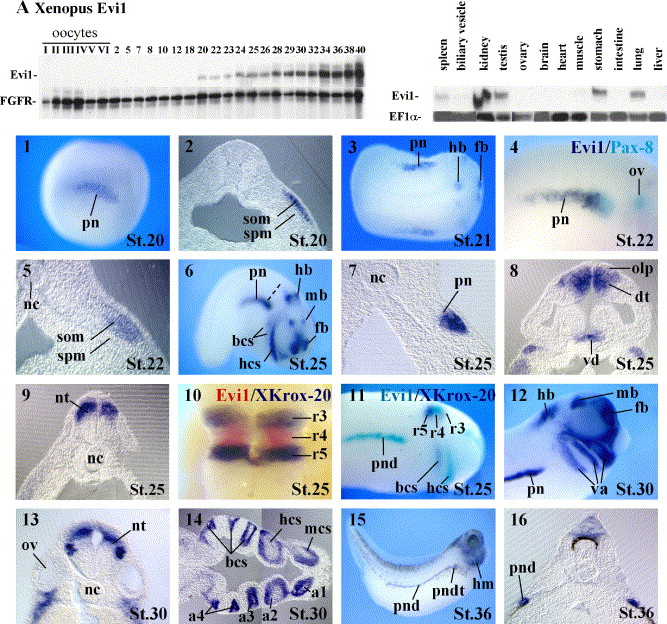
Fig. 2 Embryonic expression pattern of Evi1 and MEL1. (A) Evi1 in Xenopus. Top panels: RNAase protection analysis of Evi1 transcripts during oogenesis and embryogenesis (left) and in adult organs (right). FGFR or EF1α are used as internal controls. Bottom panels: whole-mount in situ hybridization analysis of Evi1 expression. Embryos were fixed at the indicated Nieuwkoop-Faber stages and analyzed with the probes as indicated. (1–5) Expression of Evi1 at late neurula stage. (1) Initial expression of Evi1 is observed in the pronephric mesoderm. (2) Transversal section of the embryo shown in (1) showing restricted Evi1 expression in the somatic layer. (3) Dorsal view of a slightly later stage embryo showing Evi1 expression in selected regions of the developing brain. (4) Comparison of Evi1 (purple) and Pax8 (light blue) expression in the pronephros; note that Evi1 expression is restricted to the ventral posterior part of the Pax8 expression domain. (5) Transversal section at the level of the pronephros. (6–11) Evi1 expression at tailbud stages. (6) Lateral view of an embryo; note Evi1 expression outside the pronephros in the forebrain, midbrain and hindbrain and in cranial neural crest cells. (7) Transversal section of the embryo shown in (6) at the level indicated; note that Evi1 expression can be seen only in the ventral part of the pronephros anlagen. (8) Horizontal section showing Evi1 expression in the dorsal telencephalon, ventral diencephalons and in the olfactory placodes. (9) Transversal section at the level of r4 in the hindbrain; note that only the dorsal part of the neural tube is labeled by the Evi1 probe. (10) High-magnification view of the hindbrain with anterior towards the top double stained with Evi1 (red) and XKrox20 (purple) probes indicating that Evi1 is expressed in r4. (11) Embryo double stained with Evi1 (light blue) and XKrox-20 (purple); note that the Evi1 strongest staining in the migrating neural crest cells is found anteriorly to the XKrox-20 expressing cells. (12–16) Evi1 expression at tadpole stages. (12) High-magnification view of the head; note that Evi1 expression is restricted to the posterior part of the pronephros and that in the hindbrain, its expression initially restricted to r4 has expanded anteriorly and posteriorly. (13) Transversal section at the level of the otic vesicles; note that Evi1 expression can be seen in addition to the dorsal neural tube in a subset of differentiated neurons. (14) Horizontal section through the visceral arches, with anterior towards the right; note Evi1 expression in the neural crest component of the branchial arches. (15) Lateral view of a late tadpole; note that within the pronephros, Evi1 expression is restricted to the distal tubule and duct compartments. (16) Transversal section at the level of the trunk showing Evi1 expressing pronephric duct cells. (B) Whole-mount in situ analysis of Evi1 expression in zebrafish. (1) In 4S stage embryos, strong Evi1 staining is apparent in the telencephalon. (2, 4) Lateral view, dorsal view and transversal section of a 8S stage embryo; note additional Evi1 staining in the intermediate mesoderm and in the head region. (5) Dorsal view of an embryo stained with a Sim1 probe; note that Sim1 expression in the intermediate mesoderm extends further anteriorly than Evi1. (6, 7) Lateral and dorsal view of stages 17S–20S embryos. Note that Evi1 is expressed in various regions of the brain, in head neural crest cells and in the posterior part of the pronephric duct. (8) Lateral view of a pharyngula stage embryo; note that Evi1 expression in the hindbrain has extended posteriorly and that it is also strongly expressed in the pectoral fins. (C) Whole-mount in situ analysis of Evi1 in chick. (1) Dorsal view of a stage 9 embryo showing Evi1 expression in the intermediate mesoderm. (2, 3) Dorsal view and transversal section of a stage 11 embryo showing Evi1 expression in the nephric duct. (4) In 19S embryos, Evi1 expression in the duct has disappeared and strong staining is detected in the limb buds. (D) Whole-mount in situ analysis of MEL1 in Xenopus. (1, 2) Lateral views of early tailbud stage embryos. Note MEL1 expression in cranial crest cells, in the head mesenchyme and in the otic vesicle. (3, 5) High magnification view of the head and transversal sections of early tadpoles. Note that MEL1 staining is now also apparent in various parts of the brain and in the retinal pigment epithelium. (6–8) Lateral view and horizontal sections of late tadpoles. Note that additional MEL1 staining is seen in the heart and in the pronephric distal tubule and duct. Abbreviations: a1–a4, arch 1–4; ba, branchial arches; bcs, branchial crest segment; cnc, cranial neural crest; dd, dorsal diencephalon; dt, dorsal telencephalon; e, eye; fb, forebrain; hb, hindbrain; hcs, hyoid crest segment; h, heart; hm, head mesenchyme; im, intermediate mesoderm; lb, limb buds; mcs, mandibular crest segment; mb, midbrain; nc, notochord; nd, nephric duct; nt, neural tube; olp, olfactory placodes; ov, otic vesicle; pf, pectoral fins; pn, pronephros; pnd, pronephric duct; pndt, pronephric distal tubule; ppnd, posterior pronephric duct; rpe, retinal pigment epithelium; r3–5, rhombomere 3–5; sc, spinal cord; som, somatic lateral plate mesoderm; spm, splanchnic lateral plate mesoderm; tel, telencephalon; tg, tegmentum; va, visceral arches; vd, ventral diencephalon.
Reprinted from Developmental Biology, 294(1), Van Campenhout, C., Nichane, M., Antoniou, A., Pendeville, H., Bronchain, O.J., Marine, J.C., Mazabraud, A., Voz, M.L., and Bellefroid, E.J., Evi1 is specifically expressed in the distal tubule and duct of the Xenopus pronephros and plays a role in its formation, 203-219, Copyright (2006) with permission from Elsevier. Full text @ Dev. Biol.