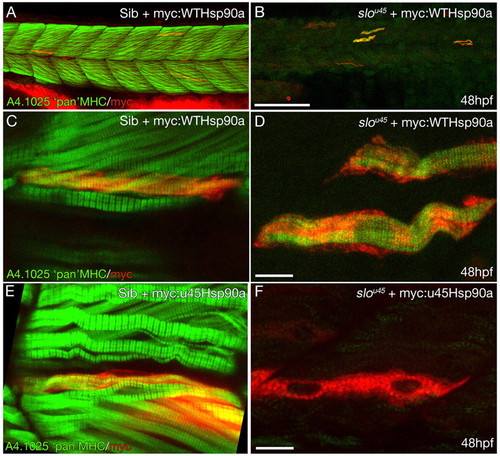
Wild-type Hsp90a rescues thick filament formation in slou45 muscle cells whereas Hsp90au45 does not. (A-D) Muscle fibres from a slou45 mutant (B,D) and wild-type (Sib) (A,C) injected with DNA encoding myc:WTHsp90a double stained for myc (red) and MHC (green). MHC staining in siblings decorates the cells with organised bands (C) indicative of mature A-bands. In the slou45 mutant, striations in the MHC staining are only evident in myc+ cells (D). (E,F) Muscle fibres in sibling embryos injected with myc-tagged hsp90au45 DNA show organised MHC staining of myc+ cells (E). Myc+ cells in slou45 mutants injected with the same construct do not show organised myofibrils (F). Scale bars: 100 µm in A,B; 10 μm in C-F. |
|
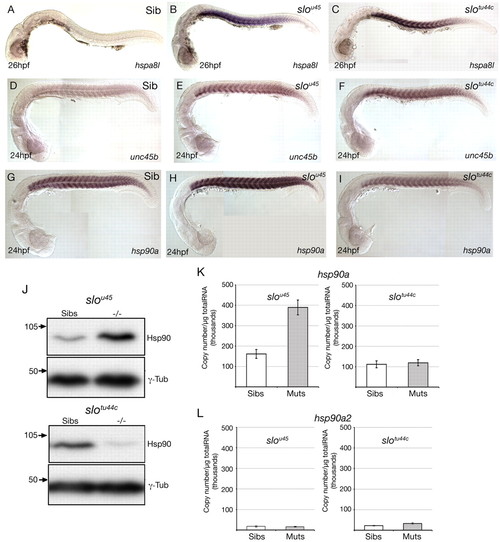
Abrogation of hsp90a but not hsp90a2 function leads to upregulation of genes encoding proteins likely to be involved in sarcomere assembly. (A-I) Lateral views, at the stage indicated bottom left, showing expression of the genes indicated bottom right in wild-type (Sib) and mutant zebrafish embryos. Note that although hspa8l and unc45b are upregulated in the slotu44c allele, hsp90a itself is not. (J) Western blots using lysates of slou45 and slotu44c mutant and sibling embryos. More Hsp90 protein was present in slou45 mutant lysates than in the corresponding siblings. By contrast, slotu44c mutant lysates contained less Hsp90 protein than siblings. (K,L) Quantitative PCR (qPCR) analyses of levels of hsp90a and hsp90a2 expression in wild type (Sibs), slou45 and slotu44c mutants (Muts). Absolute expression levels of hsp90a (normalised to μg of total RNA) were significantly increased (K; P=0.002, n=5) in slou45 mutants compared with siblings; levels of expression of hsp90a in slotu44c mutants were unchanged compared with siblings (K; P=0.75, n=3); and levels of expression of hsp90a2 were not significantly different between slou45mutants and siblings nor between slotu44c mutants and siblings (L; P=0.813, n=5 and P=0.074, n=3, respectively). EXPRESSION / LABELING:
|
|
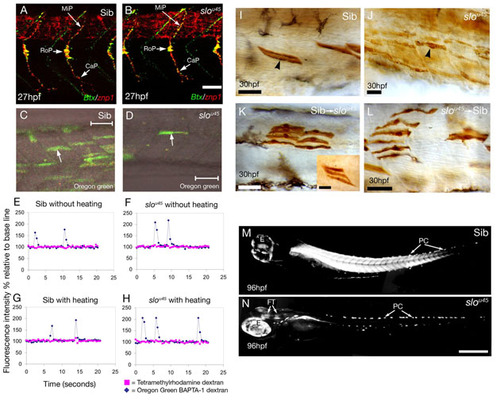
The phenotype of sloθ45 mutants is intrinsic to the muscle fibres. (A,B) Confocal projections of wild-type (A) and slou45 (B) embryos stained with alpha-bungarotoxin (Btx, green) to mark motor endplates and with the ZNP1 antibody to label primary motoneuron axons (znp1, red). The trajectories of the three primary motoneuron axons (MiP, medial; RoP, rostral; CaP, caudal) in the slou45 mutant (B) are similar to its sibling (A) with multiple bungarotoxin-positive puncta (neuromuscular junctions) associated with the axons in both cases. The only defect we detected was a slight reduction in the branching of some CaP axons in the ventral myotome of slou45 mutants. Bungarotoxin-positive spots are also evident in the furrow between the myotomes as noted previously (Behra et al., 2002; Lefebvre et al., 2007). (C-H) Calcium fluxes in the muscle of slou45 mutants are normal. (C,D) Muscle fibres (arrows) labelled with the calcium-sensitive dye Oregon-Green 488 BAPTA-1 dextran co-labelled with tetramethylrhodamine-dextran (to detect calcium-insensitive fluorescent changes) in myotomes of a slou45 mutant (D) and a sibling (C). Recordings of fluorescence in fibres from a mutant (F,H) and a sibling (E,G) with and without an external stimulus of heating show no differences except that transients are slightly larger in the mutant because movement in wild types took the dye-containing cells out of focus. To control for this movement, the ratio of peaks in calcium and rhodamine fluorescence was calculated (see Table S1). (I-L) Muscle fibres of slou45 mutants have abnormal morphologies. (I,J) Mosaic labelling of muscle cells by injection of biotinylated dextran and ABC Kit (Vector labs) and DAB in a sibling (I) and a slou45 mutant (J). slou45 muscle fibres have an inconsistent, uneven and wavy morphology lacking crisp attachments at the myotome boundaries, in contrast to the regimented, straight and crisply attached appearance of muscle fibres in siblings. (K,L) The slou45 mutation has a cell-autonomous effect. Dextran-labelled cells were transplanted from/to wild-type and slou45 embryos at 30% epiboly, fixed and stained at 24-36 hpf. Most wild-type sibling muscle cells show near-normal morphology in a slou45 mutants (K), although isolated wild-type cells tended to have a slightly defective morphology - when the cells grouped together (inset), their morphology is more normal. slou45 mutant cells in a wild-type sibling show abnormal morphology (L) similar to non-transplanted mosaically labelled mutant cells (compare with J). This suggests that the slo phenotype is primarily due to a cell-autonomous defect with only minor, likely secondary, non-autonomous defects. (M,N) Lateral views of trunk muscles of a slou45 mutant (N) and a sibling (M) visualised with polarised light. Muscle fibres in the wild type are highly birefringent, whereas in the slou45 mutant, the muscle completely lacks birefringence. Birefringence is still seen in the pigment cells (PC), eye (E) and in fibre-tracts of the brain (FT) and appears brighter in mutants due to longer photographic exposure. Scale bars: 20 μm in A,B,I-L; 50 μm in C,D; 200 μm in M,N. PHENOTYPE:
|
|
slotθ44c mutants have a similar but slightly less severe muscle structure defect than sloθ45 mutants. (A,B) Muscle fibres double stained for primary sarcomeric components Actin and Myosin using phalloidin (red) and A4.1025 anti-MHC antibodies (green) in slotu44c mutants and siblings. slotu44c mutant fibres have some apparent organisation in both Actin and anti-MHC staining; however, compared to the sibling, the staining is disordered. Boxes indicate approximately similar fields of view shown in the electron micrographs shown in C,D. (C-F) Ultrastructural analysis of wild-type and slotu44c muscle fibres shows that the slotu44c fibres contain electron-dense bodies (arrowheads, D) located approximately between adjacent I-Z-I brushes. These bodies appear to be thickest in the region equidistant between adjacent Z-lines tapering away towards the Z-lines suggesting deformed A-bands. These electron-dense bodies were sometimes found to contain disordered thick-filament-like structures (arrows, F). Scale bars: 10 μm in A,B; 2 μm in C,D; 500 nm in E,F. PHENOTYPE:
|
|
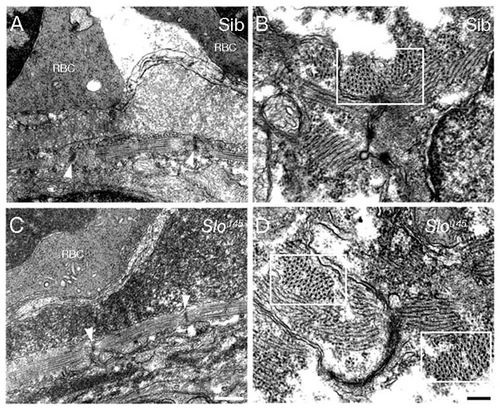
Heart muscle ultrastructure is unaffected by the sloθ45 mutation. Transverse TEM sections through the heart tube at 48 hpf reveal a thin myofibrillar band in the heart wall in both siblings (A) and slou45 mutants (C). Z-disc to Z-disc distances show no obvious differences between the mutant and sibling (arrowheads, A and C). Longitudinal sections reveal some myofibrils in cross section (boxes, B and D) and in these cases it is clear that the myofibrils contain both thick and thin filaments. RBC, red blood cell. Scale bars: 500 nm in A,C; 200 nm in B,D. PHENOTYPE:
|
|
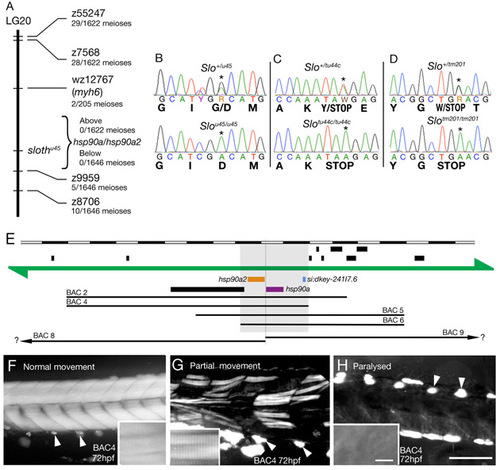
The slo phenotype is caused by mutations in hsp90a. (A) Schematic of LG20 around the slou45 mutation. slou45 was mapped to a region between SSLP markers z7568 and z9959 (SSLP primers sequences from MGH: http://zebrafish.mgh.harvard.edu/zebrafish/index.htm). Genes/ESTs located between these markers were identified and enzyme-scorable SNPs within these genes were found by sequencing genomic DNA from mutants and siblings using primers designed from public genome sequence (www.ensembl.org). These SNPs were tested for linkage: hsp90a mapped very close to the slo locus. (B-D) Sequence traces from embryos of genotypes as shown above each trace illustrating mutations (asterisks). Both hsp90a and hsp90a2 were sequenced from genomic DNA for all genotypes; primers were designed using the LIMSTILL program (http://limstill.niob.knaw.nl/); all primer sequences available on request. (E) Diagram of the genomic region around the slo locus showing BACs that all rescued the phenotype of slou45 mutants. The shaded area indicates the genomic region responsible for the phenotype found through use of overlapping BACs. The three genes within are: hsp90a2, orange; hsp90a, purple; si:dkey-241I7.6, blue. Black boxes are neighbouring genes. (F-H) Polarised light micrographs of trunk muscle from BAC4 rescue experiments. Panels show normally moving (F), paralysed (H) and partially moving (G) embryos. Striated birefringence, where present, is shown in insets. Arrowheads point to birefringent pigment cells. Scale bars: ruler divisions in E, 20 kb; 100 μm in F-H, 5 μm in insets. |
|
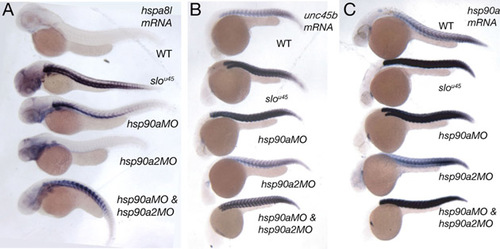
hsp90a morphants show upregulation of chaperone transcripts whereas hsp90a2 morphants do not. Lateral views of wild-type, slou45, hsp90a morphant, hsp90a2 morphant and hsp90a/hsp90a2 double morphants (condition indicated above individual embryos) examined for expression of hspa81 (A), unc45b (B) and hsp90a (C). Abrogation of hsp90a2 activity had little effect on gene expression. The bent body of the hsp90a2 morphants might not be a specific phenotype as an alternative hsp90a2 MO produced embryos with normal morphology (see Materials and methods). EXPRESSION / LABELING:
|